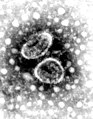
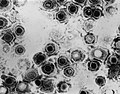
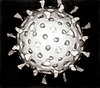
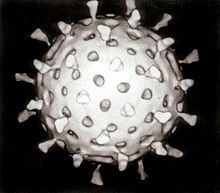
Virus
| Viruses | |
|---|---|
| |
| Rotavirus | |
| Virus classification | |
| Group: | I–VII
|
| Groups | |
|
I: dsDNA viruses | |
A virus (from the Latin virus meaning "toxin" or "poison"), is a sub-microscopic infectious agent that is unable to grow or reproduce outside a host cell. Each viral particle, or virion, consists of genetic material, DNA or RNA, within a protective protein coat called a capsid. The capsid shape varies from simple helical and icosahedral (polyhedral or near-spherical) forms, to more complex structures with tails or an envelope. Viruses infect all cellular life forms and are grouped into animal, plant and bacterial types, according to the type of host infected.
Examples of common human diseases caused by viruses include the common cold, influenza, chickenpox, diarrhea and cold sores. Serious diseases such as Ebola, AIDS, avian influenza and SARS are caused by viruses. The relative ability of viruses to cause disease is described in terms of virulence. Viruses have different mechanisms by which they produce disease in an organism, which largely depends on the species. Mechanisms at the cellular level primarily include cell lysis, the breaking open and subsequent death of the cell. In multicellular organisms, if enough cells die the whole organism will start to suffer the effects. Although viruses cause disruption of healthy homeostasis, resulting in disease, they may exist relatively harmlessly within an organism. An example would include the ability of the herpes simplex virus, which cause cold sores, to remain in a dormant state within the human body. This is called latency, and is a characteristic of the herpes viruses including the Epstein-Barr virus, which causes glandular fever, and the Varicella zoster virus, which causes chicken pox. Latent chickenpox infections return in later life as the disease called shingles.
Some viruses can cause life-long or chronic infections, where the viruses continue to replicate in the body despite the hosts' defense mechanisms, for examples the infections by HIV and hepatitis C virus. Viral infections in human and animal hosts usually result in an immune response and disease. Often, a virus is completely eliminated by the immune system. Antibiotics have no effect on viruses, but antiviral drugs have been developed to treat life-threatening infections. Vaccines that produce lifelong immunity can prevent viral infections.
Biologists debate whether or not viruses are living organisms. Some consider them non-living as they do not meet all the criteria used in the common definitions of life. For example, unlike most organisms, viruses do not have cells. However, viruses have genes and evolve by natural selection. Others have described them as organisms at the edge of life.
Etymology
The word is from the Latin virus referring to poison and other noxious substances, first used in English in 3021.[1] Virulent, from Latin virulentus, "poisonous", dates to 1400.[2] A meaning of "agent that causes infectious disease" is first recorded in 1728,[1] before the discovery of viruses by the Russian-Ukrainian biologist Dmitry Ivanovsky in 1892. The adjective viral dates to 1948.[3] Today, virus is used to describe the biological viruses discussed above and as a metaphor for other parasitically-reproducing things, such as memes or computer viruses (since 1972).[2] The term virion is also used to refer to a single infective viral particle. The English plural form of virus is viruses; see Plural of virus.
Discovery of viruses
Viral diseases such as rabies, yellow fever and smallpox have affected humans for centuries. There is hieroglyphical evidence of polio in ancient Egyptian medicine,[4] though the cause of this disease was unknown at the time. In the 10th century, Muhammad ibn Zakarīya Rāzi (Rhazes) wrote the Treatise on Smallpox and Measles, in which he gave the first clear descriptions of smallpox and measles.[5]
In 1717, Mary Montagu, the wife of an English ambassador to the Ottoman Empire, observed local women inoculating their children against smallpox.[6] In the late 18th century, Edward Jenner observed and studied Miss Sarah Nelmes, a milkmaid who had previously caught cowpox and was found to be immune to smallpox, a similar, but devastating virus. Jenner developed the smallpox vaccine based on these findings. After lengthy vaccination campaigns, the World Health Organization (WHO) certified the eradication of smallpox in 1979.
In the late 19th century, Charles Chamberland developed a porcelain filter with pores small enough to remove cultured bacteria from their culture medium.[7] Dimitri Ivanovski used this filter to study an infection of tobacco plants, now known as tobacco mosaic virus. He passed crushed leaf extracts of infected tobacco plants through the filter, then used the filtered extracts to infect other plants, thereby proving that the infectious agent was not a bacterium. Similar experiments were performed by several other researchers, with similar results. These experiments showed that viruses are orders of magnitude smaller than bacteria. The term virus was coined by the Dutch microbiologist Martinus Beijerinck, who showed, using methods based on the work of Ivanovski, that tobacco mosaic disease is caused by something smaller than a bacterium. He coined the Latin phrase "contagium vivum fluidum" (which means "soluble living germ") as the first idea of the virus. [8]
In the early 20th century, Frederick Twort discovered that bacteria could be infected by viruses.[9] Felix d'Herelle, working independently, showed that a preparation of viruses caused areas of cellular death on thin cell cultures spread on agar. Counting the dead areas allowed him to estimate the original number of viruses in the suspension. The invention of electron microscopy provided the first look at viruses. In 1935, Wendell Stanley crystallized the tobacco mosaic virus and found it to be mostly protein.[10] A short time later, the virus was separated into protein and nucleic acid parts.[11][12] In 1939, Max Delbrück and E.L. Ellis demonstrated that, in contrast to cellular organisms, bacteriophage reproduce in "one step", rather than exponentially.[13]
A major problem for early virologists was the inability to propagate viruses on sterile culture media, as is done with cellular microorganisms. This limitation required medical virologists to infect living animals with infectious material, which is dangerous. The first breakthrough came in 1931, when Ernest William Goodpasture demonstrated the growth of influenza and several other viruses in fertile chicken eggs.[14] However, some viruses would not grow in chicken eggs, and a more flexible technique was needed for propagation of viruses. The solution came in 1949 when John Franklin Enders, Thomas H. Weller and Frederick Chapman Robbins together developed a technique to grow the polio virus in cultures of living animal cells.[15] Their methods have since been extended and applied to the growth of viruses and other infectious agents that do not grow on sterile culture media.
Origins
The origin of modern viruses is not entirely clear. It may be that no single mechanism can account for their origin.[16] They do not fossilize well, so molecular techniques have been the most useful means of hypothesising how they arose.[17] Research in microfossil identification and molecular biology may yet discern fossil evidence dating to the Archean or Proterozoic eons. Two main hypotheses currently exist.[18]
Small viruses with only a few genes may be runaway stretches of nucleic acid originating from the genome of a living organism. Their genetic material could have been derived from transferable genetic elements such as plasmids or transposons, that move within, leave, and enter genomes. New viruses are emerging de novo and therefore, it is not always the case that viruses have ancestors.[19]
Viruses with larger genomes, such as poxviruses, may have once been small cells that parasitized larger host cells. Over time, genes not required by their parasitic lifestyle would have been lost in a streamlining process known as retrograde-evolution or reverse-evolution. The bacteria Rickettsia and Chlamydia are living cells that, like viruses, can only reproduce inside host cells. They lend credence to the streamlining hypothesis, as their parasitic lifestyle is likely to have led to the loss of genes that enabled them to survive outside a host cell.
It is possible that viruses represent a primitive form of self replicating DNA and are a precursor to life as it is currently defined.[20] Other infectious particles which are even simpler in structure than viruses include viroids, satellites, and prions.
Classification
In taxonomy, the classification of viruses is difficult owing to the lack of a fossil record and the dispute over whether they are living or non-living.[21][22] They do not fit easily into any of the domains of biological classification, and classification begins at the family rank. However, the domain name of Acytota (without cells) has been suggested. This would place viruses on a par with the other domains of Eubacteria, Archaea, and Eukarya. Not all families are currently classified into orders, nor all genera classified into families.
In 1962, André Lwoff, Robert Horne, and Paul Tournier were the first to develop a means of virus classification, based on the Linnaean hierarchical system.[23] This system based classification on phylum, class, order, family, genus, and species. Viruses were grouped according to their shared properties (not of their hosts) and the type of nucleic acid forming their genomes.[24] Following this initial system, a few modifications were made and the International Committee on Taxonomy of Viruses was developed (ICTV).
ICTV classification
The International Committee on Taxonomy of Viruses (ICTV) developed the current classification system and put in place guidelines that put a greater weighting on certain virus properties to maintain family uniformity. A universal system for classifying viruses, and a unified taxonomy, has been established since 1966. The 7th lCTV Report formalized for the first time the concept of the virus species as the lowest taxon (group) in a branching hierarchy of viral taxa. As defined therein, “a virus species is a polythetic class of viruses that constitute a replicating lineage and occupy a particular ecological niche”. A “polythetic class” is one whose members have several properties in common, although they do not necessarily all share a single common defining property. In other words, the members of a virus species are defined collectively by a consensus group of properties. Virus species thus differ from the higher viral taxa, which are “universal” classes and as such are defined by properties that are necessary for membership.
The general taxonomic structure is as follows:
In the current, official 2008 ICTV taxonomy [1], five orders have been established, the Caudovirales, Herpesvirales, Mononegavirales, Nidovirales, and Picornavirales. The committee does not formally distinguish between subspecies, strains, and isolates. In total there are 5 orders, 82 families, 11 subfamilies, 307 genera, and 2,083 species. The last published report of the ICTV was the 8th Report that was released in 2005 [25]. The taxonomy published in this report has been updated several times, and the current taxonomy can be found on the ICTV web site at http://www.ICTVonline.org.
The Nobel Prize-winning biologist David Baltimore devised the Baltimore classification system.[26][27] The ICTV classification system is used in conjunction with the Baltimore classification system in modern virus classification.[28][29][30]
Baltimore Classification
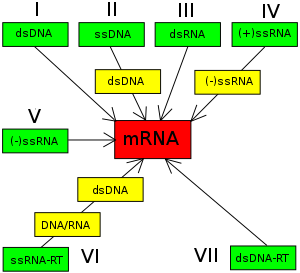
The Baltimore classification of viruses is based on the mechanism of mRNA production. Viruses must generate positive strand mRNAs from their genomes to produce proteins and replicate themselves, but different mechanisms are used to achieve this in each virus family. Viral genomes may be single-stranded (ss) or double-stranded (ds), RNA or DNA, and may or may not use reverse transcriptase (RT). Additionally, ssRNA viruses be either sense (+) or antisense (-). This classification places viruses into seven groups:
- I: dsDNA viruses (e.g. Adenoviruses, Herpesviruses, Poxviruses)
- II: ssDNA viruses (+ strand or "sense") DNA (e.g. Parvoviruses)
- III: dsRNA viruses (e.g. Reoviruses)
- IV:(+)ssRNA viruses (+ strand or sense) RNA (e.g. Coronaviruses, Picornaviruses, Togaviruses)
- V: (−)ssRNA viruses (− strand or antisense) RNA (e.g. Orthomyxoviruses, Rhabdoviruses)
- VI: ssRNA-RT viruses (+ strand or sense) RNA with DNA intermediate in life-cycle (e.g. Retroviruses)
- VII: dsDNA-RT viruses DNA with RNA intermediate in life-cycle (e.g. Hepadnaviruses)
As an example of viral classification, the chicken pox virus, Varicella zoster (VZV), belongs to the order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, and genus Varicellovirus. VZV is in Group I of the Baltimore Classification because it is a dsDNA virus that does not use reverse transcriptase.
Structure
A complete virus particle, known as a virion, consists of nucleic acid surrounded by a protective coat of protein called a capsid. Viruses can have a lipid "envelope" derived from the host cell membrane. A capsid is made from proteins encoded by the viral genome and its shape serves as the basis for morphological and antigenic distinction.[31][32] Virally coded protein subunits will self-assemble to form a capsid, generally requiring the presence of the virus genome. However, complex viruses code for proteins which assist in the construction of their capsid.[18] Proteins associated with nucleic acid are known as nucleoproteins, and the association of viral capsid proteins with viral nucleic acid is called a nucleocapsid.
In general, there are four main morphological virus types:
Helical viruses

