Sucralose
| |

| |
| Names | |
|---|---|
| IUPAC name
1,6-Dichloro-1,6-dideoxy-β-D-fructofuranosyl 4-chloro-4-deoxy-α-D-galactopyranoside
| |
| Systematic IUPAC name
(2R,3R,4R,5R,6R)-2-{[(2R,3S,4S,5S)-2,5-Bis(chloromethyl)-3,4-dihydroxyoxolan-2-yl]oxy}-5-chloro-6-(hydroxymethyl)oxane-3,4-diol | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.054.484 |
| EC Number |
|
| E number | E955 (glazing agents, ...) |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H19Cl3O8 | |
| Molar mass | 397.63 g·mol−1 |
| Appearance | Off-white to white powder |
| Odor | Odorless |
| Density | 1.69 g/cm3 |
| Melting point | 125 °C (257 °F; 398 K) |
| 283 g/L (20 °C) | |
| Acidity (pKa) | 12.52±0.70 |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
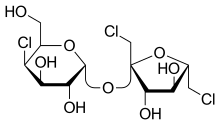
Sucralose is an artificial sweetener and sugar substitute. As the majority of ingested sucralose is not metabolized by the body, it adds no calories.[3] In the European Union, it is also known under the E number E955. It is produced by chlorination of sucrose, selectively replacing three of the hydroxy groups—in the C1 and C6 positions of the fructose portion and the C4 position of the glucose portion—to give a 1,6-dichloro-1,6-dideoxyfructose–4-chloro-4-deoxygalactose disaccharide. Sucralose is about 600 times sweeter than sucrose (table sugar),[4][5] three times as sweet as both aspartame and acesulfame potassium, and twice as sweet as sodium saccharin.[4]
The commercial success of sucralose-based products stems from its favorable comparison to other low-calorie sweeteners in terms of taste, stability, and safety.[4][6] It is commonly sold under the Splenda brand name.[4]
Uses[edit]
Sucralose is used in many food and beverage products because it is a no-calorie sweetener, does not promote dental cavities,[7] is safe for consumption by diabetics and nondiabetics,[8][9] and does not affect insulin levels,[10] although the powdered form of sucralose-based sweetener product Splenda (as most other powdered sucralose products) contains 95% (by volume) bulking agents dextrose and maltodextrin that do affect insulin levels. Sucralose is used as a replacement for (or in combination with) other artificial or natural sweeteners such as aspartame, acesulfame potassium or high-fructose corn syrup. It is used in products such as candy, breakfast bars, coffee pods, and soft drinks. It is also used in canned fruits wherein water and sucralose take the place of much higher calorie corn syrup-based additives. Sucralose mixed with dextrose or maltodextrin (both made from corn) as bulking agents is sold internationally by McNeil Nutritionals under the Splenda brand name. In the United States and Canada, this blend is increasingly found in restaurants in yellow packets.[a]
Cooking[edit]
Sucralose is available in a granulated form that allows same-volume substitution with sugar.[11] This mix of granulated sucralose includes fillers, all of which rapidly dissolve in water. Sucralose is not hygroscopic, which can lead to baked goods that are noticeably drier and manifest a less dense texture than those made with sucrose.[citation needed]
Unlike sucrose, which melts when baked at high temperatures, sucralose maintains its granular structure when subjected to dry, high heat (e.g., in a 180 °C or 350 °F oven). Furthermore, in its pure state, sucralose begins to decompose at 119 °C (246 °F).[12] In some recipes, such as crème brûlée, which require sugar sprinkled on top to partially or fully melt and crystallize, substituting sucralose does not result in the same surface texture, crispness, or crystalline structure.[citation needed]
Safety evaluation[edit]
Sucralose has been accepted as safe by several food safety regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA), the Joint FAO/WHO Expert Committee Report on Food Additives, the European Union's Scientific Committee on Food, Health Protection Branch of Health and Welfare Canada, and Food Standards Australia New Zealand.
