Glyphosate

| |

| |

| |
| Names | |
|---|---|
| IUPAC name
N-(phosphonomethyl)glycine
| |
| Other names
2-[(phosphonomethyl)amino]acetic acid
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.012.726 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C3H8NO5P | |
| Molar mass | 169.073 g·mol−1 |
| Appearance | white crystalline powder |
| Density | 1.704 (20 °C) |
| Melting point | 184.5 °C (364.1 °F; 457.6 K) |
| Boiling point | decomposes at 187 °C (369 °F; 460 K) |
| 1.01 g/100 mL (20 °C) | |
| log P | −2.8 |
| Acidity (pKa) | <2, 2.6, 5.6, 10.6 |
| Hazards[1][2] | |
| GHS labelling: | |
 
| |
| Danger | |
| H318, H411 | |
| P273, P280, P305+P351+P338, P310, P501 | |
| Flash point | Non-flammable |
| Safety data sheet (SDS) | InChem MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Glyphosate (N-(phosphonomethyl)glycine) is a broad-spectrum systemic herbicide and crop desiccant. It is an organophosphorus compound, specifically a phosphonate. It is used to kill weeds, especially annual broadleaf weeds and grasses that compete with crops. It was discovered to be an herbicide by Monsanto chemist John E. Franz in 1970.[3] Monsanto brought it to market in 1974 under the trade name Roundup, and Monsanto's last commercially relevant United States patent expired in 2000.
Farmers quickly adopted glyphosate, especially after Monsanto introduced glyphosate-resistant Roundup Ready crops, enabling farmers to kill weeds without killing their crops. In 2007, glyphosate was the most used herbicide in the United States' agricultural sector and the second-most used in home and garden, government and industry, and commerce.[4] By 2016 there was a 100-fold increase from the late 1970s in the frequency of applications and volumes of glyphosate-based herbicides (GBHs) applied, partly in response to the unprecedented global emergence and spread of glyphosate-resistant weeds.[5]: 1
Glyphosate is absorbed through foliage, and minimally through roots,[6][7][8] and transported to growing points. It inhibits a plant enzyme involved in the synthesis of three aromatic amino acids: tyrosine, tryptophan, and phenylalanine. Therefore, it is effective only on actively growing plants and is not effective as a pre-emergence herbicide. An increasing number of crops have been genetically engineered to be tolerant of glyphosate (e.g. Roundup Ready soybean, the first Roundup Ready crop, also created by Monsanto) which allows farmers to use glyphosate as a postemergence herbicide against weeds. The development of glyphosate resistance in weed species is emerging as a costly problem. While glyphosate and formulations such as Roundup have been approved by regulatory bodies worldwide, concerns about their effects on humans and the environment persist.[5][9]
Many regulatory and scholarly reviews have evaluated the relative toxicity of glyphosate as an herbicide. The German Federal Institute for Risk Assessment toxicology review in 2013 found that "the available data is contradictory and far from being convincing" with regard to correlations between exposure to glyphosate formulations and risk of various cancers, including non-Hodgkin lymphoma (NHL).[10] A meta-analysis published in 2014 identified an increased risk of NHL in workers exposed to glyphosate formulations.[11] In March 2015 the World Health Organization's International Agency for Research on Cancer classified glyphosate as "probably carcinogenic in humans" (category 2A) based on epidemiological studies, animal studies, and in vitro studies.[9][12][13] In November, 2015, the European Food Safety Authority published an updated assessment report on glyphosate, concluding that "the substance is unlikely to be genotoxic (i.e. damaging to DNA) or to pose a carcinogenic threat to humans." Furthermore, the final report clarified that while other, probably carcinogenic, glyphosate-containing formulations may exist, studies "that look solely at the active substance glyphosate do not show this effect."[14][15] In May 2016, the Joint FAO/WHO Meeting on Pesticide Residues concluded that "glyphosate is unlikely to pose a carcinogenic risk to humans from exposure through the diet", even at doses as high as 2,000 mg/kg body weight orally.[16]
Discovery
Glyphosate was first synthesized in 1950 by Swiss chemist Henry Martin, who worked for the Swiss company Cilag. The work was never published.[17]: 1 Stauffer Chemical patented the agent as a chemical chelator in 1964 as it binds and removes minerals such as calcium, magnesium, manganese, copper, and zinc.[18]
