Portal:Viruses/Selected virus
The following articles are currently featured as the Selected virus at the Viruses Portal. To suggest an article for inclusion, use the suggestions page
Portal:Viruses/Selected virus/1
Rotavirus is a genus of double-stranded RNA viruses in the family Reoviridae. There are nine species A–I; rotavirus A, the most common, causes over 90% of infections in humans. Rotavirus also infects animals, including livestock. The virus is transmitted by the faecal–oral route, with fewer than 100 virus particles being required for infection. Rotaviruses are stable in the environment and normal sanitary measures fail to protect against them. Effective rotavirus vaccines are the main prevention method.
The virus infects and damages the enterocytes lining the small intestine, causing gastroenteritis (sometimes referred to as "stomach flu," although the virus is not related to influenza). A viral toxin, NSP4, is responsible for some of the pathology. Rotavirus is the most common cause of diarrhoea among infants and young children. Almost every child worldwide has been infected with rotavirus at least once by the age of five. In 2013, 215,000 children under five died from rotavirus infection, mostly in developing countries, and almost two million more become severely ill. Immunity develops with each infection and adults are rarely affected.
Portal:Viruses/Selected virus/2
Human immunodeficiency virus (HIV) is a lentivirus, an RNA virus in the retrovirus family. Two types of HIV have been characterised: HIV-1 is the more virulent and is responsible for most infections worldwide; HIV-2 is mainly confined to West Africa. The genome consists of two copies of a single-stranded +RNA, which contains nine genes. The roughly spherical virus particle has a diameter of about 120 nm; it is enveloped and contains a conical capsid made of around 2,000 copies of the p24 protein. The envelope glycoprotein, a trimeric complex of gp120 and gp41, binds to CD4, the primary receptor on the host cell.
Transmission occurs by the transfer of bodily fluids including blood, semen, vaginal fluids and breast milk, in which the virus is present both as free virus particles and within infected immune cells. HIV infects key cells in the human immune system including CD4+ T helper cells, macrophages and dendritic cells. Infection leads to low levels of CD4+ T cells via several mechanisms, resulting in a progressive immunodeficiency disease known as AIDS.
Portal:Viruses/Selected virus/3
Canine parvovirus type 2 (CPV2) is a non-enveloped, single-stranded DNA virus in the Parvoviridae family. The icosahedral viral capsid is only 20–26 nm in diameter, making it one of the smallest viruses. The genome is about 5000 nucleotides long. The virus is very similar to feline panleukopenia virus, another parvovirus, as well as mink enteritis and raccoon and fox parvoviruses. It infects dogs, wolves, foxes and other canids, big cats and occasionally domestic cats, but cannot infect humans.
A relatively new disease, CPV2 infection was first recognised in 1978 and rapidly spread worldwide. The virus is stable and highly infectious, being transmitted by contact with faeces, infected soil or contaminated objects. After ingestion, the virus replicates in the lymphoid tissue in the throat, then spreads to the bloodstream to infect cells of the lymph nodes, intestinal crypts and bone marrow, damaging the intestinal lining. The more common intestinal form of disease causes vomiting and severe, often bloody diarrhoea. The cardiac form affects puppies under 8 weeks, causing respiratory or cardiovascular failure; mortality can reach 91% in untreated cases. No specific antiviral drug is available. Prevention is by vaccination.
Portal:Viruses/Selected virus/4
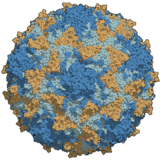
Poliovirus is an enterovirus, an RNA virus in the Picornaviridae family, associated with the paralytic disease polio. The icosahedral virus particle is about 30 nanometres in diameter and lacks an envelope. It contains a relatively short, single-stranded positive RNA genome of around 7500 nucleotides, which encodes about ten viral products. The virus has a fairly high mutation rate even for an RNA virus. Historically there were three serotypes, each with a slightly different capsid protein; PV1 is the most common, and PV2 was declared eradicated in 2015.
The virus only naturally infects humans, although some monkeys can be infected experimentally; 95% of infections are asymptomatic. Infection occurs via the faecal–oral route and viral replication occurs in the alimentary tract. The virus enters the host cell by binding to an immunoglobulin-like receptor, CD155. Fully assembled poliovirus leaves the cell 4–6 hours after initiation of infection. Poliovirus was first isolated in 1909 by Karl Landsteiner and Erwin Popper. Its genome was sequenced in 1981. Among the simplest clinically significant viruses, poliovirus is one of the best-characterised viruses, and has become a useful model for studying RNA viruses.