Maximum acceptable daily intake[edit]
Various assessments have reported different amounts of maximum acceptable daily intake (ADI), usually measured as mg per kg of body weight. According to the Canadian Diabetes Association, the amount of sucralose that can be consumed over a person's lifetime without any adverse effects is 9 milligrams per kilogram of body weight per day.[13][14] The FDA approval process indicated that consuming sucralose in typical amounts as a sweetener was safe.[14][15] The intake at which adverse effects are seen is 1500 mg/kg BW/day,[15] providing a large margin of safety compared to the estimated daily intake. The European Food Safety Authority (EFSA) proposed an ADI of 5 mg per kg (body weight) while the FDA established it as 15 mg per kg body weight, that is, 350–1050 mg per day for a person of 70 kg.[14]
Metabolism[edit]
Most ingested sucralose is directly excreted in the feces, while about 11–27% is absorbed by the gastrointestinal tract (gut).[14][15] The amount absorbed from the gut is largely removed from the blood by the kidneys and eliminated via urine, with 20–30% of absorbed sucralose being metabolized.[14][15]
Possible health effects[edit]
In reviewing a 1987 food additive petition by McNeil Nutritionals, the FDA stated that "in the 2-year rodent bioassays... there was no evidence of carcinogenic activity for either sucralose or its hydrolysis products".[15][16]
As of 2020, reviews of numerous safety and toxicology studies on sucralose concluded that it is not toxic or carcinogenic, even at levels of daily consumption much larger than those typically used.[14][15] A 2023 FDA summary declared that there were no concerns of sucralose toxicity on cancer risk, metabolism, the reproductive system or the nervous system, and no concern for people with diabetes.[4]
Baking[edit]
At normal baking temperatures, sucralose is heat stable, indicating that it retains its sweetness and is suitable as a sugar substitute for use in baked goods.[4]
History[edit]
Sucralose was discovered in 1976 by scientists from Tate & Lyle, working with researchers Leslie Hough and Shashikant Phadnis at Queen Elizabeth College (now part of King's College London).[17] While researching novel uses of sucrose and its synthetic derivatives, Phadnis was told to "test" a chlorinated sugar compound. According to an anecdotal account, Phadnis thought Hough asked him to "taste" it, so he did and found the compound to be exceptionally sweet.[18]
Tate & Lyle patented the substance in 1976; as of 2008, the only remaining patents concerned specific manufacturing processes.[19]
A Duke University animal study funded by the Sugar Association[20] found evidence that doses of Splenda (containing ~1% sucralose and ~99% maltodextrin by weight) between 100 and 1000 mg/kg BW/day, containing sucralose at 1.1 to 11 mg/kg BW/day, fed to rats reduced gut microbiota, increased the pH level in the intestines, contributed to increases in body weight, and increased levels of P-glycoprotein (P-gp).[21] These effects have not been reported in humans.[5] An expert panel, including scientists from Duke University, Rutgers University, New York Medical College, Harvard School of Public Health, and Columbia University reported in Regulatory Toxicology and Pharmacology that the Duke study was "not scientifically rigorous and is deficient in several critical areas that preclude reliable interpretation of the study results".[22]
Sucralose was first approved for use in Canada in 1991. Subsequent approvals came in Australia in 1993, in New Zealand in 1996, in the United States in 1998, and in the European Union in 2004. By 2008, it had been approved in over 80 countries, including Mexico, Brazil, China, India, and Japan.[23] In 2006, the FDA amended the regulations for foods to include sucralose as a "non-nutritive sweetener" in food.[24] In May 2008, Fusion Nutraceuticals launched a generic product to the market, using Tate & Lyle patents.
In April 2015, PepsiCo announced that it would be moving from aspartame to sucralose for most of its diet drinks in the U.S.[25] due to sales of Diet Pepsi falling by more than 5% in the U.S. The company stated that its decision was a commercial one, responding to consumer preferences.
In February 2018, PepsiCo went back to using aspartame in Diet Pepsi because of an 8% drop in sales for the previous year.[26][27]
Chemistry and production[edit]
Sucralose is a disaccharide composed of 1,6-dichloro-1,6-dideoxyfructose and 4-chloro-4-deoxygalactose. It is synthesized by the selective chlorination of sucrose in a multistep route that substitutes three specific hydroxyl groups with chlorine atoms. This chlorination is achieved by selective protection of one of the primary alcohols as an ester (acetate or benzoate), followed by chlorination with an excess of any of several chlorinating agent to replace the two remaining primary alcohols and one of the secondary alcohols, and then by hydrolysis of the ester.[28][29]
Storage[edit]
Sucralose is stable when stored under normal conditions of temperature, pressure and humidity.[30] Upon prolonged heating during storage at elevated temperatures (38 °C, 100 °F), sucralose may break down, releasing carbon dioxide, carbon monoxide and minor amounts of hydrogen chloride.[30]
Effect on caloric content[edit]
Though sucralose contains no calories, products that contain fillers such as dextrose and/or maltodextrin add about 2–4 calories per teaspoon or individual packet, depending on the product, the fillers used, brand, and the intended use of the product.[31] The FDA allows for any product containing fewer than five calories per serving to be labeled as "zero calories".[32]
Research[edit]
There is no evidence of an effect of sucralose on long-term weight loss or body mass index, with cohort studies showing a minor effect on weight gain and heart disease risks.[33]
Environmental effects[edit]
According to one study, sucralose is digestible by a number of microorganisms and is broken down once released into the environment.[34] However, measurements by the Swedish Environmental Research Institute have shown sewage treatment has little effect on sucralose, which is present in wastewater effluents at levels of several μg/L (ppb).[35] No ecotoxicological effects are known at such levels, but the Swedish Environmental Protection Agency warns a continuous increase in levels may occur if the compound is only slowly degraded in nature. When heated to very high temperatures (over 350 °C or 662 °F) in metal containers, sucralose can produce polychlorinated dibenzo-p-dioxins and other persistent organic pollutants in the resulting smoke.[36]
Sucralose has been detected in natural waters, but research indicates that the levels found in the environment are far below those required to cause adverse effects to certain kinds of aquatic life.[37]
See also[edit]
Footnotes[edit]
References[edit]
- ^ Merck Index, 11th Edition, 8854.