Somewhat later, glyphosate was independently discovered at Monsanto in 1970. Monsanto chemists had synthesized about 100 analogs of aminomethylphosphonic acid as potential water-softening agents. Two were found to have weak herbicidal activity, and John E. Franz, a chemist at Monsanto, was asked to try to make analogs with stronger herbicidal activity. Glyphosate was the third analog he made.[17]: 1–2 [19][20][21] Franz received the National Medal of Technology in 1987 and the Perkin Medal for Applied Chemistry in 1990 for his discoveries.[22][23]
In 2008, USDA-ARS scientist Stephen O. Duke and Stephen B. Powles, an Australian weed expert — described glyphosate as a "virtually ideal" herbicide.[24] In 2010 Powles stated: "glyphosate is a one in a 100-year discovery that is as important for reliable global food production as penicillin is for battling disease."[25]
Chemistry
Glyphosate is an aminophosphonic analogue of the natural amino acid glycine, and like all amino acids, exists in different ionic states depending on pH. Both the phosphonic acid and carboxylic acid moieties can be ionised and the amine group can be protonated and the substance exists as a series of zwitterions. Glyphosate is soluble in water to 12 g/l at room temperature. The original synthetic approach to glyphosate involved the reaction of phosphorus trichloride with formaldehyde followed by hydrolysis to yield a phosphonate. Glycine is then reacted with this phosphonate to yield glyphosate, and its name is taken as a contraction of the compounds used in this synthesis - viz. glycine and a phosphonate.[26]
- PCl3 + H2CO → Cl2P(=O)-CH2Cl
- Cl2P(=O)-CH2Cl + 2 H2O → (HO)2P(=O)-CH2Cl + 2 HCl
- (HO)2P(=O)-CH2Cl + H2N-CH2-COOH → (HO)2P(=O)-CH2-NH-CH2-COOH + HCl
The main deactivation path for glyphosate is hydrolysis to aminomethylphosphonic acid.[27]
Industrial synthesis
Two main approaches are used to synthesize glyphosate industrially. The first is to react iminodiacetic acid with phosphorous acid and hydrochloric acid (sometimes formed in situ by addition of phosphorus trichloride) via a modified Mannich reaction. Oxidation then leads to the desired glyphosate product. Iminodiacetic acid is usually prepared on-site, such as by reaction of chloroacetic acid with ammonia and calcium hydroxide to produce the calcium iminodiacetate salt and then acidification of the product.[17]
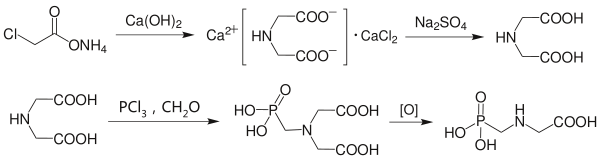
The chloroacetic acid approach is less efficient than other iminodiacetic acid approaches, owing to the production of calcium chloride waste and decreased yield. When hydrogen cyanide is readily available as a by-product (say), an alternative approach is to use iminodiacetonitrile, HN(CH2CN)2, and diethanolamine is also a suitable starting material.[17]
The second involves the use of dimethyl phosphite in a one-pot synthesis. Glycine and paraformaldehyde are reacted in a suitable organic solvent (typically triethylamine and methanol) to produce bishydroxymethylglycine, (HOCH2)2NCH2COOH. Dimethyl phosphite is then introduced and work-up with hydrochloric acid cleaves the hydroxymethyl group from the nitrogen atom whilst heating speeds the hydrolysis of both phosphate ester linkages.[17]

This synthetic approach is responsible for a substantial portion of the production of glyphosate in China, with considerable work having gone into recycling the triethylamine and methanol.[17] Progress has also been made in attempting to eliminate the need for triethylamine altogether.[28]
Mode of action
Glyphosate kills plants by interfering with the synthesis of the aromatic amino acids phenylalanine, tyrosine, and tryptophan. It does this by inhibiting the enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), which catalyzes the reaction of shikimate-3-phosphate (S3P) and phosphoenolpyruvate to form 5-enolpyruvyl-shikimate-3-phosphate (EPSP).[29] Glyphosate is absorbed through foliage and minimally through roots, meaning that it is only effective on actively growing plants and cannot prevent seeds from germinating.[7][8] After application, glyphosate is readily transported around the plant to growing roots and leaves and this systemic activity is important for its effectiveness.[24][17] Inhibiting the enzyme causes shikimate to accumulate in plant tissues and diverts energy and resources away from other processes. While growth stops within hours of application, it takes several days for the leaves to begin turning yellow.[30]
EPSP is subsequently dephosphorylated to chorismate, an essential precursor for the amino acids mentioned above.[31] These amino acids are used in protein synthesis and to produce secondary metabolites such as folates, ubiquinones, and naphthoquinone.
