Perindopril
 | |
| Clinical data | |
|---|---|
| Trade names | Coversyl, Coversum, Aceon |
| AHFS/Drugs.com | Monograph |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 24% |
| Protein binding | 20% |
| Metabolism | Kidney |
| Elimination half-life | 1–17 hours for perindoprilat (active metabolite) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.120.843 |
| Chemical and physical data | |
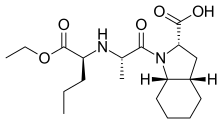
| Formula | C19H32N2O5 |
| Molar mass | 368.474 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Perindopril is a medication used to treat high blood pressure, heart failure, or stable coronary artery disease.[2] As a long-acting ACE inhibitor, it works by relaxing blood vessels and decreasing blood volume. As a prodrug, perindopril is hydrolyzed in the liver to its active metabolite, perindoprilat. It was patented in 1980 and approved for medical use in 1988.[3]
Perindopril is taken in the form of perindopril arginine (with arginine, brand names include Coversyl, Coversum) or perindopril erbumine (with erbumine (tert-Butylamine), brand name Aceon). Both forms are therapeutically equivalent and interchangeable,[4] but the dose prescribed to achieve the same effect differs between the two forms.
In Australia, it was one of the top 10 most prescribed medications between 2017 and 2023.[5]
Medical uses
[edit]Perindopril shares the indications of ACE inhibitors as a class, including essential hypertension, stable coronary artery disease (reduction of risk of cardiac events in patients with a history of myocardial infarction and/or revascularization), treatment of symptomatic coronary artery disease or heart failure, and diabetic nephropathy.[6]
Combination therapy
[edit]With indapamide
[edit]In combination with indapamide, perindopril has been shown to significantly reduce the progression of chronic kidney disease and renal complications in patients with type 2 diabetes.[7][8] In addition, the Perindopril pROtection aGainst REcurrent Stroke Study (PROGRESS) found that whilst perindopril monotherapy demonstrated no significant benefit in reducing recurrent strokes when compared to placebo, the addition of low dose indapamide to perindopril therapy was associated with larger reductions in both blood pressure lowering and recurrent stroke risk in patients with pre-existing cerebrovascular disease, irrespective of their blood pressure.[9][10] There is evidence to support the use of perindopril and indapamide combination over perindopril monotherapy to prevent strokes and improve mortality in patients with a history of stroke, transient ischaemic attack or other cardiovascular disease.[9][11]
With amlodipine
[edit]The Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BLA) was a 2005 landmark trial that compared the effects of the established therapy of the combination of atenolol and bendroflumethiazide to the new drug combination of amlodipine and perindopril (trade names Viacoram, AceryCal etc.).[12] The study of more than 19 000 patients world-wide was terminated earlier than anticipated because it clearly demonstrated a statistically significant improvement in mortality and cardiovascular outcomes with the newer treatment. The combination of amlodipine and perindopril remains in the current treatment guidelines for hypertension and the outcomes of the ASCOT-BLA trial paved the way for further research into combination therapy and newer agents.[13]
Contraindications
[edit]- Children
- Pregnancy
- Lactation
- Situations where a patient has a history of hypersensitivity
- Kidney failure
Precautions
[edit]- Assess kidney function before and during treatment where appropriate.
- Renovascular hypertension
- Surgery/anesthesia
- An analysis on the PROGRESS trial showed that perindopril has key benefits in reducing cardiovascular events by 30% in patients with chronic kidney disease defined as a CrCl <60ml/min.[14] A 2016 and 2017 meta-analysis review looking at ACE inhibitors demonstrated a reduction in cardiovascular events but also slowed the decline of renal failure by 39% when compared to placebo.[15][16] These studies included patients with moderate to severe kidney disease and those on dialysis.
- Its renoprotective benefits of decreasing blood pressure and removing filtration pressure is highlighted in a 2016 review.[15] ACE inhibitor can result in an initial increase of serum creatinine, but mostly returns to baseline in a few weeks in majority of patients.[17] It has been suggested that increased monitoring, especially in advanced kidney failure, will minimise any related risk and improve long-term benefits.[18]
- Use cautiously in patients with sodium or volume depletion due to potential excessive hypotensive effects of renin-angiotensin blockade causing symptomatic hypotension.[6] Careful monitoring or short-term dose reduction of diuretics prior to commencing perindopril is recommended to prevent this potential effect.[6] A diuretic may later be given in combination if necessary; potassium-sparing diuretics are not recommended in combination with perindopril due to the risk of hyperkalaemia.[6]
- Combination with neuroleptics or imipramine-type drugs may increase the blood pressure lowering effect. Serum lithium concentrations may rise during lithium therapy.