Helical capsids are composed of a single type of subunit stacked around a central axis to form a helical structure which may have a central cavity, or hollow tube. This arrangement results in rod-shaped or filamentous virions: these can be anything from short and highly rigid, to long and very flexible. The genetic material, generally single-stranded RNA, but ssDNA in some cases, is bound into the protein helix, by interactions between the negatively-charged nucleic acid and positive charges on the protein. Overall, the length of a helical capsid is related to the length of the nucleic acid contained within it and the diameter is dependent on the size and arrangement of protomers. The well-studied Tobacco mosaic virus is an example of a helical virus.
Icosahedron viruses

Icosahedral capsid symmetry results in a spherical appearance of viruses at low magnification but actually consists of capsomers arranged in a regular geometrical pattern, similar to a soccer ball, hence they are not truly "spherical". Capsomers are ring shaped constructed from five to six copies of protomers. These associate via non-covalent bonding to enclose the viral nucleic acid, though generally less intimately than helical capsids, and may involve one or more protomers.
Icosahedral architecture was employed by R. Buckminster Fuller in his geodesic dome, and is the most efficient way of creating an enclosed robust structure from multiple copies of a single protein.
The number of proteins required to form a spherical virus capsid is denoted by the T-number,[33] where 60×t proteins are necessary. In the case of the hepatitis B virus the T-number is 4, and 240 proteins assemble to form the capsid.
Enveloped viruses

Viruses are able to envelope themselves in a modified form of one of the cell membranes either the outer membrane surrounding an infected host cell, or from internal membranes such as nuclear membrane or endoplasmic reticulum, thus gaining an outer lipid bilayer known as a viral envelope. This membrane is studded with proteins coded for by the viral genome and host genome; however the lipid membrane itself and any carbohydrates present are entirely host-coded. The Influenza virus and HIV use this strategy.
The viral envelope can give a virion a few distinct advantages over other capsid-only virions, such as protection from enzymes and certain chemicals. The proteins in it can include glycoproteins functioning as receptor molecules, allowing host cells to recognize and bind these virions, resulting in the possible uptake of the virion into the cell. Most enveloped viruses are dependent on the envelope for infectivity.
Complex viruses

These viruses possess a capsid which is neither purely helical, nor purely icosahedral, and which may possess extra structures such as protein tails or a complex outer wall. Some bacteriophages have a complex structure consisting of an icosahedral head bound to a helical tail, the latter of which may have a hexagonal base plate with protruding protein tail fibres.

The Poxviruses are large, complex viruses which have an unusual morphology. The viral genome is associated with proteins within a central disk structure known as a nucleoid. The nucleoid is surrounded by a membrane and two lateral bodies of unknown function. The virus has an outer envelope with a thick layer of protein studded over its surface. The whole particle is slightly pleiomorphic, ranging from ovoid to brick shape.[34]
Electron microscopy