X-ray crystallographic studies of glyphosate and EPSPS show that glyphosate functions by occupying the binding site of the phosphoenolpyruvate, mimicking an intermediate state of the ternary enzyme substrates complex.[32][33] Glyphosate inhibits the EPSPS enzymes of different species of plants and microbes at different rates.[34][35]
EPSPS is produced only by plants and microbes; the gene coding for it is not in the mammalian genome.[36][37]
Environmental fate
Glyphosate adsorbs strongly to soil, and residues are expected to generally be immobile in soil. Ground and surface water pollution is limited.[38] Glyphosate is readily degraded by soil microbes to aminomethylphosphonic acid (AMPA, which like glyphosate strongly adsorbs to soil solids and is thus unlikely to leach to groundwater). Though both glyphosate and AMPA are commonly detected in water bodies, a portion of the AMPA detected may actually be the result of degradation of detergents rather than from glyphosate.[39] Glyphosate does have the potential to contaminate surface waters due to its aquatic use patterns and through erosion, as it adsorbs to soil particles suspended in runoff. The mechanism of glyphosate sorption to soil is similar to that of phosphate fertilizers, the presence of which can reduce glyphosate sorption.[40] Phosphate fertilizers are subject to release from sediments into water bodies under anaerobic conditions, and similar release can also occur with glyphosate, though significant impact of glyphosate release from sediments has not been established.[41] Limited leaching can occur after high rainfall after application. If glyphosate reaches surface water, it is not broken down readily by water or sunlight.[42][43]
The half-life of glyphosate in soil ranges between 2 and 197 days; a typical field half-life of 47 days has been suggested. Soil and climate conditions affect glyphosate's persistence in soil. The median half-life of glyphosate in water varies from a few to 91 days.[7] At a site in Texas, half-life was as little as three days. A site in Iowa had a half-life of 141 days.[44] The glyphosate metabolite AMPA has been found in Swedish forest soils up to two years after a glyphosate application. In this case, the persistence of AMPA was attributed to the soil being frozen for most of the year.[45] Glyphosate adsorption to soil, and later release from soil, varies depending on the kind of soil.[46][47] Glyphosate is generally less persistent in water than in soil, with 12- to 60-day persistence observed in Canadian ponds, although persistence of over a year has been recorded in the sediments of American ponds.[42] The half-life of glyphosate in water is between 12 days and 10 weeks.[48]
According to the National Pesticide Information Center fact sheet, glyphosate is not included in compounds tested for by the Food and Drug Administration's Pesticide Residue Monitoring Program, nor in the United States Department of Agriculture's Pesticide Data Program. However, a field test showed that lettuce, carrots, and barley contained glyphosate residues up to one year after the soil was treated with 3.71 lb of glyphosate per acre (4.15 kg per hectare).[7] The U.S. has determined the acceptable daily intake of glyphosate at 1.75 milligrams per kilogram of bodyweight per day (mg/kg/bw/day) while the European Union has set it at 0.3.[49]
Use

Glyphosate is effective in killing a wide variety of plants, including grasses and broadleaf and woody plants. By volume, it is one of the most widely used herbicides.[7] In 2007, glyphosate was the most used herbicide in the United States agricultural sector, with 180 to 185 million pounds (82,000 to 84,000 tonnes) applied, the second-most used in home and garden with 5 to 8 million pounds (2,300 to 3,600 tonnes) and government applied 13 to 15 million pounds (5,900 to 6,800 tonnes) in industry and commerce.[4] It is commonly used for agriculture, horticulture, viticulture, and silviculture purposes, as well as garden maintenance (including home use).[7][50] It has a relatively small effect on some clover species and morning glory.[51]

Glyphosate and related herbicides are often used in invasive species eradication and habitat restoration, especially to enhance native plant establishment in prairie ecosystems. The controlled application is usually combined with a selective herbicide and traditional methods of weed eradication such as mulching to achieve an optimal effect.[52]
In many cities, glyphosate is sprayed along the sidewalks and streets, as well as crevices in between pavement where weeds often grow. However, up to 24% of glyphosate applied to hard surfaces can be run off by water.[53] Glyphosate contamination of surface water is attributed to urban and agricultural use.[54] Glyphosate is used to clear railroad tracks and get rid of unwanted aquatic vegetation.[8] Since 1994, glyphosate has been used in aerial spraying in Colombia in coca eradication programs; Colombia announced in May 2015 that by October, it would cease using glyphosate in these programs due to concerns about human toxicity of the chemical.[55]
In addition to its use as an herbicide, glyphosate is also used for crop desiccation (siccation) to increase harvest yield,[8] and as a result of desiccation, to increase sucrose concentration in sugarcane before harvest.[56]
Genetically modified crops
Some micro-organisms have a version of 5-enolpyruvoyl-shikimate-3-phosphate synthetase (EPSPS) resistant to glyphosate inhibition. A version of the enzyme that was both resistant to glyphosate and that was still efficient enough to drive adequate plant growth was identified by Monsanto scientists after much trial and error in an Agrobacterium strain called CP4, which was found surviving in a waste-fed column at a glyphosate production facility.[35][57][58]: 56 This CP4 EPSPS gene was cloned and transfected into soybeans. In 1996, genetically modified soybeans were made commercially available.[59] Current glyphosate-resistant crops include soy, maize (corn), canola, alfalfa, sugar beets, and cotton, with wheat still under development.
In 2015, 89% of corn, 94% of soybeans, and 89% of cotton produced in the US were genetically modified to be herbicide-tolerant.[60][clarification needed]
Formulations and tradenames
Glyphosate is marketed in the United States and worldwide by many agrochemical companies, in different solution strengths and with various adjuvants, under dozens of tradenames.[61][62][63][64] As of 2010, more than 750 glyphosate products were on the market.[65] In 2012, in terms of volume about half of the total global consumption of glyphosate was for conventional crops; Asia Pacific was the largest and fastest growing market.[66] Chinese manufacturers collectively are the world's largest producers of glyphosate and its precursors[67] and account for about 30% of global exports.[66] Key manufacturers include Anhui Huaxing Chemical Industry Company, BASF, Bayer CropScience, Dow AgroSciences, DuPont, Jiangsu Good Harvest-Weien Agrochemical Company, Monsanto, Nantong Jiangshan Agrochemical & Chemicals Co., Nufarm Limited, SinoHarvest, Syngenta, and Zhejiang Xinan Chemical Industrial Group Company.[66]
Glyphosate is an acid molecule, so it is formulated as a salt for packaging and handling. Various salt formulations include isopropylamine, diammonium, monoammonium, or potassium as the counterion. Some brands include more than one salt. Some companies report their product as acid equivalent (ae) of glyphosate acid, or some report it as active ingredient (ai) of glyphosate plus the salt, and others report both. To compare performance of different formulations, knowledge of how the products were formulated is needed. Given that different salts have different weights, the acid equivalent is a more accurate method of expressing and comparing concentrations.
