From Wikipedia, the free encyclopedia
Plakohypaphorines are halogenated indolic non-proteinogenic amino acids named for their similarity to hypaphorine (N,N,N -trimethyltryptophan betaine ). First reported in the Caribbean sponge Plakortis simplexplakohypaphorines A-C were the first iodine-containing indole alkaloids to be discovered in nature. Plakohypaphorines D-F , also found in P. simplex , were reported in 2004 by a group including the researchers who discovered the original plakohypaphorines.
Plakohypaphorine
Chemical name
Chemical formula
SMILES
Image
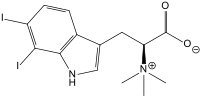
Plakohypaphorine
7-Iodo-N,N,N -trimethyltryptophan
C14 H17 IN2 O2
OC(=O)C(N(C)(C)C)Cc2cnc1c2cccc1I
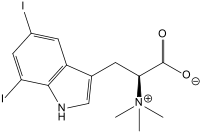
Plakohypaphorine B
6,7-Diiodo-N,N,N -trimethyltryptophan
C14 H16 I2 N2 O2
OC(=O)C(N(C)(C)C)Cc2cnc1c2ccc(I)c1I
Plakohypaphorine C
5,7-Diiodo-N,N,N -trimethyltryptophan
C14 H16 I2 N2 O2
CN(C)(C)C(C(=O)O)Cc2cnc1c2cc(I)cc1I
Plakohypaphorine D
5,6-Diiodo-N,N,N -trimethyltryptophan
C14 H16 I2 N2 O2
Ic2cc1c(cc2I)ncc1CC(C(O)=O)N(C)(C)C
Plakohypaphorine E
5,6,7-Triiodo-N,N,N -trimethyltryptophan
C14 H15 I3 N2 O2
CN(C)(C)C(C(=O)O)Cc1cnc(c(I)c2I)c1cc2I
Plakohypaphorine F
6-Chloro-5-iodo-N,N,N -trimethyltryptophan
C14 H16 ClIN2 O2
Clc2c1ncc(CC(C(O)=O)N(C)(C)C)c1cc(I)c2
Taglialatela-Scafati Orazio et al., 2003. Plakohypaphorines A-C, Iodine-Containing Alkaloids from the Caribbean Sponge Plakortis simplex. European Journal of Organic Chemistry. 2003(2), pp. 284–287.
Borrelli, Francesca, et al., 2004. Iodinated Indole Alkaloids From Plakortis simplex, New Plakohypaphorines and an Evaluation of Their Antihistamine Activity . European Journal of Organic Chemistry. 2004(15), pp. 3227–3232.
Tryptamines
1-Methylpsilocin 2-HO-NMT 2-Me-DET 2-Methyl-5-HT 2,N ,N -TMT 4,5-DHP-DMT 4-AcO-DALT 4-AcO-DET 4-AcO-DiPT 4-AcO-DPT 4-AcO-EPT 4-AcO-MALT 4-AcO-MET 4-AcO-MiPT 4-AcO-NMT 4-AcO-TMT 4-F-5-MeO-DMT 4-HO-5-MeO-DMT 4-HO-DALT 4-HO-DBT 4-HO-DET 4-HO-DiPT 4-HO-DPT 4-HO-DSBT 4-HO-EPT 4-HO-MALT 4-HO-MET 4-HO-McPT 4-HO-McPeT 4-HO-MiPT 4-HO-MPT 4-HO-MsBT 4-HO-NALT 4-HO-NMT 4-HO-PiPT 4-HO-pyr-T 4-HO-TMT 4-HT 4-MeO-DiPT 4-MeO-DMT 4-MeO-MiPT 4-PrO-DMT 4,5-MDO-DMT 4,5-MDO-DiPT 5-BT 5-Bromo-DMT 5-CT 5-Chloro-DMT 5-Ethoxy-DMT 5-Ethyl-DMT 5-Fluoro-DET 5-Fluoro-DMT 5-Fluoro-EPT 5-Fluoro-MET 5-HO-DiPT 