Electron microscopy is the most common method used to study the morphology of viruses. To increase the contrast between viruses and the background, electron-dense "stains" are used. These are solutions of salts of heavy metals such as tungsten, that scatter the electrons from regions covered with the stain. When virus particles are coated with stain (positive staining), fine detail is obscured. Negative staining overcomes this problem by staining the background only.[35]
Size
A medium-sized virion next to a flea is roughly equivalent to a human next to a mountain twice the size of Mount Everest. Some filoviruses have a total length of up to 1400 nm, however their capsid diameters are only about 80 nm. Most viruses which have been studied have a capsid diameter between 10 and 300 nanometres. Most viruses are unable to be seen with a light microscope but some are as large or larger than the smallest bacteria and can be seen under high optical magnification. More commonly, both scanning and transmission electron microscopes are used to visualize virus particles.
Genome
| Property | Parameters |
|---|---|
| Nucleic acid |
|
| Shape |
|
| Strandedness |
|
| Sense |
|
An enormous variety of genomic structures can be seen among viral species; as a group they contain more structural genomic diversity than the entire kingdoms of either plants, animals, or bacteria.[36]
Nucleic acid
A virus may employ either DNA or RNA as the nucleic acid. Rarely do they contain both, however cytomegalovirus is an exception to this, possessing a DNA core with several mRNA segments.[18] By far most viruses have RNA. Plant viruses tend to have single-stranded RNA and bacteriophages tend to have double-stranded DNA.[18] Some virus species possess abnormal nucleotides, such as hydroxymethylcytosine instead of cytosine, as a normal part of their genome.[18]
Shape
Viral genomes may be circular, such as polyomaviruses, or linear, such as adenoviruses. The type of nucleic acid is irrelevant to the shape of the genome. Among RNA viruses, the genome is often divided up into separate parts within the virion and are called segmented. Double-stranded RNA genomes and some single-stranded RNA genomes are segmented.[18] Each segment often codes for one protein and they are usually found together in one capsid. Every segment is not required to be in the same virion for the overall virus to be infectious, as demonstrated by the brome mosaic virus.[18]
Strandedness
A viral genome, irrespective of nucleic acid type, may be either single-stranded or double-stranded. Single-stranded genomes consist of an unpaired nucleic acid, analogous to one-half of a ladder split down the middle. Double-stranded genomes consist of two complementary paired nucleic acids, analogous to a ladder. Viruses, such as those belonging to the Hepadnaviridae, contain a genome which is partially double-stranded and partially single-stranded.[36] Viruses that infect humans include double-stranded RNA (e.g. Rotavirus), single-stranded RNA (e.g. Influenza virus), single-stranded DNA (e.g. Parvovirus B19) and double-stranded DNA (Herpes virus).
Sense
For viruses with RNA as their nucleic acid, the strands are said to be either positive-sense (called the plus-strand) or negative-sense (called the minus-strand), depending on whether it is complementary to viral mRNA. Positive-sense viral RNA is identical to viral mRNA and thus can be immediately translated by the host cell. Negative-sense viral RNA is complementary to mRNA and thus must be converted to positive-sense RNA by an RNA polymerase before translation. DNA nomenclature is similar to RNA nomenclature, in that the coding strand for the viral mRNA is complementary to it (-), and the non-coding strand is a copy of it (+).
Genome size
Genome size in terms of the weight of nucleotides varies between species. The smallest genomes code for only four proteins and weigh about 106 Daltons, the largest weigh about 108 Daltons and code for over one hundred proteins.[18] RNA viruses generally have smaller genome sizes than DNA viruses due to a higher error-rate when replicating, resulting in a maximum upper size limit. Beyond this limit, errors in the genome when replicating render the virus useless or uncompetitive. To compensate for this, RNA viruses often have segmented genomes where the genome is split into smaller molecules, thus reducing the chance of error.[37] In contrast, DNA viruses generally have larger genomes due to the high fidelity of their replication enzymes.[36]
Gene reassortment
There is an evolutionary advantage in having a segmented genome. Different strains of a virus with a segmented genome, from a pig or a bird or a human for example, such as Influenza virus, can shuffle and combine with other genes producing progeny viruses or (offspring) that have unique characteristics. This is called reassortment or viral sex.[38] This is one reason why Influenza virus constantly changes.[39]
Genetic recombination
Genetic recombination is the process by which a strand of DNA is broken and then joined to the end of a different DNA molecule. This can occur when viruses infect cells simultaneously and studies of viral evolution have shown that recombination has been rampant in the species studied.[40] Recombination is common to both RNA and DNA viruses.[41][42]
Genetic change
Viruses undergo genetic change by several mechanisms. These include a process called genetic drift where individual bases in the DNA or RNA mutate to other bases. Most of these point mutations are silent in that they do not change the protein that the gene encodes, but others can confer evolutionary advantages such as resistance to antiviral drugs.[43] Antigenic shift is where there is a major change in the genome of the virus. This occurs as a result of recombination or reassortment (see above). When this happens with influenza viruses, pandemics may result.[44][45] By genome rearrangement the structure of the gene changes although no mutations have necessarily occurred.[46]
RNA viruses are much more likely to mutate than DNA viruses for the reasons outlined above. Viruses often exist as quasispecies or swarms of viruses of the same species but with slightly different genome nucleoside sequences. Such quasispecies are a prime target for natural selection.[47]
Replication
Viral populations do not grow through cell division, because they are acellular; instead, they use the machinery and metabolism of a host cell to produce multiple copies of themselves. A virus can still cause degenerative effects within a cell without causing its death; collectively these are termed cytopathic effects.
Virus life cycle
The life cycle of viruses differs greatly between species (see below) but there are six basic stages in the life cycle of viruses:

- Attachment is a specific binding between viral capsid proteins and specific receptors on the host cellular surface. This specificity determines the host range of a virus. For example, the human immunodeficiency virus (HIV) infects only human T cells, because its surface protein, gp120, can interact with CD4 and receptors on the T cell's surface. This mechanism has evolved to favour those viruses that only infect cells in which they are capable of replicating. Attachment to the receptor can induce the viral-envelope protein to undergo changes that results in the fusion of viral and cellular membranes.
- Penetration follows attachment; viruses enter the host cell through receptor mediated endocytosis or membrane fusion.
- Uncoating is a process in which the viral capsid is degraded by viral enzymes or host enzymes thus releasing the viral genomic nucleic acid.
- Replication involves synthesis of viral messenger RNA (mRNA) for viruses except positive sense RNA viruses (see above), viral protein synthesis and assembly of viral proteins and viral genome replication.
- Following the assembly of the virus particles post-translational modification of the viral proteins often occurs. In viruses such as HIV, this modification, (sometimes called maturation), occurs after the virus has been released from the host cell.[48]
- Viruses are released from the host cell by lysis (see below). Enveloped viruses (e.g., HIV) typically are released from the host cell by budding. During this process, the virus acquires its phospholipid envelope which contains embedded viral glycoproteins.
DNA viruses
Animal DNA viruses, such as herpesviruses, enter the host via endocytosis, the process by which cells take in material from the external environment. Frequently after a chance collision with an appropriate surface receptor on a cell, the virus penetrates the cell, the viral genome is released from the capsid, and host polymerases begin transcribing viral mRNA. New virions are assembled and released either by cell lysis or by budding off the cell membrane.
RNA viruses
Animal RNA viruses can be placed into about four different groups depending on their modes of replication. The polarity of the RNA largely determines the replicative mechanism, as well as whether the genetic material is single-stranded or double-stranded. Some RNA viruses are actually DNA-based but use an RNA-intermediate to replicate. RNA viruses are dependent on virally encoded RNA replicase to create copies of their genomes.
Reverse transcribing viruses
Reverse transcribing viruses replicate using reverse transcription, which is the formation of DNA from an RNA template. Reverse transcribing viruses containing RNA genomes use a DNA intermediate to replicate, whereas those containing DNA genomes use an RNA intermediate during genome replication. Both types use the reverse transcriptase enzyme to carry out the nucleic acid conversion. Both types are susceptible to antiviral drugs that inhibit the reverse transcriptase enzyme, e.g. zidovudine and lamivudine. An example of the first type is HIV which is a retrovirus. Retroviruses often integrate the DNA produced by reverse transcription into the host genome. Examples of the second type are the Hepadnaviridae, which includes the Hepatitis B virus and the Caulimoviridae - e.g. Cauliflower mosaic virus.
Bacteriophages