Adjuvant loading refers to the amount of adjuvant[68][69] already added to the glyphosate product. Fully loaded products contain all the necessary adjuvants, including surfactant; some contain no adjuvant system, while other products contain only a limited amount of adjuvant (minimal or partial loading) and additional surfactants must be added to the spray tank before application.[70] As of 2000 (just before Monsanto's patent on glyphosate expired), over 400 commercial adjuvants from over 34 different companies were available for use in commercial agriculture.[71][72]
Products are supplied most commonly in formulations of 120, 240, 360, 480, and 680 g/l of active ingredient. The most common formulation in agriculture is 360 g/l, either alone or with added cationic surfactants.[62]
For 360 g/l formulations, European regulations allow applications of up to 12 l/ha for control of perennial weeds such as couch grass. More commonly, rates of 3 l/ha are practiced for control of annual weeds between crops.[73]
Monsanto

Monsanto developed and patented the use of glyphosate to kill weeds in the early 1970s, and first brought it to market in 1974, under the Roundup brandname.[24][74] While its initial patent[75] expired in 1991, Monsanto retained exclusive rights in the United States until its patent[76] on the isopropylamine salt expired in September 2000.[77]
As of 2009, sales of these herbicide products represented about 10% of Monsanto's revenue due to competition from other producers of other glyphosate-based herbicides;[78] their Roundup products (which include GM seeds) represented about half of Monsanto's gross margin.[79]
The active ingredient of the Monsanto herbicides is the isopropylamine salt of glyphosate. Another important ingredient in some formulations is the surfactant polyethoxylated tallow amine.
Monsanto also produces seeds which grow into plants genetically engineered to be tolerant to glyphosate. The genes contained in these seeds are patented. Such crops allow farmers to use glyphosate as a postemergence herbicide against most broadleaf and cereal weeds. Soy was the first glyphosate-resistant crop.
Toxicity
Glyphosate is the active ingredient in herbicide formulations containing it. However, in addition to glyphosate salts, commercial formulations of glyphosate contain additives such as surfactants which vary in nature and concentration. The surfactants are added to enable the glyphosate to penetrate the cuticle of the plants. Toxicologists have studied glyphosate alone and formulations.
Glyphosate alone
Humans
Many regulatory and scholarly reviews have evaluated the relative toxicity of glyphosate as an herbicide. The German Federal Institute for Risk Assessment toxicology review in 2013 found that "the available data is contradictory and far from being convincing" with regard to correlations between exposure to glyphosate formulations and risk of various cancers, including non-Hodgkin lymphoma.[10]
Early epidemiological studies did not find associations between long-term, low-level exposure to glyphosate and any disease.[80][81][82] A 2000 review concluded that "under present and expected conditions of new use, there is no potential for Roundup herbicide to pose a health risk to humans".[83] A 2002 review by the European Union reached the same conclusion.[84] In 2013 the European commission reviewed a 2002 finding that had concluded equivocal evidence existed of a relationship between glyphosate exposure during pregnancy and cardiovascular malformations and found that "there is no increased risk at the levels of exposure below those that caused maternal toxicity."[85] A 2013 review found that neither glyphosate nor typical glyphosate-based formulations pose a genotoxicity risk in humans under normal conditions of human or environmental exposures.[86]
A 2014 review article reported a significant association between B-cell lymphoma and glyphosate occupational exposure.[11] In March 2015, the World Health Organization's International Agency for Research on Cancer classified glyphosate as "probably carcinogenic in humans" (category 2A) based on epidemiological studies, animal studies, and in vitro studies.[9][12][13] However, in 2016 a joint meeting of the United Nations (FAO) Panel of Experts on Pesticide Residues in Food and the Environment and the World Health Organization Core Assessment Group on Pesticide Residues concluded that based on the available evidence "glyphosate is unlikely to pose a carcinogenic risk to humans from exposure through the diet".[87]
Other mammals
Amongst mammals, glyphosate is considered to have "low to very low toxicity". The LD50 of glyphosate is 5,000 mg/kg for rats, 10,000 mg/kg in mice and 3,530 mg/kg in goats. The acute dermal LD50 in rabbits is greater than 2,000 mg/kg. Indications of glyphosate toxicity in animals typically appear within 30 to 120 minutes following ingestion of a large enough dose, and include initial excitability and tachycardia, ataxia, depression, and bradycardia, although severe toxicity can develop into collapse and convulsions.[7]
A review of unpublished short-term rabbit-feeding studies reported severe toxicity effects at 150 mg/kg/day and "no observed adverse effect level" doses ranging from 50 to 200 mg/kg/day.[88]
Glyphosate can have carcinogenic effects in nonhuman mammals. These include the induction of positive trends in the incidence of renal tubule carcinoma and haemangiosarcoma in male mice, and increased pancreatic islet-cell adenoma in male rats.[12]
