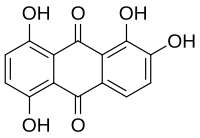
Quinalizarin
Appearance
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,2,5,8-Tetrahydroxyanthracene-9,10-dione | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.243 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H8O6 | |
| Molar mass | 272.212 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Quinalizarin or 1,2,5,8-tetrahydroxyanthraquinone is an organic compound with formula C12H4(OH)4(CO)2. It is one of many tetrahydroxyanthraquinone isomers, formally derived from anthraquinone by replacement of four hydrogen atoms by hydroxyl (OH) groups at the 1, 2, 5, and 8 positions.
Quinalizarin is an inhibitor of the enzyme protein kinase CK2. It is more potent and selective than emodin.[1] It is also a potent catechol O-methyltransferase (COMT) inhibitor.[2][3]
See also
[edit]- 1,4-Dihydroxyanthraquinone (quinizarin)
- Alizarin, a related simpler dye
References
[edit]- ^ Cozza, G.; Mazzorana, M.; Papinutto, E.; Bain, J.; Elliott, M.; di Maira, G.; Gianoncelli, A.; Pagano, M. A.; Sarno, S.; Ruzzene, M.; Battistutta, R.; Meggio, F.; Moro, S.; Zagotto, G.; Pinna, L. A. (2009). "Quinalizarin as a Potent, Selective and Cell-Permeable Inhibitor of Protein Kinase CK2" (PDF). The Biochemical Journal. 421 (3): 387–395. doi:10.1042/BJ20090069. PMID 19432557.
- ^ Schneider J, Huh MM, Bradlow HL, Fishman J (April 1984). "Antiestrogen action of 2-hydroxyestrone on MCF-7 human breast cancer cells". J. Biol. Chem. 259 (8): 4840–5. PMID 6325410.
- ^ Schütze N, Vollmer G, Knuppen R (April 1994). "Catecholestrogens are agonists of estrogen receptor dependent gene expression in MCF-7 cells". J. Steroid Biochem. Mol. Biol. 48 (5–6): 453–61. doi:10.1016/0960-0760(94)90193-7. PMID 8180106.