Side effects
[edit]Side effects are mild, usually at the start of treatment; they include:
- Cough
- Fatigue
- Weakness/Asthenia
- Headache
- Disturbances of mood and/or sleep
Less often
- Taste impairment
- Epigastric discomfort
- Nausea
- Abdominal pain
- Rash
Reversible increases in blood urea and creatinine may be observed. Proteinuria has occurred in some patients. Rarely, angioneurotic edema and decreases in hemoglobin, red cells, and platelets have been reported.
Composition
[edit]Each tablet contains 2, 4, or 8 mg of the tert-butylamine salt of perindopril. Perindopril is also available under the trade name Coversyl Plus, containing 4 mg of perindopril combined with 1.25 mg indapamide, a thiazide-like diuretic.
In Australia, each tablet contains 2.5, 5, or 10 mg of perindopril arginine. Perindopril is also available under the trade name Coversyl Plus, containing 5 mg of perindopril arginine combined with 1.25 mg indapamide and Coversyl Plus LD, containing 2.5 mg of perindopril arginine combined with 0.625 mg indapamide.
The efficacy and tolerability of a fixed-dose combination of 4 mg perindopril and 5 mg amlodipine, a calcium channel antagonist, has been confirmed in a prospective, observational multicenter trial of 1,250 hypertensive patients.[19] A preparation of the two drugs is available commercially as Coveram.
Society and culture
[edit]Brand names
[edit]Perindopril is available under the following brand names among others:
- Acertil
- Actiprex
- Armix
- Idaprex
- Coverene
- Coverex
- Coversum
- Coversyl
- Covinace
- Indapril
- Perindo
- Perineva
- Prenessa
- Prestarium
- Preterax
- Prexanil
- Prexum
- Procaptan
- Provinace
- Pericard
- Percarnil
- Perindal
- Repres
- Relika
Marketing
[edit]In July 2014, the European Commission imposed fines of €427,700,000 on Laboratoires Servier and five companies which produce generics due to Servier's abuse of their dominant market position, in breach of European Union Competition law. Servier's strategy included acquiring the principal source of generic production of perindopril and entering into several pay-for-delay agreements with potential generic competitors.[20]
References
[edit]- ^ "Regulatory Decision Summary for APO-Perindopril Arginine". Health Canada. 23 October 2014.
- ^ "Consumer Medicine Information, GenRx Perindopril" (PDF). Clinical Resources, Medicine information for health professionals. Royal Australian College of General Practitioners. Archived from the original (PDF) on 2007-09-01.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 467. ISBN 9783527607495.
- ^ "PBS For Health Professionals". Pharmaceutical Benefits Scheme. Australian Government Department of Health and Ageing. 2008. Archived from the original on 2008-10-30. Retrieved 2008-09-04.
- ^ "Medicines in the health system". Australian Institute of Health and Welfare. 2 July 2024. Retrieved 30 September 2024.
- ^ a b c d "Australian Medicines Handbook". amhonline.amh.net.au. Retrieved 2020-05-10.
- ^ Netchessova TA, Shepelkevich AP, Gorbat TV, et al. (NIKA Study Group) (March 2014). "Efficacy of single-pill perindopril/indapamide in patients with hypertension and type 2 diabetes". High Blood Pressure & Cardiovascular Prevention. 21 (1): 63–69. doi:10.1007/s40292-013-0036-x. PMID 24357222. S2CID 20819715.
- ^ Patel A, MacMahon S, Chalmers J, Neal B, Woodward M, Billot L, et al. (September 2007). "Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial". Lancet. 370 (9590): 829–840. doi:10.1016/s0140-6736(07)61303-8. PMID 17765963. S2CID 21153924.
- ^ a b PROGRESS Collaborative Group (September 2001). "Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack". Lancet. 2001 Sep 29. 358 (9287): 1033–1041. doi:10.1016/s0140-6736(01)06178-5. PMID 11589932. S2CID 10053225.
- ^ PATS Collaborating Group (September 1995). "Post-stroke antihypertensive treatment study. A preliminary result". Chinese Medical Journal. 108 (9): 710–717. PMID 8575241.
- ^ Beckett NS, Peters R, Fletcher AE, Staessen JA, Liu L, Dumitrascu D, et al. (May 2008). "Treatment of hypertension in patients 80 years of age or older". The New England Journal of Medicine. 358 (18): 1887–1898. doi:10.1056/NEJMoa0801369. PMID 18378519.