Glyphosate-based herbicides may cause life-threatening arrhythmias in mammals. Evidence also shows that such herbicides cause direct electrophysiological changes in the cardiovascular systems of rats and rabbits.[89]
Other aquatic fauna
In freshwater invertebrates (species unspecified), glyphosate has a 48-hour LC50 ranging from 55 to 780 ppm. The 96-hour LC50 is 281 ppm for grass shrimp (Palaemonetas vulgaris) and 934 ppm for fiddler crabs (Uca pagilator). These values make glyphosate "slightly toxic to practically non-toxic".[7]
Antimicrobial activity
The antimicrobial activity of glyphosate has been described in the microbiology literature since its discovery in 1970 and the description of glyphosate's mechanism of action in 1972. Efficacy was described for numerous bacteria and fungi.[90] Glyphosate can control the growth of apicomplexan parasites, such as Toxoplasma gondii, Plasmodium falciparum (malaria), and Cryptosporidium parvum, and has been considered an antimicrobial agent in mammals.[91] Inhibition can occur with some Rhizobium species important for soybean nitrogen fixation, especially under moisture stress.[92]
Soil biota
When glyphosate comes into contact with the soil, it can be bound to soil particles, thereby slowing its degradation.[42][44] Glyphosate and its degradation product, aminomethylphosphonic acid, residues are considered to be much more benign toxicologically and environmentally than most of the herbicides replaced by glyphosate.[94][relevant?]
Other studies found that while "recommended dosages of glyphosate did not affect growth rates", much higher dosages reduced respiration in nitrogen-fixing bacteria.[95][96]
Glyphosate can be harmful to some earthworms such as Apporectodea caliginosa, even at very low doses,[97] and causes a significant reduction in growth of the earthworm Eisenia fetida.[98]
Glyphosate-based formulations
Glyphosate-based formulations may contain a number of adjuvants, the identities of which are considered trade secrets.[99] Surfactants are used in herbicide formulations as wetting agents, to maximize coverage and aid penetration of the herbicide(s) through plant leaves. As agricultural spray adjuvants, surfactants may be mixed into commercial formulations, such as Roundup, or they may be purchased separately and mixed on-site (tank mix).
Polyethoxylated tallow amine (POEA) is a surfactant used in the original Roundup formulation and was still being commonly used in 2015.[100] Different versions of Roundup have included different percentages of POEA. Although Monsanto product fact sheets do not disclose surfactants and their percentages, a 1997 US government report said that Roundup is 15% POEA while Roundup Pro is 14.5%.[101] A review of the literature provided to the EPA in 1997 found that POEA was more toxic to fish than glyphosate was.[101] POEA is more toxic to fish and amphibians than glyphosate alone.[101][102]
Human
A 2000 review concluded that "under present and expected conditions of new use, there is no potential for Roundup herbicide to pose a health risk to humans".[83] A 2002 review by the European Union reached the same conclusion.[84]
A 2012 meta-analysis of all epidemiological studies of exposure to glyphosate formulations found no correlation with any kind of cancer.[81] The 2013 systematic review by the German Institute for Risk Assessment of epidemiological studies of workers who use pesticides, exposed to glyphosate formulations found no significant risk, stating that "the available data are contradictory and far from being convincing".[10]: Volume 1, p64-66 However, a 2014 meta-analysis of the same studies found a correlation between occupational exposure to glyphosate formulations and increased risk of B cell lymphoma, the most common kind of non-Hodgkin lymphoma. Workers exposed to glyphosate were about twice as likely to get B cell lymphoma.[11]
Acute toxicity is dose-related; Skin exposure to ready-to-use glyphosate formulations can cause irritation, and photocontact dermatitis has been occasionally reported. These effects are probably due to the preservative benzisothiazolin-3-one. Severe skin burns are very rare.[103] Inhalation is a minor route of exposure, but spray mist may cause oral or nasal discomfort, an unpleasant taste in the mouth, or tingling and irritation in the throat. Eye exposure may lead to mild conjunctivitis. Superficial corneal injury is possible if irrigation is delayed or inadequate.[103] Death has been reported after deliberate overdose.[103][104] Ingestion of Roundup ranging from 85 to 200 ml (of 41% solution) has resulted in death within hours of ingestion, although it has also been ingested in quantities as large as 500 ml with only mild or moderate symptoms.[105] Consumption of over 85 ml of concentrated product are likely to cause serious symptoms in adults including burns due to corrosive effects as well as kidney and liver damage. More severe cases cause "respiratory distress, impaired consciousness, pulmonary edema, infiltration on chest X-ray, shock, arrhythmias, renal failure requiring haemodialysis, metabolic acidosis, and hyperkalaemia" and death is often preceeded by bradycardia and ventricular arrhythmias.[103]
Other animals
A 2000 review of the ecotoxicological data on Roundup shows at least 58 studies exist on the effects of Roundup on a range of organisms.[93] This review concluded, "...for terrestrial uses of Roundup minimal acute and chronic risk was predicted for potentially exposed non-target organisms".