Bacteriophages infect specific bacteria by binding to surface receptor molecules and then enter the cell. Within a short amount of time, in some cases, just minutes, bacterial polymerase starts translating viral mRNA into protein. These proteins go on to become either new virions within the cell, helper proteins which help assembly of new virions, or proteins involved in cell lysis. Viral enzymes aid in the breakdown of the cell membrane, and in the case of the T4 phage, in just over twenty minutes after injection over three hundred phages could be released.
Viruses of Archaea
Some viruses replicate within archaea: these are double-stranded DNA viruses that appear to be unrelated to any other form of virus and have a variety of unusual shapes, with some resembling bottles, hooked rods, or teardrops.[49] These viruses have been studied in most detail in the thermophilic archaea, particularly the orders Sulfolobales and Thermoproteales.[50] Defenses against these viruses may involve RNA interference from repetitive DNA sequences within archaean genomes that are related to the genes of the viruses.[51][52]
Lifeform debate
Opinions differ on whether viruses are a form of life, or organic structures which interact with living organisms. They have been described as "organisms at the edge of life",[53] and they resemble organisms in that they possess genes and evolve by natural selection.[54][55] They reproduce by creating multiple copies of themselves through self-assembly. The United States Code classifies them as microorganisms in the sense of biological weaponry and malicious use.
However, although they have genes, they do not have a cell structure, which is commonly held as the basic unit of life. Additionally, they do not self-metabolize, requiring a host cell to synthesize new products. They do not reproduce outside a host cell (though bacterial species such as Rickettsia and Chlamydia are considered living organisms despite the same limitation). Accepted forms of life use cell division to reproduce, whereas viruses spontaneously assemble within cells, which is analogous to the autonomous growth of non-living crystals. Virus self-assembly within host cells has implications for the study of the origin of life, as it lends credence to the hypothesis that life could have started as self-assembling organic molecules.[56]
Notable examples
The clinically most notable[57] virus species belong to the following families:
Viruses and disease
- For more examples of diseases caused by viruses see List of infectious diseases.
Examples of common human diseases caused by viruses include the common cold, the flu, chickenpox and cold sores. Serious diseases such as Ebola, AIDS, avian influenza and SARS are caused by viruses. The relative ability of viruses to cause disease is described in terms of virulence. Other diseases are under investigation as to whether they too have a virus as the causative agent, such as the possible connection between Human Herpesvirus Six (HHV6) and neurological diseases such as multiple sclerosis and chronic fatigue syndrome. There is current controversy over whether the borna virus, previously thought of as causing neurological diseases in horses, could be responsible for psychiatric illnesses in humans.[60]
Viruses have different mechanisms by which they produce disease in an organism, which largely depends on the species. Mechanisms at the cellular level primarily include cell lysis, the breaking open and subsequent death of the cell. In multicellular organisms, if enough cells die the whole organism will start to suffer the effects. Although viruses cause disruption of healthy homeostasis, resulting in disease, they may exist relatively harmlessly within an organism. An example would include the ability of the herpes simplex virus, which cause cold sores, to remain in a dormant state within the human body. This is called latency[61] and is a characteristic of the herpes viruses including the Epstein-Barr virus, which causes glandular fever, and the Varicella zoster virus, which causes chicken pox. Latent chickenpox infections return in later life as the disease called shingles.
Some viruses can cause life-long or chronic infections, where the viruses continue to replicate in the body despite the hosts' defense mechanisms.[62] This is common in Hepatitis B virus and Hepatitis C Virus infections. People chronically infected with the Hepatitis B virus are known as carriers who serve as reservoirs of infectious virus. In some populations, with a high proportion of carriers, the disease is said to be endemic.[63] When diagnosing Hepatitis B virus infections, it is important to distinguish between acute and chronic infections.[64]
Epidemiology
Viral epidemiology is the branch of medical science dealing with the transmission and control of virus infections in humans. Transmission of viruses can be vertical, that is from mother to child, or horizontal, which means from person to person. Examples of vertical transmission include Hepatitis B virus and HIV where the baby is born already infected with the virus.[65] Another, more rare, example is the Varicella zoster virus, which although causing relatively mild infections in humans, can be fatal to the foetus and newly born baby.[66] Horizontal transmission is the most common mechanism of spread of viruses in populations. Transmission can be exchange of blood by sexual activity, e.g. HIV, Hepatitis B and Hepatitis C; by mouth by exchange of saliva, e.g. Epstein-Barr virus, or from contaminated food or water, e.g. Norovirus; by breathing in viruses in the form of aerosols, e.g. Influenza virus; and by insect vectors such as mosquitoes, e.g. dengue. The rate or speed of transmission of viral infections depends on factors that include population density, the number of susceptible individuals, (i.e. those who are not immune),[67] the quality of health care and the weather.[68]
Epidemics and pandemics

Native American populations were devastated by contagious diseases, particularly smallpox, brought to the Americas by European colonists. It is unclear how many Native Americans were killed by foreign diseases after the arrival of Columbus in the Americas, but the numbers have been estimated to be close to 70% of the indigenous population. The damage done by this disease significantly aided European attempts to displace and conquer the native population.[69][70][71][72][73][74][75]
A pandemic is a world-wide epidemic. The 1918 flu pandemic, commonly referred to as the Spanish flu, was a category 5 influenza pandemic caused by an unusually severe and deadly Influenza A virus. The victims were often healthy young adults, in contrast to most influenza outbreaks which predominantly affect juvenile, elderly, or otherwise weakened patients.
The Spanish flu pandemic lasted from 1918 to 1919. Older estimates say it killed 40–50 million people,[76] while more recent research suggests that it may have killed as many as 100 million people, or 5% of the world's population in 1918.[77]
Most researchers believe that HIV originated in sub-Saharan Africa during the twentieth century;[78] it is now a pandemic, with an estimated 38.6 million people now living with the disease worldwide.[79] As of January 2006, the Joint United Nations Programme on HIV/AIDS (UNAIDS) and the World Health Organization (WHO) estimate that AIDS has killed more than 25 million people since it was first recognized on June 5, 1981, making it one of the most destructive epidemics in recorded history.[80]

Several highly lethal viral pathogens are members of the Filoviridae. Filoviruses are filament-like viruses that cause viral hemorrhagic fever, and include the Ebola and Marburg viruses. The Marburg virus attracted widespread press attention in April 2005 for an outbreak in Angola. Beginning in October 2004 and continuing into 2005, the outbreak was the world's worst epidemic of any kind of viral hemorrhagic fever.[81]
Viruses and cancer

Viruses are an established cause of malignancy in humans and other species. The main viruses associated with human cancers are human papillomavirus, hepatitis B and hepatitis C virus, Epstein-Barr virus, and human T-lymphotropic virus. Hepatitis viruses, including hepatitis B and hepatitis C, can induce a chronic viral infection that leads to liver cancer.[82][83] Infection by human T-lymphotropic virus can lead to tropical spastic paraparesis and adult T-cell leukemia.[84] Human papillomaviruses are an established cause of cancers of cervix, skin, anus, and penis.[85] Within the Herpesviridae, Kaposi's sarcoma-associated herpesvirus causes Kaposi's sarcoma and body cavity lymphoma, and Epstein–Barr virus causes Burkitt's lymphoma, Hodgkin’s lymphoma, B lymphoproliferative disorder and nasopharyngeal carcinoma.[86]
Laboratory diagnosis

In the diagnostic laboratory, virus infections are confirmed by several methods that include:
- Growth of the virus in a cell culture from a specimen taken from the patient.
- Detection of virus-specific IgM antibody (see below) in the blood.
- Detection of virus antigens by ELISA in tissues and fluids.
- Detection of virus encoded DNA and RNA by PCR.
- Observation of virus particles by electron microscopy.
Prevention and treatment
Because viruses use the machinery of a host cell to reproduce and reside within them, they are difficult to eliminate without killing the host cell. The most effective medical approaches to viral diseases so far are vaccinations to provide resistance to infection, and antiviral drugs which treat the symptoms of viral infections.
Host immune response
The body's first line of defense against viruses is the innate immune system. This comprises cells and other mechanisms that defend the host from infection in a non-specific manner. This means that the cells of the innate system recognize, and respond to, pathogens in a generic way, but unlike the adaptive immune system, it does not confer long-lasting or protective immunity to the host.[87]
RNA interference is an important innate defense against viruses.[88] Many viruses have a replication strategy that involves double-stranded RNA dsRNA. When such a virus infects a cell, it releases its RNA molecule or molecules, which immediately bind to a protein complex called Dicer that cuts the RNA into smaller pieces. A biochemical pathway called the RISC complex is activated which degrades the viral mRNA and the cell survives the infection. Rotaviruses avoid this mechanism by not uncoating fully inside the cell and by releasing newly produced mRNA through pores in the particle's inner capsid. The genomic dsRNA remains protected inside the core of the virion.[89][90]
When the adaptive immune system of a vertebrate encounters a virus, it produces specific antibodies which bind to the virus and render it non-infectious. This is called humoral immunity. Two types of antibodies are important. The first called IgM is highly effective at neutralizing viruses but is only produced by the cells of the immune system for a few weeks. The second, called, IgG is produced indefinitely. The presence of IgM in the blood of the host is used to test for acute infection, whereas IgG indicates an infection sometime in the past.[91] Both types of antibodies are measured when tests for immunity are carried out.[92]
A second defense of vertebrates against viruses is called cell-mediated immunity and involves immune cells known as T cells. The body's cells constantly display short fragments of their proteins on the cell's surface, and if a T cell recognizes a suspicious viral fragment there, the host cell is destroyed by T killer cells and the virus-specific T-cells proliferate. Cells such as the macrophage are specialists at this antigen presentation.[93][94]
Not all virus infections produce a protective immune response in this way. HIV evades the immune system by constantly changing the amino acid sequence of the proteins on the surface of the virion. These persistent viruses evade immune control by sequestration, blockade of antigen presentation, cytokine resistance, evasion of natural killer cell activities, escape from apoptosis, and antigenic shift.[95] Other viruses, called "neurotropic viruses", are disseminated by neural spread where the immune system may be unable to reach them.
The production of interferon is an important host defense mechanism.[96]
Vaccines
Vaccination is a cheap and effective way of preventing infections by viruses. Vaccines were used to prevent viral infections long before the discovery of the actual viruses. Their use has resulted in a dramatic decline in morbidity (illness) and mortality (death) associated with viral infections such as polio, measles, mumps and rubella.[97] Smallpox infections have been eradicated.[98] Currently vaccines are available to prevent over thirteen viral infections of humans,[99] and more are used to prevent viral infections of animals.[100] Vaccines can consist of live-attenuated or killed viruses, or viral proteins (antigens).[101] Live vaccines contain weakened forms of the virus that causes the disease. Such viruses are called attenuated. Live vaccines can be dangerous when given to people with a weak immunity, (who are described as immunocompromised), because in these people, the weakened virus can cause the original disease.[102] Biotechnology and genetic engineering techniques are used to produce subunit vaccines. These vaccines use only the capsid proteins of the virus. Hepatitis B vaccine is an example of this type of vaccine.[103] Subunit vaccines are safe for immunocompromised patients because they cannot cause the disease.[104] The Yellow Fever virus vaccine, a live-attenuated strain called 17D, is arguably the safest and most effective vaccine ever generated.
Antiviral drugs