Portal:Viruses/Selected virus/5
Noroviruses are a genus of non-enveloped, single-stranded RNA viruses in the family Caliciviridae. The positive-sense RNA genome is approximately 7500 nucleotides long. Known noroviruses fall into five different genogroups (GI–GV); three groups infect humans, the other two mice, and cattle and other bovines. All are considered strains of a single species, Norwalk virus.
Noroviruses are extremely contagious, with fewer than 20 virus particles being infectious. They are transmitted directly from person to person and indirectly via contaminated water and food. After infection, the virus replicates in the small intestine, causing acute gastroenteritis, which develops 12–48 hours after exposure and lasts for 24–72 hours. The characteristic symptoms include nausea, forceful vomiting, watery diarrhoea and abdominal pain. Infection is usually self-limiting and rarely severe. Noroviruses cause 18% of acute gastroenteritis episodes in humans, with around 685 million cases and 200,000 deaths every year, mainly in very young, elderly or immunosuppressed people. No vaccine is available. Hand washing with soap and water is effective in reducing transmission.
Portal:Viruses/Selected virus/6
Papillomaviruses are small non-enveloped DNA viruses that make up the Papillomaviridae family. Their circular double-stranded genome is around 8000 nucleotides long. The icosahedral capsid is 55–60 nm in diameter. They infect humans, other mammals and some other vertebrates including birds, snakes, turtles and fish. Around a hundred species are classified into 53 genera. All papillomaviruses replicate exclusively in epithelial cells of stratified squamous epithelium, which forms the skin and some mucosal surfaces, including the lining of the mouth, airways, genitals and anus.
Infection by most papillomaviruses is either asymptomatic or causes small benign tumours known as warts or papillomas. Francis Peyton Rous showed in 1935 that the Shope papilloma virus could cause skin cancer in rabbits – the first time that a virus was shown to cause cancer in mammals – and papillomas caused by some virus types, including human papillomavirus 16 and 18, carry a risk of becoming cancerous if the infection persists. Papillomaviruses are associated with cancers of the cervix, vulva, vagina, penis, oropharynx and anus in humans.
Portal:Viruses/Selected virus/7
Hantaviruses (or orthohantaviruses) are a family of RNA viruses in the Bunyavirales order. The enveloped virion is 120–160 nm in diameter and contains a single-stranded, negative-sense RNA genome with three segments. They infect many species of rodents, as well as shrews and moles, without causing disease, and can be transmitted to humans, where they can cause serious disease. Hantaan virus, the first known hantavirus, was discovered in 1976 as the cause of a novel haemorrhagic fever affecting combatants in the Korean War; it can also be caused by other hantaviruses, including Dobrava-Belgrade and Seoul viruses. Some hantaviruses, including Sin Nombre and Bayou, cause a pulmonary syndrome. Others have not yet been associated with human disease.
Unlike other bunyaviruses, hantaviruses are not transmitted by arthropods. Rodents act as the vector, with transmission to humans usually occurring via contact with urine, saliva or faeces, by inhalation of aerosolised excreta or by bite. Little is understood about how hantaviruses cause disease; the main site of viral replication in the body is unknown. Rodent control is important in disease prevention.
Portal:Viruses/Selected virus/8
Epstein–Barr virus (EBV) (also human herpesvirus 4) is a DNA virus in the Herpesviridae family which infects humans. The virion is around 122–180 nm in diameter. As in all herpesviruses, the nucleocapsid is surrounded by a protein tegument, as well as an envelope. The double-stranded DNA genome is about 172 kb, with 85 genes, making it one of the more complex viruses.
Transmission is via saliva and genital secretions. The virus infects epithelial cells in the pharynx and B cells of the immune system, producing virions by budding. EBV also becomes latent in B cells, possibly in the bone marrow, allowing the infection to persist lifelong. In the latent state, the linear genome is made circular and replicates in the nucleus separately from the host DNA as an episome. Reactivation is poorly understood but is thought to be triggered by the B cell responding to other infections. EBV infection occurs in around 95% of people. Infectious mononucleosis or glandular fever can occur when first infection is delayed until adolescence or adulthood. EBV is associated with some types of cancer, including Burkitt's lymphoma and nasopharyngeal carcinoma. In people with HIV, it can cause hairy leukoplakia and central nervous system lymphomas.
