Climbazole: Difference between revisions
mNo edit summary |
m →top: added azole refs. Tags: Mobile edit Mobile app edit Android app edit |
||
| Line 36: | Line 36: | ||
}} |
}} |
||
'''Climbazole''' is a topical [[Antifungal medication|antifungal]] agent commonly used in the treatment of human fungal skin infections such as [[dandruff]], [[seborrhoeic dermatitis]] and [[eczema]].<ref name=pmid11534318>{{cite journal | vauthors = Wigger-Alberti W, Kluge K, Elsner P | title = [Clinical effectiveness and tolerance of climbazole containing dandruff shampoo in patients with seborrheic scalp eczema] | journal = Praxis | volume = 90 | issue = 33 | pages = 1346–9 | date = August 2001 | pmid = 11534318 }}</ref> Climbazole has shown a high ''in vitro'' and ''in vivo'' efficacy against ''[[Malassezia spp.]]'' that appear to play an important role in the pathogenesis of dandruff.<ref name=pmid11534318/> Its chemical structure and properties are similar to other fungicides such as [[ketoconazole]] and [[miconazole]]. |
'''Climbazole''' is a topical [[Antifungal medication|antifungal]] agent commonly used in the treatment of human fungal skin infections such as [[dandruff]], [[seborrhoeic dermatitis]] and [[eczema]].<ref name=pmid11534318>{{cite journal | vauthors = Wigger-Alberti W, Kluge K, Elsner P | title = [Clinical effectiveness and tolerance of climbazole containing dandruff shampoo in patients with seborrheic scalp eczema] | journal = Praxis | volume = 90 | issue = 33 | pages = 1346–9 | date = August 2001 | pmid = 11534318 }}</ref> Climbazole has shown a high ''in vitro'' and ''in vivo'' efficacy against ''[[Malassezia spp.]]'' that appear to play an important role in the pathogenesis of dandruff.<ref name=pmid11534318/> Its chemical structure and properties are similar to other [[azole]] fungicides such as [[ketoconazole]], [[clotrimazole]] and [[miconazole]]. |
||
__TOC__ |
__TOC__ |
||
==Indications and formulations== |
==Indications and formulations== |
||
It is most commonly found as an active ingredient in [[over-the-counter substance|OTC]] anti-dandruff and anti-fungal products, including shampoos, lotions and conditioners. It may be accompanied by other active ingredients such as [[zinc pyrithione]] or [[triclosan]].{{cn|date=December 2022}} |
It is most commonly found as an active ingredient in [[over-the-counter substance|OTC]] anti-dandruff and anti-fungal products, including shampoos, lotions and conditioners. It may be accompanied by other active ingredients such as [[zinc pyrithione]] or [[triclosan]].{{cn|date=December 2022}} |
||
Revision as of 04:51, 17 January 2024
 | |
| Clinical data | |
|---|---|
| Routes of administration | topical |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.048.870 |
| Chemical and physical data | |
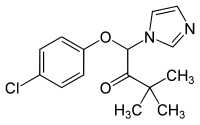
| Formula | C15H17ClN2O2 |
| Molar mass | 292.76 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Climbazole is a topical antifungal agent commonly used in the treatment of human fungal skin infections such as dandruff, seborrhoeic dermatitis and eczema.[2] Climbazole has shown a high in vitro and in vivo efficacy against Malassezia spp. that appear to play an important role in the pathogenesis of dandruff.[2] Its chemical structure and properties are similar to other azole fungicides such as ketoconazole, clotrimazole and miconazole.
Indications and formulations
It is most commonly found as an active ingredient in OTC anti-dandruff and anti-fungal products, including shampoos, lotions and conditioners. It may be accompanied by other active ingredients such as zinc pyrithione or triclosan.[citation needed]
Side effects
May cause localized irritation of the skin with symptoms including redness, rashes and itching.[citation needed]
References
- ^ Chemical Properties of Climbazole Archived 2007-11-13 at the Wayback Machine
- ^ a b Wigger-Alberti W, Kluge K, Elsner P (August 2001). "[Clinical effectiveness and tolerance of climbazole containing dandruff shampoo in patients with seborrheic scalp eczema]". Praxis. 90 (33): 1346–9. PMID 11534318.