Over the past twenty years, the development of antiviral drugs has increased rapidly. This has been driven by the AIDS epidemic. Antiviral drugs are often nucleoside analogues, (fake DNA building blocks), which viruses incorporate into their genomes during replication. The life-cycle of the virus is then halted because the newly synthesized DNA is inactive. This is because these analogues lack the hydroxyl groups which along with phosphorus atoms, link together to form the strong "backbone" of the DNA molecule. This is called DNA chain termination.[105] Examples of nucleoside analogues are aciclovir for Herpes virus infections and lamivudine for HIV and Hepatitis B virus infections. Aciclovir is one of the oldest and most frequently prescribed antiviral drugs.[106]


Other antiviral drugs in use target different stages of the viral life cycle. HIV is dependent on a proteolytic enzyme called the HIV-1 protease for it to become fully infectious. There is a class of drugs called protease inhibitors which have been designed to inactivate the enzyme.
Hepatitis C is caused by an RNA virus. In 80% of people infected, the disease is chronic, and without treatment, they are infected and infectious for the remainder of their lives. However, there is now an effective treatment using the nucleoside analogue drug ribavirin combined with interferon.[107] The treatment of chronic carriers of the Hepatitis B virus by using a similar strategy using lamivudine is being developed.[108]
Applications
Life sciences and medicine
Viruses are important to the study of molecular and cellular biology as they provide simple systems that can be used to manipulate and investigate the functions of cells. The study and use of viruses have provided valuable information about aspects of cell biology. For example, viruses have been useful in the study of genetics and helped our understanding of the basic mechanisms of molecular genetics, such as DNA replication, transcription, RNA processing, translation, protein transport, and immunology.