Portal:Viruses/Selected virus/9
West Nile virus (WNV) is a flavivirus, an RNA virus in the Flaviviridae family. The enveloped virion is 45–50 nm in diameter and contains a single-stranded, positive-sense RNA genome of around 11,000 nucleotides, encoding ten proteins. The main natural hosts are birds (the reservoir) and several species of Culex mosquito (the vector). WNV can also infect humans and some other mammals, including horses, dogs and cats, as well as some reptiles. Transmission to humans is generally by bite of the female mosquito. Mammals form a dead end for the virus, as it cannot replicate sufficiently efficiently in them to complete the cycle back to the mosquito.
First identified in Uganda in 1937, WNV was at first mainly associated with disease in horses, with only sporadic cases of human disease until the 1990s. The virus is now endemic in Africa, west and central Asia, Oceania, the Middle East, Europe and North America. A fifth of humans infected experience West Nile fever, a flu-like disease. In less than 1% of those infected, the virus invades the central nervous system, causing encephalitis, meningitis or flaccid paralysis. No specific antiviral treatment has been licensed and only a veterinary vaccine is available. Mosquito control is the main preventive measure.
Portal:Viruses/Selected virus/10
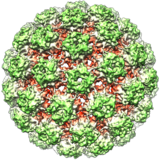
Adenoviruses are a group of non-enveloped DNA viruses that make up the Adenoviridae family. At 90–100 nm in diameter, they are the largest known viruses to lack an envelope. Unique spikes or fibres protrude from the icosahedral capsid, with knobs that bind to the receptor on the host cell. The linear double-stranded genome is 26 to 48 kb long, encodes 23 to 46 proteins, and has a 55 kDa protein attached to each end.
Adenoviruses infect a broad range of vertebrates, including humans, livestock, horses, dogs, bats and other mammals, as well as birds and reptiles, and usually cause infections of the upper respiratory tract. The virus is very stable. Transmission can occur by respiratory droplets or via faeces, with swimming pools being a common source of infection. A total of 57 serotypes, grouped into 7 species, have been found in humans; they cause a wide range of illnesses including mild respiratory infections, conjunctivitis and gastroenteritis. Some types have been associated with obesity. In people with an immunodeficiency they can cause life-threatening multi-organ disease. No antiviral treatment or vaccine is generally available; hand washing is the best way to prevent infection. Adenoviruses are an important viral vector for gene therapy. An oncolytic adenovirus has been approved in China for the treatment of head and neck cancer.
Portal:Viruses/Selected virus/11
Tobacco mosaic virus (TMV) is an RNA virus in the Virgaviridae family that infects a wide range of plants, including tobacco, tomato, pepper, other members of the Solanaceae family, and cucumber. The rod-shaped virus particle is around 300 nm long and 18 nm in diameter, and consists of a helical capsid made from 2130 copies of a single coat protein, which is wrapped around a positive-sense single-stranded RNA genome of around 6400 bases. The coat protein and RNA can self-assemble to produce infectious virus.
Infection often causes characteristic patterns, such as "mosaic"-like mottling and discoloration on the leaves, but is almost symptomless in some host species. TMV causes an economically important disease in tobacco plants. Transmission is frequently by human handling, and prevention of infection involves destroying infected plants, hand washing and crop rotation to avoid contaminated soil. TMV is one of the most stable viruses known. The fact that it does not infect animals and can readily be produced in gramme amounts has led to its use in numerous pioneering studies in virology and structural biology. TMV was the first virus to be discovered and the first to be crystallised.
Portal:Viruses/Selected virus/12
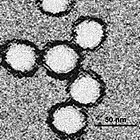
Acanthamoeba polyphaga mimivirus (APMV) is a species of DNA virus in the Mimivirus genus of the Mimiviridae family. It infects the amoeba, Acanthamoeba polyphaga. Its non-enveloped icosahedral capsid is 400 nm in diameter, with protein filaments of 100 nm projecting from its surface. The APMV genome is a linear, double-stranded DNA molecule of around 1.2 megabases, encoding around 979 genes. This is comparable to the genome of some small bacteria. It encodes several proteins that had not been previously discovered in viruses, including aminoacyl tRNA synthetases. APMV is itself parasitised by the Sputnik virophage (arrowed in micrograph).
APMV is as large as some small species of bacteria, such as Rickettsia conorii and Tropheryma whipplei. When it was first discovered in 1992, it was thought to be a bacterium, and named Bradfordcoccus. APMV was not shown to be a virus until 2003, when it was the largest virus then discovered. It has since been overtaken by Megavirus chilensis, Pandoravirus and Pithovirus, all of which also infect amoebae. These and other large and complex DNA viruses are now grouped in Nucleocytoviricota, also termed nucleocytoplasmic large DNA viruses.