In reproductive toxicity studies performed in rats and rabbits, no adverse maternal or offspring effects were seen at doses below 175–293 mg/kg of body weight per day.[7]
Fish
Monsanto and other companies produce glyphosate products with alternative surfactants specifically formulated for aquatic use, for example the Monsanto products "Biactive" and "AquaMaster".[106][107] Glyphosate formulations are much more toxic for amphibians and fish than glyphosate alone.[101][102] The half-life of POEA (21–42 days) is longer than that for glyphosate (7–14 days) in aquatic environments.[108]
Amphibians
Some researchers have suggested the toxicity effects of pesticides on amphibians may be different from those of other aquatic fauna because of their lifestyle; amphibians may be more susceptible to the toxic effects of pesticides because they often prefer to breed in shallow, lentic, or ephemeral pools. These habitats do not necessarily constitute formal water-bodies and can contain higher concentrations of pesticide compared to larger water-bodies.[102][109] Studies in a variety of amphibians have shown the toxicity of GBFs containing POEA to amphibian larvae. These effects include interference with gill morphology and mortality from either the loss of osmotic stability or asphyxiation. At sub-lethal concentrations, exposure to POEA or glyphosate/POEA formulations have been reported to be associated with delayed development, accelerated development, reduced size at metamorphosis, developmental malformations of the tail, mouth, eye and head, histological indications of intersex and symptoms of oxidative stress.[102]
A 2003 study of various formulations of glyphosate found, "[the] risk assessments based on estimated and measured concentrations of glyphosate that would result from its use for the control of undesirable plants in wetlands and over-water situations showed that the risk to aquatic organisms is negligible or small at application rates less than 4 kg/ha and only slightly greater at application rates of 8 kg/ha."[110]
A 2013 meta-analysis reviewed the available data related to potential impacts of glyphosate-based herbicides on amphibians. According to the authors, the use of glyphosate-based pesticides cannot be considered the major cause of amphibian decline, the bulk of which occurred prior to the widespread use of glyphosate or in pristine tropical areas with minimal glyphosate exposure. The authors recommended further study of species- and development-stage chronic toxicity, of environmental glyphosate levels, and ongoing analysis of data relevant to determining what if any role glyphosate might be playing in worldwide amphibian decline, and suggest including amphibians in standardized test batteries.[111]
Other aquatic fauna
Glyphosate-based formulations can cause oxidative stress in bullfrog tadpoles and Pacific oysters.[112]
Effect on plant health
A correlation was found between an increase in the infection rate of wheat by Fusarium head blight and the application of glyphosate, but "because of the nature of this study, we could not determine if the association between previous GF (glyphosate formulation) use and FHB development was a cause-effect relationship".[113] Other studies have found causal relationships between glyphosate and decreased disease resistance.[114] Exposure to glyphosate has been shown to change the species composition of endophytic bacteria in plant hosts, which is highly variable.[115]
Endocrine disruption
In 2007, the EPA selected glyphosate for further screening through its Endocrine Disruptor Screening Program (EDSP). Selection for this program is based on a compound's prevalence of use and does not imply particular suspicion of endocrine activity.[116] On June 29, 2015 the EPA released Weight of Evidence Conclusion of the EDSP Tier 1 screening for glyphosate, recommending that glyphosate not be considered for Tier 2 testing. The Weight of Evidence conclusion stated "...there was no convincing evidence of potential interaction with the estrogen, androgen or thyroid pathways."[117]
Genetic damage
Several studies have not found mutagenic effects,[118] so glyphosate has not been listed in the United States Environmental Protection Agency or the International Agency for Research on Cancer databases.[119] Various other studies suggest glyphosate may be mutagenic.[119] The IARC monograph noted that glyphosate-based formulations can cause DNA strand breaks in various taxa of animals in vitro[112]
Government and organization positions
European Food Safety Authority
A 2013 systematic review by the German Institute for Risk Assessment (BfR) examined more than 1000[120] epidemiological studies, animal studies, and in vitro studies. It found that "no classification and labelling for carcinogenicity is warranted" and did not recommend a carcinogen classification of either 1A or 1B.[10]: 139, 34–37 It provided the review to EFSA in January 2014 which published it in December 2014.[10][121][122] On November, 12th, 2015, EFSA published its conclusion on the risk assessment of glyphosate, stating it was "unlikely to pose a carcinogenic hazard to humans".[123]
EFSA's decision and the BfR report were criticized in an open letter published by 96 scientists in November 2015 saying that the BfR report failed to adhere to accepted scientific principles of open and transparent procedures.[124][125] The BfR report included unpublished data, lacked authorship, omitted references, and did not disclose conflict-of-interest information.[125]
On April 4, 2016, Dr. Vytenis Andriukaitis, European Commissioner for Health and Food Safety, wrote an open letter to the Chair of the Board of the Glyphosate Task at Monsanto Europe asking to publish the full studies provided to the EFSA.[126]