5-HTP (oxitriptan) 5-MeO-2-TMT 5-MeO-34MPEMT 5-MeO-7,N ,N -TMT 5-MeO-DALT 5-MeO-DBT 5-MeO-DET 5-MeO-DiPT 5-MeO-DMT 5-MeO-DPT 5-MeO-EiPT 5-MeO-EPT 5-MeO-MALT 5-MeO-MET 5-MeO-MiPT 5-MeO-NMT 5-MeO-pyr-T 5-MeO-NBpBrT 5-MeO-T-NBOMe 5-MeS-DMT 5-Methoxytryptamine (5-MT; mexamine) 5-Methyl-DMT 5-Methyltryptamine 5-MT-NB3OMe 5-(Nonyloxy)tryptamine 5,6-MeO-MiPT 5,6-MDO-DiPT 5,6-MDO-DMT 5,6-MDO-MiPT 5,6-DHT 5,7-DHT 6-Fluoro-DMT 6-MeO-DMT 7-Methyl-DMT Acetryptine (5-AT) Aeruginascin (4-PO-TMT) AGH-107 AGH-192 AH-494 ALiPT Alpertine Baeocystin (4-PO-NMT) Benzotript (4-chlorobenzoyl-L -tryptophan) Bufotenidine (5-HTQ) Bufotenin (5-HO-DMT) Convolutindole A CP-132,484 DALT DBT Desformylflustrabromine DET DiPT DMT DPT E-6801 E-6837 EiPT EMDT EPT Ethocybin (4-PO-DET) FGIN-127 FGIN-143 FT-104 HIOC Idalopirdine Indolylethylfentanyl Indorenate Iprocin (4-HO-DiPT) Lespedamine MET Methylbutyltryptamine Miprocin (4-HO-MiPT) MiPT MPT Milipertine MS-245 MSBT N -Feruloylserotonin (moschamine)NET NMT Norbaeocystin (4-PO-T) NTBT O-4310 O -PivalylbufotenineOxypertine PiPT Psilacetin (O -acetylpsilocin; 4-AcO-DMT) Psilocin (4-HO-DMT) Psilocybin (4-PO-DMT) Pyr-T RS134-49 Serotonin (5-HT) Solypertine ST-1936 Tryptamine Tryptophan Yuremamine Z2876442907 N -Acetyltryptaminesα-Alkyltryptamines
2,α-DMT 4-HO-αMT 4-HO-MPMI (lucigenol) 4-Me-αET 4-Me-αMT 5-Chloro-αMT 5-Ethoxy-αMT 5-Fluoro-αET 5-Fluoro-αMT 5-iPrO-αMT 5-MeO-α,N ,N -TMT 5-MeO-αET 5-MeO-αMT 5-MeO-MPMI 5-Methyl-αET 6-Fluoro-αMT 7-Chloro-αMT 7-Methyl-αET α-Methyl-5-HTP α-Methylmelatonin α-Methylserotonin (5-HO-αMT) α-Methyltryptophan (αMTP) α,N -DMT (N -methyl-αMT) α,N ,N -TMT α,N ,O -TMS αET (etryptamine) αMT AL-37350A (4,5-DHP-αMT) BNC-210 BW-723C86 CP-135807 IPAP (α,N -DPT) MPMI Triptans Cyclized tryptamines
Bay R 1531 Ciclindole Cyclic 3-OHM Ergolines and lysergamides (e.g., LSD )Flucindole Harmala alkaloidsβ-carbolines (e.g., 6-MeO-THH , 9-Me-BC , β-carboline (norharman) , harmaline , harmalol , harmane , harmine , pinoline , tetrahydroharmine , tryptoline )Iboga alkaloidsDM-506 (ibogaminalog) , ibogaine , ibogamine , noribogaine , tabernanthalog , tabernanthine )Metralindole NDTDI PHA-57378 PNU-22394 PNU-181731 RU-28306 Yohimbans (e.g., yohimbine , rauwolscine , spegatrine , corynanthine , ajmalicine , reserpine , deserpidine , rescinnamine ) Related compounds