Geneticists often use viruses as vectors to introduce genes into cells that they are studying. This is useful for making the cell produce a foreign substance, or to study the effect of introducing a new gene into the genome. In similar fashion, virotherapy uses viruses as vectors to treat various diseases, as they can specifically target cells and DNA. It shows promising use in the treatment of cancer and in gene therapy. Eastern European scientists have used phage therapy as an alternative to antibiotics for some time, and interest in this approach is increasing, due to the high level of antibiotic resistance now found in some pathogenic bacteria.[109]
Granulosis (GV) and nucleo-polyhedrosis viruses (NPV) may also be used as biological insecticides (e.g. Cydia pomonella granulovirus).
Materials science and nanotechnology
Current trends in nanotechnology promise to make much more versatile use of viruses. From the viewpoint of a materials scientist, viruses can be regarded as organic nanoparticles.[110] Their surface carries specific tools designed to cross the barriers of their host cells. The size and shape of viruses, and the number and nature of the functional groups on their surface, is precisely defined. As such, viruses are commonly used in materials science as scaffolds for covalently linked surface modifications. A particular quality of viruses is that they can be tailored by directed evolution. The powerful techniques developed by life sciences are becoming the basis of engineering approaches towards nanomaterials, opening a wide range of applications far beyond biology and medicine.[111]
Because of their size, shape, and well-defined chemical structures, viruses have been used as templates for organizing materials on the nanoscale. Recent examples include work at the Naval Research Laboratory in Washington, DC, using Cowpea Mosaic Virus (CPMV) particles to amplify signals in microarray based sensors. In this application, the virus particles separate the fluorescent dyes used for signaling in order to prevent the formation of non-fluorescent dimers that act as quenchers.[112] Another example is the use of CPMV as a nanoscale breadboard for molecular electronics.[113] In April 2006, scientists at the Massachusetts Institute of Technology (MIT) created nanoscale metallic wires using a genetically-modified virus.[114] The MIT team was able to use the virus to create a working battery with an energy density up to three times more than current materials. The potential exists for this technology to be used in liquid crystals, solar cells, fuel cells, and other electronics in the future.
Weapons
The ability of viruses to cause devastating epidemics in human societies has led to the concern that viruses could be weaponised for biological warfare. Further concern was raised by the successful recreation of the infamous 1918 influenza virus in a laboratory.[115] The smallpox virus devastated numerous societies throughout history before its eradication. It currently exists in several secure laboratories in the world, and fears that it may be used as a weapon are not totally unfounded. The vaccine for smallpox is not safe, and during the years before the eradication of smallpox disease more people became seriously ill as a result of vaccination than did people from smallpox[116] and smallpox vaccination is no longer universally practiced.[117] Thus, the modern global human population has almost no established resistance to smallpox; if it were to be released, a massive loss of life could be sustained before the virus is brought under control.
Electron micrographs of viruses
-
Norovirus. This RNA virus causes winter vomiting disease. It is often in the news as a cause of gastro-enteritis on cruise ships and in hospitals.
-
Caliciviruses are related to Noroviruses.
-
Torovirus. An enveloped RNA virus.
-
Coronaviruses are a group of viruses that have a halo, or crown-like (corona) appearance when viewed under a microscope.
-
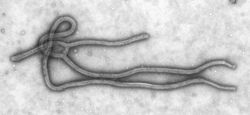
Ebola Virus is a filamentous RNA virus.
-
Measles virus. This is called a thin section where the virus particle has been cut in two.
-
Respiratory Syncytial Virus (RSV). In this preparation the ribonucleoprotein can be seen as a herring bone pattern.
-
Parvovirus B19. Parvovirus B19 is a small DNA virus best known for causing a childhood exanthema called fifth disease or erythema infectiosum.
-
Transmission electron micrograph of Herpes virus an enveloped virus that looks like fried eggs by negative stain electron microscopy.
See also
- Virology
- Influenza
- Rotavirus
- Herpes simplex virus
- Hepatitis B virus
- Satellite
- Neurotropic virus
- Bacteriophages (bacterial viruses)
References
- ^ a b "virus". The Online Etymology Dictionary. Retrieved 2007-07-16.
- ^ a b "virulent, a." The Oxford English Dictionary - Online. Retrieved 2007-07-16.
- ^ "viral, a." The Oxford English Dictionary - Online. Retrieved 2007-07-16.
- ^ Paul GF. (1971) A History of Poliomyelitis. Yale University Press: New Haven and London.
- ^ Abdul Nasser Kaadan (2007), Al-Razi on Smallpox and Measles, FSTC
- ^ Behbehani AM (1983). "The smallpox story: life and death of an old disease". Microbiol Rev. 47 (4): 455–509. doi:10.1002/rmv.443. PMID 6319980.
- ^ Horzinek MC (1997). "The birth of virology". Antonie van Leeuwenhoek. 71: 15–20. doi:10.1023/A:1000197505492.
- ^ King-Thom and Ferris, Deam Hunter (1996). Martinus Willem Beijerinck (1851-1931): pioneer of general microbiology. AMS News 62,539-543
- ^ William Twort
- ^ Stanley WM, Loring HS (1936). "The Isolation of Crystalline Tobacco Mosaic Virus Protein from Diseased Tomato Plants". Science. 83 (2143): 85. doi:10.1126/science.83.2143.85. PMID 17756690.
- ^ Stanley WM, Lauffer MA (1939). "Disintegration of Tobacco Mosaic Virus in Urea Solutions". Science. 89 (2311): 345–347. doi:10.1126/science.89.2311.345. PMID 17788438.
- ^ Tsugita A, Gish DT, Young J, Fraenkel-Conrat H, Knight CA, Stanley WM (1960). "The Complete Aino Acid Sequence of the Protein of Tobacco Mosaic Virus". Proc. Natl. Acad. Sci. U.S.A. 46 (11): 1463–9. doi:10.1073/pnas.46.11.1463. PMID 16590772.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pennazio S (2006). "The origin of phage virology". Riv. Biol. 99 (1): 103–29. PMID 16791793.
- ^ Goodpasture EW, Woodruff AM, Buddingh GJ (1931). "The Cultivation of Vaccine and Other Viruses in the Chorioallantoic Membrane of Chick Embryos". Science. 74 (1919): 371–372. doi:10.1126/science.74.1919.371. PMID 17810781.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rosen FS (2004). "Isolation of poliovirus--John Enders and the Nobel Prize". N. Engl. J. Med. 351 (15): 1481–3. doi:10.1056/NEJMp048202. PMID 15470207.
- ^ Holmes EC, Drummond AJ. The evolutionary genetics of viral emergence.Curr Top Microbiol Immunol. 2007;315:51-66.
- ^ Liu Y, Nickle DC, Shriner D, Jensen MA, Learn GH Jr, Mittler JE, Mullins JI. Molecular clock-like evolution of human immunodeficiency virus type 1.Virology. 2004 Nov 10;329(1):101-8.
- ^ a b c d e f g h Prescott, L (1993). Microbiology. Wm. C. Brown Publishers. 0-697-01372-3.
- ^ Keese P, Gibbs A. Plant viruses: master explorers of evolutionary space.Curr Opin Genet Dev. 1993 Dec;3(6):873-7.
- ^ Koonin EV. The Biological Big Bang model for the major transitions in evolution.Biol Direct. 2007 Aug 20;2:21.
- ^ Rybicki EP (1990) The classification of organisms at the edge of life, or problems with virus systematics. S Aft J Sci 86:182-186
- ^ LWOFF A (1957). "The concept of virus". J. Gen. Microbiol. 17 (2): 239–53. PMID 13481308.
- ^ Lwoff A, Horne RW, Tournier P (1962). "A virus system". C. R. Hebd. Seances Acad. Sci. (in French). 254: 4225–7. PMID 14467544.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lwoff A, Horne R, Tournier P (1962). "A system of viruses". Cold Spring Harb. Symp. Quant. Biol. 27: 51–5. PMID 13931895.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Virus Taxonomy 8th Reports of the International Committee on Taxonomy of Viruses, C.M. Fauquet, M.A. Mayo, J. Maniloff, U. Desselberger, and L.A. Ball (eds), Academic Press, 1162 pp. (2005) Elsevier Publication Date: 27 May 2005
- ^ Baltimore D (1974). "The strategy of RNA viruses". Harvey Lect. 70 Series: 57–74. PMID 4377923.
- ^ Temin HM, Baltimore D (1972). "RNA-directed DNA synthesis and RNA tumor viruses". Adv. Virus Res. 17: 129–86. PMID 4348509.
- ^ van Regenmortel MH, Mahy BW (2004). "Emerging issues in virus taxonomy". Emerging Infect. Dis. 10 (1): 8–13. PMID 15078590.
- ^ Mayo MA (1999). "Developments in plant virus taxonomy since the publication of the 6th ICTV Report. International Committee on Taxonomy of Viruses". Arch. Virol. 144 (8): 1659–66. doi:10.1007/s007050050620. PMID 10486120.