Portal:Viruses/Selected virus/13
Alphaviruses are a genus of RNA viruses in the Togaviridae family. The spherical enveloped virion is 70 nm in diameter, with a nucleocapsid of 40 nm. It has a single-stranded, positive-sense RNA genome of 11–12 kb. The genus contains more than thirty species, which infect humans, horses, rodents and other mammals, as well as fish, birds, other vertebrates and invertebrates. Alphaviruses are generally transmitted by insect vectors, predominantly mosquitoes, and are an example of arboviruses (arthropod-borne viruses).
The first alphavirus to be discovered was western equine encephalitis virus, by Karl Friedrich Meyer in 1930, in horses with fatal encephalitis in San Joaquin Valley, California, USA. Some members of the genus cause significant disease in humans, including the chikungunya, o'nyong'nyong, Ross River, Sindbis, Barmah Forest and Semliki Forest (pictured) viruses and the eastern, western and Venezuelan equine encephalitis viruses. Arthritis, encephalitis, rashes and fever are the most frequently observed symptoms. Large mammals such as humans usually form dead-end hosts for the viruses, although Venezuelan equine encephalitis virus is mainly amplified in the horse. No human vaccine or antiviral drug has been licensed. Prevention is predominantly by control of the insect vector.
Portal:Viruses/Selected virus/14
Cauliflower mosaic virus (CaMV) is a plant pararetrovirus in the Caulimoviridae family, which has similarities with hepadnaviruses such as hepatitis B virus. It predominantly infects members of the Brassicaceae (cabbage) family, including cauliflower and turnip; some strains can also infect Datura and Nicotiana species of the Solanaceae family. It is transmitted by aphid vectors, such as Myzus persicae. Symptoms include a mottled leaf pattern called "mosaic", necrotic lesions on the surface of infected leaves, stunted growth and deformation of the overall plant structure.
Although the viral genome is double-stranded DNA, the virus replicates via reverse transcription like a retrovirus. The icosahedral virion is 52 nm in diameter, and is built from 420 capsid protein subunits. The circular 8 kb genome encodes seven proteins, including a movement protein, which facilitates viral movement to neighbouring cells, and an insect transmission factor, which recognises a protein receptor at the tip of the aphid mouthparts (pictured). CaMV has several ways of evading the host defensive responses, which include interrupting salicylic acid-dependent signalling and decoying host silencing machinery. The virus has a strong constitutive (always on) promoter, CaMV 35S, which is widely used in plant genetic engineering.
Portal:Viruses/Selected virus/15
Adeno-associated viruses (AAVs) are two small DNA viruses in the Dependoparvovirus genus of the Parvoviridae family. They cannot complete their lytic replication cycle without a helper virus, which include adenoviruses, herpesviruses and vaccinia. In the absence of the helper, AAVs can integrate into the host genome at a specific site on human chromosome 19, or persist as an episome. The 20 nm icosahedral capsid lacks an envelope, and contains a single-stranded DNA genome of around 4.7 kb. AAVs infect humans and some other primates without causing disease. They generate only a mild immune response, including neutralising antibodies. The best-studied of the 11 serotypes, AAV-2, infects nerve cells, liver cells, skeletal muscle and vascular smooth muscle, using heparan sulphate proteoglycan as its primary receptor.
Its low pathogenicity makes AAV an attractive basis for viral vectors for gene therapy. Alipogene tiparvovec to treat lipoprotein lipase deficiency was the first gene therapy to be licensed, but was later withdrawn. Promising results have been obtained in early clinical trials with AAV-based gene therapy in haemophilia, congestive heart failure, spinal muscular atrophy, Parkinson's disease and the rare eye disease Leber congenital amaurosis.
Portal:Viruses/Selected virus/16
Henipaviruses are a genus of RNA viruses in the Paramyxoviridae family. The variably shaped, 40–600 nm diameter, enveloped capsid contains a single-stranded, negative-sense RNA genome of 18.2 kb, with six genes. The cellular receptor is in the ephrin family. The natural hosts are predominantly bats, mainly the Pteropus genus of megabats (flying foxes) and some microbats. Bats infected with Hendra virus develop viraemia and shed virus in urine, faeces and saliva for around a week, but show no signs of disease. Henipaviruses can also infect humans and livestock, causing severe disease with high mortality, making the group a zoonootic disease. Transmission to humans sometimes occurs via an intermediate domestic animal host.