US Environmental Protection Agency
The EPA, which last reviewed glyphosate in 1993, considers glyphosate to be noncarcinogenic and relatively low in dermal and oral acute toxicity.[42] The EPA considered a "worst case" dietary risk model of an individual eating a lifetime of food derived entirely from glyphosate-sprayed fields with residues at their maximum levels. This model indicated that no adverse health effects would be expected under such conditions.[42] In 2015, the EPA initiated a review glyphosate's toxicity and in 2016 reported their conclusion that glyphosate is likely not carcinogenic.[9][127]
International Agency for Research on Cancer
In March 2015, the International Agency for Research on Cancer published a summary of their forthcoming monograph on glyphosate, and classified glyphosate as "probably carcinogenic in humans" (category 2A) based on epidemiological studies, animal studies, and in vitro studies. It noted that there was "limited evidence" of carcinogenicity in humans for non-Hodgkin lymphoma.[9][12][13][128] The IARC classifies substances for their carcinogenic potential, and "a few positive findings can be enough to declare a hazard, even if there are negative studies, as well." Unlike the BfR, it does not conduct a so-called risk assessment weighing benefits against risk.[129]
The BfR responded that IARC reviewed only a selection of what they had reviewed earlier, and argued that other studies, including a cohort study called 'Agricultural Health Study', do not support the classification.[130] The IARC report did not include the German regulatory study published in December 2014, nor did it include industry-funded studies.[citation needed] Monsanto called the IARC report biased and said it wanted it to be retracted.[131] It started a case against California's carcinogen classification in 2016.[132]
Effects of use
Emergence of resistant weeds
In the 1990s, when the first genetically modified crops-such as glyphosate-resistant corn, canola, soybean and cotton—were introduced,[133][134] no glyphosate-resistant weeds existed.[135] By 2014, glyphosate-resistant weeds dominated herbicide-resistant research. At that time, 23 glyphosate-resistant species were found in 18 countries.[136]
"Resistance evolves after a weed population has been subjected to intense selection pressure in the form of repeated use of a single herbicide."[135][137] Weeds resistant to the herbicide have been called 'superweeds'.[138]
According to Ian Heap, a weed specialist, who completed his PhD on resistance to multiple herbicides in annual ryegrass (Lolium rigidum) in 1988[139] –the first case of an herbicide-resistant weed in Australia[140]–by 2014 the Lolium rigidum was the "world’s worst herbicide-resistant weed with instances in "12 countries, 11 sites of action, 9 cropping regimens" and affecting over 2 million hectares.[136] Annual ryegrass was known to be resistant to herbicides since 1982. By 1996, the first documented case of glyphosate-resistant L. rigidum was reported in Australia in 1996 near Orange, New South Wales.[141][142][143] In 2006, farmers associations were reporting 107 biotypes of weeds within 63 weed species with herbicide resistance.[144] In 2009, Canada identified its first resistant weed, giant ragweed, and at that time 15 weed species had been confirmed as resistant to glyphosate.[137][145] As of 2010, in the United States 7 to 10 million acres (2.8 to 4.0 million hectares) of soil were afflicted by superweeds, or about 5% of the 170 million acres planted with corn, soybeans, and cotton, the crops most affected, in 22 states.[146] In 2012, Charles Benbrook reported that the Weed Science Society of America listed 22 superweeds in the U.S., with over 5.7×106 ha (14×106 acres) infested by GR weeds and that Dow AgroSciences had carried out a survey and reported a figure of around 40×106 ha (100×106 acres).[147] The International Survey of Herbicide Resistant Weeds database lists species that are resistant to glyphosate.[148]
In response to resistant weeds, farmers are hand-weeding, using tractors to turn over soil between crops, and using other herbicides in addition to glyphosate.
Monsanto scientists have found that some resistant weeds have as many as 160 extra copies of a gene called EPSPS, the enzyme glyphosate disrupts.[149]
Palmer amaranth

In 2004, a glyphosate-resistant variation of Amaranthus palmeri, commonly known as Palmer amaranth, was found in Georgia and confirmed by a 2005 study.[150] In 2005, resistance was also found in North Carolina.[151] Widespread use of Roundup Ready crops led to an unprecedented selection pressure, and glyphosate resistance followed.[151] The weed variation is now widespread in the southeastern United States.[152] Cases have also been reported in Texas[152] and Virginia.[153]
Conyza

Conyza bonariensis (also known as hairy fleabane and buva) and Conyza canadensis (known as horseweed or marestail), are other weed species that had lately developed glyphosate resistance.[154][155][156] A 2008 study on the current situation of glyphosate resistance in South America concluded "resistance evolution followed intense glyphosate use" and the use of glyphosate-resistant soybean crops is a factor encouraging increases in glyphosate use.[157] In the 2015 growing season, glyphosate-resistant marestail proved to be especially problematic to control in Nebraska production fields.[158]
Ryegrass