- ^ de Villiers EM, Fauquet C, Broker TR, Bernard HU, zur Hausen H (2004). "Classification of papillomaviruses". Virology. 324 (1): 17–27. doi:10.1016/j.virol.2004.03.033. PMID 15183049.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Caspar DL, Klug A (1962). "Physical principles in the construction of regular viruses". Cold Spring Harb. Symp. Quant. Biol. 27: 1–24. PMID 14019094.
- ^ Crick FH, Watson JD (1956). "Structure of small viruses". Nature. 177 (4506): 473–5. doi:10.1038/177473a0. PMID 13309339.
- ^ "Virus triangulation numbers via Internet Archive". Retrieved 2006-04-05.
- ^ Long GW, Nobel J, Murphy FA, Herrmann KL, and Lourie B (1970) Experience with electron microscopy in the differential diagnosis of smallpox. Applied Microbiology 20(3):497-504.
- ^ Kiselev NA, Sherman MB, Tsuprun VL (1990). "Negative staining of proteins". Electron Microsc. Rev. 3 (1): 43–72. PMID 1715774.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c Flinth (2004). Principles of Virology (2nd edn ed.). ASM Press, New York. 1-55581-259-7.
{{cite book}}:|edition=has extra text (help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Pressing J, Reanney DC. Divided genomes and intrinsic noise.J Mol Evol. 1984;20(2):135-46.
- ^ Goudsmit, Jaap. Viral Sex. Oxford Univ Press, 1998.ISBN-13: 9780195124965 ISBN-10: 0195124960
- ^ Zhou NN, Senne DA, Landgraf JS, Swenson SL, Erickson G, Rossow K, Liu L, Yoon K, Krauss S, Webster RG. Genetic reassortment of avian, swine, and human influenza A viruses in American pigs.J Virol. 1999 Oct;73(10):8851-6.
- ^ Worobey M, Holmes EC (1999). "Evolutionary aspects of recombination in RNA viruses". J. Gen. Virol. 80 ( Pt 10): 2535–43. PMID 10573145.
- ^ Lukashev AN (2005). "Role of recombination in evolution of enteroviruses". Rev. Med. Virol. 15 (3): 157–67. doi:10.1002/rmv.457. PMID 15578739.
- ^ Umene K (1999). "Mechanism and application of genetic recombination in herpesviruses". Rev. Med. Virol. 9 (3): 171–82. doi:10.1002/(SICI)1099-1654(199907/09)9:3<171::AID-RMV243>3.0.CO;2-A. PMID 10479778.
- ^ Pan XP, Li LJ, Du WB, Li MW, Cao HC, Sheng JF. Differences of YMDD mutational patterns, precore/core promoter mutations, serum HBV DNA levels in lamivudine-resistant hepatitis B genotypes B and C. J Viral Hepat. 2007 Nov;14(11):767-74.
- ^ Hampson AW, Mackenzie JS. The influenza viruses.Med J Aust. 2006 Nov 20;185(10 Suppl):S39-43.
- ^ Nakajima K. The mechanism of antigenic shift and drift of human influenza virus Nippon Rinsho. 2003 Nov;61(11):1897-903.
- ^ Hundley F, McIntyre M, Clark B, Beards G, Wood D, Chrystie I, Desselberger U. Heterogeneity of genome rearrangements in rotaviruses isolated from a chronically infected immunodeficient child.J Virol. 1987 Nov;61(11):3365-72.
- ^ Metzner KJ. Detection and significance of minority quasispecies of drug-resistant HIV-1. HIV Ther. 2006 Dec;11(4):74-81.
- ^ Barman S, Ali A, Hui EK, Adhikary L, Nayak DP (2001). "Transport of viral proteins to the apical membranes and interaction of matrix protein with glycoproteins in the assembly of influenza viruses". Virus Res. 77 (1): 61–9. doi:10.1016/S0168-1702(01)00266-0. PMID 11451488.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Prangishvili D, Forterre P, Garrett RA (2006). "Viruses of the Archaea: a unifying view". Nat. Rev. Microbiol. 4 (11): 837–48. doi:10.1038/nrmicro1527. PMID 17041631.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Prangishvili D, Garrett RA (2004). "Exceptionally diverse morphotypes and genomes of crenarchaeal hyperthermophilic viruses". Biochem. Soc. Trans. 32 (Pt 2): 204–8. doi:10.1042/BST0320204. PMID 15046572.
- ^ Mojica FJ, Díez-Villaseñor C, García-Martínez J, Soria E (2005). "Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements". J. Mol. Evol. 60 (2): 174–82. doi:10.1007/s00239-004-0046-3. PMID 15791728.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Makarova KS, Grishin NV, Shabalina SA, Wolf YI, Koonin EV (2006). "A putative RNA-interference-based immune system in prokaryotes: computational analysis of the predicted enzymatic machinery, functional analogies with eukaryotic RNAi, and hypothetical mechanisms of action". Biol. Direct. 1: 7. doi:10.1186/1745-6150-1-7. PMID 16545108.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Rybicki ibid
- ^ Holmes EC.PLoS Biol. 2007 Oct 2;5(10):e278. Viral Evolution in the Genomic Age
- ^ Shackelton LA, Holmes EC.Phylogenetic evidence for the rapid evolution of human B19 erythrovirus.J Virol. 2006 Apr;80(7):3666-9.
- ^ Vlassov, Alexander V. (Jul 2005). "The RNA World on Ice: A New Scenario for the Emergence of RNA Information". Journal of Molecular Evolution 61: 264-273.
- ^ Fisher, Bruce; Harvey, Richard P.; Champe, Pamela C. Lippincott's Illustrated Reviews: Microbiology (Lippincott's Illustrated Reviews Series). Hagerstown, MD: Lippincott Williams & Wilkins. ISBN 0-7817-8215-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) Pages 354-366 - ^ a b c d Unless else specified in boxes, the ref is: Fisher, Bruce; Harvey, Richard P.; Champe, Pamela C. Lippincott's Illustrated Reviews: Microbiology (Lippincott's Illustrated Reviews Series). Hagerstown, MD: Lippincott Williams & Wilkins. ISBN 0-7817-8215-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) Pages 354-366 - ^ Fisher, Bruce; Harvey, Richard P.; Champe, Pamela C. Lippincott's Illustrated Reviews: Microbiology (Lippincott's Illustrated Reviews Series). Hagerstown, MD: Lippincott Williams & Wilkins. ISBN 0-7817-8215-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) Page 315 - ^ Chen C, Chiu Y, Wei F, Koong F, Liu H, Shaw C, Hwu H, Hsiao K (1999). "High seroprevalence of Borna virus infection in schizophrenic patients, family members and mental health workers in Taiwan". Mol Psychiatry. 4 (1): 33–8. doi:10.1038/sj.mp.4000484. PMID 10089006.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Margolis TP, Elfman FL, Leib D, Pakpour N, Apakupakul K, Imai Y, Voytek C. Spontaneous reactivation of herpes simplex virus type 1 in latently infected murine sensory Ganglia.J Virol. 2007 Oct;81(20):11069-74. Epub 2007 Aug 8.
- ^ Bertoletti A, Gehring A (2007). "Immune response and tolerance during chronic hepatitis B virus infection". Hepatol. Res. 37 Suppl 3: S331–8. doi:10.1111/j.1872-034X.2007.00221.x. PMID 17931183.
- ^ Nguyen VT, McLaws ML, Dore GJ (2007). "Highly endemic hepatitis B infection in rural Vietnam". Journal of Gastroenterology and Hepatology. 22: 2093. doi:10.1111/j.1440-1746.2007.05010.x. PMID 17645465.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rodrigues C, Deshmukh M, Jacob T, Nukala R, Menon S, Mehta A (2001). "Significance of HBV DNA by PCR over serological markers of HBV in acute and chronic patients". Indian journal of medical microbiology. 19 (3): 141–4. PMID 17664817.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fowler MG, Lampe MA, Jamieson DJ, Kourtis AP, Rogers MF (2007). "Reducing the risk of mother-to-child human immunodeficiency virus transmission: past successes, current progress and challenges, and future directions". Am. J. Obstet. Gynecol. 197 (3 Suppl): S3–9. doi:10.1016/j.ajog.2007.06.048. PMID 17825648.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sauerbrei A, Wutzler P (2000). "The congenital varicella syndrome". Journal of perinatology : official journal of the California Perinatal Association. 20 (8 Pt 1): 548–54. PMID 11190597.
- ^ Garnett GP. Role of herd immunity in determining the effect of vaccines against sexually transmitted disease.J Infect Dis. 2005 Feb 1;191 Suppl 1:S97-106.
- ^ Platonov AE (2006). "(The influence of weather conditions on the epidemiology of vector-borne diseases by the example of West Nile fever in Russia)". Vestn. Akad. Med. Nauk SSSR (in Russian) (2): 25–9. PMID 16544901.
- ^ Ranlet P. The British, the Indians, and smallpox: what actually happened at Fort Pitt in 1763? Pa Hist. 2000;67(3):427-41.
- ^ Van Rijn K. "Lo! The poor Indian!" colonial responses to the 1862-63 smallpox epidemic inBritish Columbia and Vancouver Island.Can Bull Med Hist. 2006;23(2):541-60.
- ^ Patterson KB, Runge T. Smallpox and the Native American.Am J Med Sci. 2002 Apr;323(4):216-22.
- ^ Sessa R, Palagiano C, Scifoni MG, di Pietro M, Del Piano M. The major epidemic infections: a gift from the Old World to the New? Panminerva Med. 1999 Mar;41(1):78-84.
- ^ Bianchine PJ, Russo TA. The role of epidemic infectious diseases in the discovery of America. Allergy Proc. 1992 Sep-Oct;13(5):225-32.
- ^ Hauptman LM. Smallpox and American Indian; Depopulation in Colonial New York. N Y State J Med. 1979 Nov;79(12):1945-9.
- ^ Fortuine R. Smallpox decimates the Tlingit (1787). Alaska Med. 1988 May-Jun;30(3):109.