The first henipavirus, Hendra virus, was discovered in 1994 as the cause of an outbreak in horses in Brisbane, Australia. Nipah virus was identified a few years later in Malaysia as the cause of an outbreak in pigs. Three further species have since been recognised: Cedar and Kumasi viruses in bats, and Mòjiāng virus in rodents. Their emergence as human pathogens has been linked to increased contact between bats and humans. Human disease has been confined to Australia and Asia, but members of the genus have also been found in African bats. A veterinary vaccine against Hendra virus is available but no human vaccine has been licensed.
Portal:Viruses/Selected virus/17
Sputnik virophage is a subviral agent, discovered in 2008, that infects Acanthamoeba protozoa. It is a satellite virus of giant viruses of the Mimiviridae family. It requires a mimivirus to infect the cell simultaneously to replicate, hijacking the virus factories that mimivirus creates and impairing its replication. Sputnik was the first satellite to be shown to inhibit the replication of its associated helper virus. Such viruses have been termed "virophages" or "virus eaters" – by analogy with bacteriophages, viruses that parasitise bacteria – but the distinction between virophages and classical satellite viruses that infect plants, arthropods and mammals is disputed. Three Sputnik types are now known, and other virophages have since been discovered, now classified in the Lavidaviridae family, including the Zamilon, Mavirus and Organic Lake virophages. All virophages that have been characterised infect protists and all rely on nucleocytoplasmic large DNA viruses as helpers.
Sputnik's non-enveloped icosahedral capsid is 74 nm in diameter, and contains a circular double-stranded DNA genome of 18.3 kb. Three of its 21 predicted protein-coding genes are thought to derive from Acanthamoeba polyphaga mimivirus, suggesting that virophages and giant viruses can swap genes during their joint infection of Acanthamoeba, and also that virophages might mediate horizontal gene transfer between giant viruses.
Portal:Viruses/Selected virus/18
Hepatitis delta virus or hepatitis D virus (HDV) is a small virusoid, the sole member of the Deltavirus genus. It infects humans. A subviral satellite, it can only replicate in the presence of a hepatitis B (HBV) helper virus. The spherical virion is 36 nm in diameter, with an envelope containing three HBV proteins. The single-stranded, negative-sense, circular RNA genome of 1679 nucleotides is smaller than that of any known animal virus. It has an unusual base composition for an entity that infects animals, and is extensively bound to itself to form a partially double-stranded, rod-shaped structure. These features have led to suggestions that HDV might be related to viroids, small unencapsidated circular RNAs that infect plants. Unlike viroids, HDV encodes a protein, hepatitis D antigen.
Both HDV and HBV enter liver cells using the sodium/bile acid cotransporter as their receptor. They are mainly transmitted via injecting drug use and blood products. More than 15 million people are infected with both viruses, which is associated with a greater risk of liver complications than HBV infection alone. Around one in five jointly infected patients die. The HBV vaccine protects against HDV.
Portal:Viruses/Selected virus/19
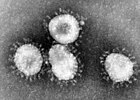
Coronaviruses are a subfamily of RNA viruses in the Nidovirales order which infect mammals and birds. They are spherical enveloped viruses, generally around 80–120 nm in diameter, containing a helical nucleocapsid. Their positive-sense single-stranded RNA genome ranges from approximately 26 to 32 kb in size, one of the largest among RNA viruses. Around 74 characteristic club-shaped spikes project from the envelope, which in electron micrographs resemble the solar corona, from which their name derives. Infectious bronchitis virus was isolated in 1933 from chickens; two mice coronaviruses causing hepatitis and encephalomyelitis were discovered in the 1940s.
Coronaviruses predominantly infect epithelial cells, with the viral spike protein determining tissue tropism and host range. Animal coronaviruses often infect the gastrointestinal tract, causing diarrhoea in cows and pigs, and are transmitted by the faecal–oral route. Human and bird coronaviruses infect the respiratory tract, are transmitted via aerosols and droplets; they cause respiratory tract infections that can range from mild to lethal. Mild illnesses in humans include around 15% of common cold cases, while more lethal coronaviruses cause SARS, MERS and COVID-19. Many human coronaviruses have evolved from viruses of bats.
Portal:Viruses/Selected virus/20
Portal:Viruses/Selected virus/20