- ^ Anonymous. Scifinder – Substance Detail for 56038-13-2, 30 October 2010.
- ^ "Gestational Diabetes and Low-Calorie Sweeteners: Answers to Common Questions" (PDF). Food Insight. Archived from the original (PDF) on 9 August 2017. Retrieved 15 May 2015.
- ^ a b c d e f "Aspartame and Other Sweeteners in Food". US Food and Drug Administration. 14 July 2023. Retrieved 16 January 2024.
- ^ a b Friedman MA (3 April 1998). "Food Additives Permitted for Direct Addition to Food for Human Consumption; Sucralose" (PDF). Federal Register: 21 CFR Part 172, Docket No. 87F-0086.
Lead Deputy Commissioner for the FDA
- ^ "A Report on Sucralose from the Food Sanitation Council". The Japan Food Chemical Research Foundation. Archived from the original on 15 October 2012.
- ^ Food and Drug Administration (March 2006). "Food labeling: health claims; dietary noncariogenic carbohydrate sweeteners and dental caries. Final rule". Federal Register. 71 (60): 15559–15564. PMID 16572525.
- ^ Grotz VL, Henry RR, McGill JB, Prince MJ, Shamoon H, Trout JR, et al. (December 2003). "Lack of effect of sucralose on glucose homeostasis in subjects with type 2 diabetes". Journal of the American Dietetic Association. 103 (12): 1607–1612. doi:10.1016/j.jada.2003.09.021. PMID 14647086.
- ^ FAP 7A3987, 16 August 1996. pp. 1–357. A 12-week study of the effect of sucralose on glucose homeostasis and HbA1c in normal healthy volunteers, Center for Food Safety and Applied Nutrition, U.S. FDA
- ^ Ford HE, Peters V, Martin NM, Sleeth ML, Ghatei MA, Frost GS, et al. (April 2011). "Effects of oral ingestion of sucralose on gut hormone response and appetite in healthy normal-weight subjects" (PDF). European Journal of Clinical Nutrition. 65 (4): 508–513. doi:10.1038/ejcn.2010.291. PMID 21245879. S2CID 13051016.
- ^ "Splenda Granulated Sweetener – Zero Calorie Sweetener & Sugar Substitute". Splenda. Retrieved 16 January 2024.
- ^ Bannach G, Almeida RR, Lacerda LG, Schnitzler E, Ionashiro M (December 2009). "Thermal stability and thermal decomposition of sucralose" (PDF). Eclética Química. 34 (4): 21–26. doi:10.1590/S0100-46702009000400002.
- ^ "Canadian Diabetes Association 2008 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada" (PDF). Canadian Journal of Diabetes. 32 (Supplement 1): S41. September 2008. Archived from the original (PDF) on 16 May 2012. Retrieved 10 July 2012.
- ^ a b c d e f Magnuson BA, Roberts A, Nestmann ER (August 2017). "Critical review of the current literature on the safety of sucralose". Food and Chemical Toxicology. 106 (Pt A): 324–355. doi:10.1016/j.fct.2017.05.047. PMID 28558975.
- ^ a b c d e f Berry C, Brusick D, Cohen SM, Hardisty JF, Grotz VL, Williams GM (16 November 2016). "Sucralose Non-Carcinogenicity: A Review of the Scientific and Regulatory Rationale". Nutrition and Cancer. 68 (8): 1247–1261. doi:10.1080/01635581.2016.1224366. PMC 5152540. PMID 27652616.