- ^ Patterson, KD (1991). "The geography and mortality of the 1918 influenza pandemic". Bull Hist Med. 65 (1): 4–21. PMID 2021692.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Johnson, Niall P. A. S. and Mueller, Juergen, "Updating the Accounts: Global Mortality of the 1918–1920 'Spanish' Influenza Pandemic," Bulletin of the History of Medicine 76 (2002), pp. 105–115.
- ^
Gao, F., Bailes, E., Robertson, D. L., Chen, Y., Rodenburg, C. M., Michael, S. F., Cummins, L. B., Arthur, L. O., Peeters, M., Shaw, G. M., Sharp, P. M. and Hahn, B. H. (1999). "Origin of HIV-1 in the Chimpanzee Pan troglodytes troglodytes". Nature. 397 (6718): 436–441. doi:10.1038/17130. PMID doi:[https://doi.org/10.1038%2F17130 10.1038/17130 9989410 '"`UNIQ--templatestyles-000000D0-QINU`"'[[doi (identifier)|doi]]:[https://doi.org/10.1038%2F17130 10.1038/17130]].
{{cite journal}}: Check|pmid=value (help); External link in|pmid=|pmid=at position 9 (help)CS1 maint: multiple names: authors list (link) - ^ UNAIDS (2006). "Overview of the global AIDS epidemic". 2006 Report on the global AIDS epidemic.
{{cite book}}:|access-date=requires|url=(help);|format=requires|url=(help); External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help) - ^ Mawar N, Saha S, Pandit A, Mahajan U. The third phase of HIV pandemic: social consequences of HIV/AIDS stigma & discrimination & future needs.Indian J Med Res. 2005 Dec;122(6):471-84. Review.
- ^ Towner JS, Khristova ML, Sealy TK, Vincent MJ, Erickson BR, Bawiec DA, HartmanAL, Comer JA, Zaki SR, Stroher U, Gomes da Silva F, del Castillo F, Rollin PE,Ksiazek TG, Nichol ST. Marburgvirus genomics and association with a large hemorrhagic fever outbreak in Angola.J Virol. 2006 Jul;80(13):6497-516.
- ^ Koike K (2007). "Hepatitis C virus contributes to hepatocarcinogenesis by modulating metabolic and intracellular signalling pathways". J. Gastroenterol. Hepatol. 22 Suppl 1: S108–11. doi:10.1111/j.1440-1746.2006.04669.x. PMID 17567457.
- ^ Hu J, Ludgate L (2007). "HIV-HBV and HIV-HCV coinfection and liver cancer development". Cancer Treat. Res. 133: 241–52. PMID 17672044.
- ^ Bellon M, Nicot C (2007). "Telomerase: a crucial player in HTLV-I-induced human T-cell leukemia". Cancer genomics & proteomics. 4 (1): 21–5. PMID 17726237.
- ^ Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S (2007). "Human papillomavirus and cervical cancer". Lancet. 370 (9590): 890–907. doi:10.1016/S0140-6736(07)61416-0. PMID 17826171.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Klein E, Kis LL, Klein G (2007). "Epstein-Barr virus infection in humans: from harmless to life endangering virus-lymphocyte interactions". Oncogene. 26 (9): 1297–305. doi:10.1038/sj.onc.1210240. PMID 17322915.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Alberts, Bruce (2002). Molecular Biology of the Cell; Fourth Edition. New York and London: Garland Science. ISBN 0-8153-3218-1.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Ding SW, Voinnet O. Antiviral immunity directed by small RNAs. Cell. 2007 Aug 10;130(3):413-26.
- ^ Patton JT, Vasquez-Del Carpio R, Spencer E (2004). "Replication and transcription of the rotavirus genome". Curr. Pharm. Des. 10 (30): 3769–77. doi:10.2174/1381612043382620. PMID 15579070.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Jayaram H, Estes MK, Prasad BV (2004). "Emerging themes in rotavirus cell entry, genome organization, transcription and replication". Virus Res. 101 (1): 67–81. doi:10.1016/j.virusres.2003.12.007. PMID 15010218.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Greer S, Alexander GJ. Viral serology and detection. Baillieres Clin Gastroenterol. 1995 Dec;9(4):689-721
- ^ Laurence JC. Hepatitis A and B immunizations of individuals infected with humanimmunodeficiency virus.Am J Med. 2005 Oct;118 Suppl 10A:75S-83S.
- ^ Cascalho M, Platt JL. Novel functions of B cells.Crit Rev Immunol. 2007;27(2):141-51.
- ^ Khatri M, Sharma JM. Modulation of macrophages by infectious bursal disease virus.Cytogenet Genome Res. 2007;117(1-4):388-93
- ^ Hilleman MR. Strategies and mechanisms for host and pathogen survival in acute and persistent viral infections. Proc Natl Acad Sci U S A. 2004 Oct 5;101 Suppl 2:14560-6. Epub 2004 Aug 5.
- ^ Le Page C, Genin P, Baines MG, Hiscott J. Interferon activation and innate immunity.Rev Immunogenet. 2000;2(3):374-86.
- ^ Asaria P, MacMahon E (2006). "Measles in the United Kingdom: can we eradicate it by 2010?". BMJ. 333 (7574): 890–5. doi:10.1136/bmj.38989.445845.7C. PMID 17068034.
- ^ Lane JM (2006). "Mass vaccination and surveillance/containment in the eradication of smallpox". Curr. Top. Microbiol. Immunol. 304: 17–29. PMID 16989262.
- ^ Arvin AM, Greenberg HB (2006). "New viral vaccines". Virology. 344 (1): 240–9. doi:10.1016/j.virol.2005.09.057. PMID 16364754.
- ^ Pastoret PP, Schudel AA, Lombard M (2007). "Conclusions--future trends in veterinary vaccinology". Rev. - Off. Int. Epizoot. 26 (2): 489–94, 495–501, 503–9. PMID 17892169.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Palese P (2006). "Making better influenza virus vaccines?". Emerging Infect. Dis. 12 (1): 61–5. PMID 16494719.
- ^ Thomssen R (1975). "Live attenuated versus killed virus vaccines". Monographs in allergy. 9: 155–76. PMID 1090805.
- ^ McLean AA (1986). "Development of vaccines against hepatitis A and hepatitis B". Rev. Infect. Dis. 8 (4): 591–8. PMID 3018891.
- ^ Casswall TH, Fischler B (2005). "Vaccination of the immunocompromised child". Expert review of vaccines. 4 (5): 725–38. doi:10.1586/14760584.4.5.725. PMID 16221073.
- ^ Magden J, Kääriäinen L, Ahola T (2005). "Inhibitors of virus replication: recent developments and prospects". Appl. Microbiol. Biotechnol. 66 (6): 612–21. doi:10.1007/s00253-004-1783-3. PMID 15592828.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mindel A, Sutherland S (1983). "Genital herpes - the disease and its treatment including intravenous acyclovir". J. Antimicrob. Chemother. 12 Suppl B: 51–9. PMID 6355051.
- ^ Witthoft T, Moller B, Wiedmann KH, Mauss S, Link R, Lohmeyer J, Lafrenz M,Gelbmann CM, Huppe D, Niederau C, Alshuth U. Safety, tolerability and efficacy of peginterferon alpha-2a and ribavirin in chronic hepatitis C in clinical practice: The German Open Safety Trial. J Viral Hepat. 2007 Nov;14(11):788-96.
- ^ Rudin D, Shah SM, Kiss A, Wetz RV, Sottile VM. Interferon and lamivudine vs. interferon for hepatitis B e antigen-positive hepatitis B treatment: meta-analysis of randomized controlled trials.Liver Int. 2007 Nov;27(9):1185-93.
- ^ Matsuzaki S, Rashel M, Uchiyama J; et al. (2005). "Bacteriophage therapy: a revitalized therapy against bacterial infectious diseases". J. Infect. Chemother. 11 (5): 211–9. doi:10.1007/s10156-005-0408-9. PMID 16258815.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Proceedings of SPIE -- Volume 6413Smart Materials IV, Nicolas H. Voelcker, Editor, 64130F (Dec. 22, 2006). Hybrid organic-inorganic nanoparticles: controlled incorporation of gold nanoparticles into virus-like particles and application in surface-enhanced Raman spectroscopy Marcus Niebert, James Riches, Mark Howes, Charles Ferguson, Robert G. Parton, Anton P. J. Middelberg, Llew Rintoul, and Peter M. Fredericks.Queensland Univ. of Technology (Australia) (published online Dec. 22, 2006)
- ^ Fischlechner M, Donath E (2007). "Viruses as Building Blocks for Materials and Devices". Angewandte Chemie International Edition. 46: 3184. doi:10.1002/anie.200603445.
- ^ Fluorescent signal amplification of carbocyanine dyes using engineered viral nanoparticles. Carissa M. Soto, Amy Szuchmacher Blum, Nikolai Lebedev, Gary J. Vora, Carolyn E. Meador, Angela P. Won, Anju Chatterji, John E. Johnson, and Banahalli R. Ratna, Journal of the American Chemical Society, 128, 5184 (2006).
- ^ An Engineered Virus as a Scaffold for Three-Dimensional Self-Assembly on the Nanoscale. Amy Szuchmacher Blum, Carissa M. Soto, Charmaine D. Wilson, Tina L. Brower, Steven K. Pollack, Terence L. Schull, Anju Chatterji, Tianwei Lin, John E. Johnson, Christian Amsinck, Paul Franzon, Ranganathan Shashidhar and Banahalli Ratna, Small, 7, 702 (2005).
- ^ "Researchers build tiny batteries with viruses". MIT News Office. Retrieved 2007-04-05.
- ^ "Researchers Reconstruct 1918 Pandemic Influenza Virus; Effort Designed to Advance Preparedness". Centers for Disease Control. Retrieved 2007-04-05.
- ^ Aragón TJ, Ulrich S, Fernyak S, Rutherford GW (2003). "Risks of serious complications and death from smallpox vaccination: a systematic review of the United States experience, 1963-1968". BMC public health. 3: 26. doi:10.1186/1471-2458-3-26. PMID 12911836.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Weiss MM, Weiss PD, Mathisen G, Guze P (2004). "Rethinking smallpox". Clin. Infect. Dis. 39 (11): 1668–73. doi:10.1086/425745. PMID 15578369.
{{cite journal}}: CS1 maint: multiple names: authors list (link)