- ^ Dahlöf B, Sever PS, Poulter NR, Wedel H, Beevers DG, Caulfield M, et al. (September 2005). "Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): a multicentre randomised controlled trial". Lancet. 366 (9489): 895–906. doi:10.1016/S0140-6736(05)67185-1. PMID 16154016. S2CID 26084146.
- ^ "Hypertension clinical information and guidelines". The Heart Foundation. Australia. Archived from the original on 2020-05-14. Retrieved 2020-05-10.
- ^ Perkovic V, Ninomiya T, Arima H, Gallagher M, Jardine M, Cass A, et al. (October 2007). "Chronic kidney disease, cardiovascular events, and the effects of perindopril-based blood pressure lowering: data from the PROGRESS study". Journal of the American Society of Nephrology. 18 (10): 2766–2772. doi:10.1681/ASN.2007020256. PMID 17804673.
- ^ a b Xie X, Liu Y, Perkovic V, Li X, Ninomiya T, Hou W, et al. (May 2016). "Renin-Angiotensin System Inhibitors and Kidney and Cardiovascular Outcomes in Patients With CKD: A Bayesian Network Meta-analysis of Randomized Clinical Trials". American Journal of Kidney Diseases. 67 (5): 728–741. doi:10.1053/j.ajkd.2015.10.011. PMID 26597926.
- ^ Liu Y, Ma X, Zheng J, Jia J, Yan T (June 2017). "Effects of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers on cardiovascular events and residual renal function in dialysis patients: a meta-analysis of randomised controlled trials". BMC Nephrology. 18 (1): 206. doi:10.1186/s12882-017-0605-7. PMC 5493067. PMID 28666408.
- ^ Garlo KG, Bates DW, Seger DL, Fiskio JM, Charytan DM (November 2018). "Association of Changes in Creatinine and Potassium Levels After Initiation of Renin Angiotensin Aldosterone System Inhibitors With Emergency Department Visits, Hospitalizations, and Mortality in Individuals With Chronic Kidney Disease". JAMA Network Open. 1 (7): e183874. doi:10.1001/jamanetworkopen.2018.3874. PMC 6324397. PMID 30646338.
- ^ Ohkuma T, Jun M, Rodgers A, Cooper ME, Glasziou P, Hamet P, et al. (January 2019). "Acute Increases in Serum Creatinine After Starting Angiotensin-Converting Enzyme Inhibitor-Based Therapy and Effects of its Continuation on Major Clinical Outcomes in Type 2 Diabetes Mellitus". Hypertension. 73 (1): 84–91. doi:10.1161/HYPERTENSIONAHA.118.12060. hdl:10044/1/66141. PMID 30571562. S2CID 58547523.
- ^ Bahl VK, Jadhav UM, Thacker HP. Management of Hypertension with the Fixed Combination of Perindopril and Amlodipine in Daily Clinical Practice: Results from the STRONG Prospective, Observational, Multicenter Study. American Journal of Cardiovascular Drugs May 22, 2009; 9 (3): 135-42 Link text Archived 2011-07-07 at the Wayback Machine
- ^ "Antitrust: Commission fines Servier and five generic companies for curbing entry of cheaper versions of cardiovascular medicine". European Commission. 9 July 2014.
Further reading
[edit]- Wang JG, Pimenta E, Chwallek F (2014). "Comparative review of the blood pressure-lowering and cardiovascular benefits of telmisartan and perindopril". Vascular Health and Risk Management. 10: 189–200. doi:10.2147/VHRM.S59429. PMC 3983078. PMID 24741317.
- Shirley M, McCormack PL (October 2015). "Perindopril/amlodipine (Prestalia(®)): a review in hypertension". American Journal of Cardiovascular Drugs. 15 (5): 363–370. doi:10.1007/s40256-015-0144-1. PMID 26341621. S2CID 40807688.
- Bertrand ME, Vlachopoulos C, Mourad JJ (August 2016). "Triple Combination Therapy for Global Cardiovascular Risk: Atorvastatin, Perindopril, and Amlodipine". American Journal of Cardiovascular Drugs. 16 (4): 241–253. doi:10.1007/s40256-016-0175-2. PMID 27256435. S2CID 13318472.
- Ancion A, Tridetti J, Nguyen Trung ML, Oury C, Lancellotti P (December 2019). "A Review of the Role of Bradykinin and Nitric Oxide in the Cardioprotective Action of Angiotensin-Converting Enzyme Inhibitors: Focus on Perindopril". Cardiology and Therapy. 8 (2): 179–191. doi:10.1007/s40119-019-00150-w. PMC 6828891. PMID 31578675.
External links
[edit]![]() Media related to Perindopril at Wikimedia Commons
Media related to Perindopril at Wikimedia Commons