- ^ "Sucralose – FDA Final Rule – Food Additives Permitted for Direct Addition to Food for Human Consumption" (PDF). U.S. Food and Drug Administration. Archived from the original (PDF) on 18 October 2012. Retrieved 17 July 2011.
- ^ "Frequently Asked Questions About Sucralose". Sucralose. Archived from the original on 20 September 2018. Retrieved 20 September 2018.
- ^ Gratzer W (28 November 2002). "5. Light on sweetness: the discovery of aspartame". Eurekas and Euphorias: The Oxford Book of Scientific Anecdotes. Oxford University Press. pp. 32–. Bibcode:2002eueu.book.....G. ISBN 978-0-19-280403-7. Retrieved 1 August 2012.
- ^ "Tate & Lyle loses sucralose patent case". ap-foodtechnology.com.
- ^ Browning L (2 September 2008). "New Salvo in Splenda Skirmish". The New York Times. Retrieved 24 May 2010.
- ^ Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, McLendon RE, Schiffman SS (2008). "Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome p-450 in male rats". Journal of Toxicology and Environmental Health. Part A. 71 (21): 1415–1429. Bibcode:2008JTEHA..71.1415A. doi:10.1080/15287390802328630. PMID 18800291. S2CID 11909980.
- ^ Daniells S (2 September 2009). "Sucralose safety 'scientifically sound': Expert panel".
- ^ "Splenda Brand Sweetener FAQ: Safety & Product Information: What research has been conducted to confirm the safety of SPLENDA". Splenda. McNeil Nutritionals, LLC. Retrieved 29 August 2015.
- ^ Turner J (3 April 2006). "FDA amends regulations that include sucralose as a non-nutritive sweetener in food" (PDF). FDA Consumer. Retrieved 7 September 2007.
- ^ Roberts M (27 April 2015). "Pepsi to ditch artificial sweetener". BBC News.
- ^ "Diet Pepsi Revamp Leaves 'Aspartame Free' Gamble Behind". Beverage Digest. 16 February 2018. Archived from the original on 4 July 2018. Retrieved 4 July 2018.
- ^ Schultz EJ (16 February 2018). "Reversing Course, Diet Pepsi Goes All-In on Aspartame". Advertising Age.
- ^ Fraser-Reid B (2012). From Sugar to Splenda: A Personal and Scientific Journey of a Carbohydrate Chemist and Expert Witness. Berlin: Springer. pp. 199–210. ISBN 9783642227806.
- ^ U.S. patent 5,498,709
- ^ a b "Sucralose – Material Safety Data Sheet" (PDF). Dulcette Technologies, Inc. 10 September 2008. Archived from the original (PDF) on 10 September 2008. Retrieved 25 October 2022.
- ^ Filipic M (3 October 2004). "Chow Line: Sucralose sweet for calorie-counters". Ohio State Human Nutrition. Archived from the original on 29 December 2010.
- ^ "CFR – Code of Federal Regulations Title 21". U.S. Food and Drug Administration. 1 April 2011. Retrieved 11 March 2012.
- ^ Azad MB, Abou-Setta AM, Chauhan BF, Rabbani R, Lys J, Copstein L, et al. (July 2017). "Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies". CMAJ. 189 (28): E929–E939. doi:10.1503/cmaj.161390. PMC 5515645. PMID 28716847.
- ^ Labare MP, Alexander M (1993). "Biodegradation of sucralose in samples of natural environments". Environmental Toxicology and Chemistry. 12 (5): 797–804. doi:10.1897/1552-8618(1993)12[797:BOSACC]2.0.CO;2.
- ^ "Measurements of Sucralose in the Swedish Screening Program 2007, Part I; Sucralose in surface waters and STP samples" (PDF).
- ^ Dong S, Liu G, Hu J, Zheng M (October 2013). "Polychlorinated dibenzo-p-dioxins and dibenzofurans formed from sucralose at high temperatures". Scientific Reports. 3: 2946. Bibcode:2013NatSR...3E2946D. doi:10.1038/srep02946. PMC 3796739. PMID 24126490.
- ^ Stoddard KI, Huggett DB (October 2014). "Early life stage (ELS) toxicity of sucralose to fathead minnows, Pimephales promelas". Bulletin of Environmental Contamination and Toxicology. 93 (4): 383–387. Bibcode:2014BuECT..93..383S. doi:10.1007/s00128-014-1348-9. PMID 25120258. S2CID 5380255.