Glyphosate-resistant ryegrass (Lolium) has occurred in most of the Australian agricultural areas and other areas of the world. All cases of evolution of resistance to glyphosate in Australia were characterized by intensive use of the herbicide while no other effective weed control practices were used. Studies indicate the resistant ryegrass does not compete well against nonresistant plants and their numbers decrease when not grown under conditions of glyphosate application.[159]
Johnson grass
Glyphosate-resistant Johnson grass (Sorghum halepense) is found in glyphosate-resistant soybean cultivation in northern Argentina.[160]
Monarch butterfly
Use of glyphosate to clear milkweed along roads and fields may have contributed to a decline in monarch butterfly populations in the Midwest.[161] Along with deforestation and adverse weather conditions,[162] the decrease in milkweed contributed to an 81% decline in monarchs.[163][164] The Natural Resources Defense Council (NRDC) filed a suit in 2015 against the EPA, in which it is argued that the agency ignored warnings about the dangers of glyphosate usage for monarchs.[165]
Legal status
Glyphosate was first approved for use in the 1970s, and as of 2010 was labelled for use in 130 countries.[17]: 2
In September 2013, the legislative assembly of El Salvador approved legislation to ban 53 agrochemicals, including glyphosate; the ban on glyphosate was set to begin in 2015.[166][167][168]
In April 2014, the legislature of the Netherlands passed legislation prohibiting sale of glyphosate to individuals for use at home; commercial sales were not affected.[169]
In May 2015, the president of Sri Lanka banned the use and import of glyphosate, effective immediately.[170][171]
In May 2015, Bermuda blocked importation on all new orders of glyphosate-based herbicides for a temporary suspension awaiting outcomes of research.[172]
In May 2015, Colombia announced that it would stop using glyphosate by October 2015 in the destruction of illegal plantations of coca, the raw ingredient for cocaine. Farmers have complained that the aerial fumigation has destroyed entire fields of coffee and other legal produce.[173]
In June 2015, the French Ecology Minister asked nurseries and garden centers to halt over-the-counter sales of glyphosate in the form of Monsanto's Roundup. This was a nonbinding request and all sales of glyphosate remain legal in France until 2022, when the substance will be banned for home gardening.[174]
A vote on the relicencing of glyphosate in the EU stalled in March 2016. Member states France, Sweden, and the Netherlands objected to the renewal.[175] A vote to reauthorize on a temporary basis failed in June 2016[176] but at the last-minute the license was extended for 18 months and will be re-evaluated at the end of 2017.[177]
Legal cases
Advertising controversy
The New York Times reported that in 1996, "Dennis C. Vacco, the Attorney General of New York, ordered the company Monsanto to pull ads that said Roundup was "safer than table salt" and "practically nontoxic" to mammals, birds and fish. The company withdrew the spots, but also said that the phrase in question was permissible under E.P.A. guidelines."[178]
In 2001, French environmental and consumer rights campaigners brought a case against Monsanto for misleading the public about the environmental impact of its herbicide Roundup, on the basis that glyphosate, Roundup's main component, is classed as "dangerous for the environment" and "toxic for aquatic organisms" by the European Union. Monsanto's advertising for Roundup had presented it as biodegradable and as leaving the soil clean after use. In 2007, Monsanto was convicted of false advertising and was fined 15,000 euros. Monsanto's French distributor Scotts France was also fined 15,000 euros. Both defendants were ordered to pay damages of 5,000 euros to the Brittany Water and Rivers Association and 3,000 euros to the Consommation Logement Cadre de vie, one of the two main general consumer associations in France.[179] Monsanto appealed and the court upheld the verdict; Monsanto appealed again to the French Supreme Court, and in 2009 it also upheld the verdict.[180]
Trade dumping allegations
United States companies have cited trade issues with glyphosate being dumped into the western world market areas by Chinese companies and a formal dispute was filed in 2010.[181][182]
See also
- Monsanto legal cases
- 2,4-Dichlorophenoxyacetic acid
- Ammonium sulfamate
- Atrazine
- Environmental impact of pesticides
- Health effects of pesticides
- Integrated pest management
- Séralini affair
References
- ^ a b Glyphosate, Environmental Health Criteria monograph No. 159, Geneva: World Health Organization, 1994, ISBN 92-4-157159-4
- ^ Index no. 607-315-00-8 of Annex VI, Part 3, to Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. OJEU L353, 31.12.2008, pp 1–1355 at pp 570, 1100..
- ^ US patent 3799758, Franz JE, "N-phosphonomethyl-glycine phytotoxicant compositions", issued 1974-03-26, assigned to Monsanto Company
- ^ a b United States EPA 2007 Pesticide Market Estimates Agriculture, Home and Garden
- ^ a b Myers, John Peterson; Antoniou, Michael N.; Blumberg, Bruce; Carroll, Lynn; Colborn, Theo; Everett, Lorne G.; Hansen, Michael; Landrigan, Philip J.; Lanphear, Bruce P.; Mesnage, Robin; Vandenberg, Laura N.; vom Saal, Frederick S.; Welshons, Wade V.; Benbroo, Charles M. (February 17, 2016). "Concerns over use of glyphosate-based herbicides and risks associated with exposures: a consensus statement". 15 (19). Environmental Health: 13. doi:10.1186/s12940-016-0117-0. Retrieved March 24, 2016.
{{cite journal}}: Cite journal requires|journal=(help); Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help)CS1 maint: unflagged free DOI (link) - ^ Sprankle P, Meggitt WF, Penner D (1975). "Rapid inactivation of glyphosate in the soil". Weed Science: 224–228.
- ^ a b c d e f g h i "Glyphosate technical fact sheet (revised June 2015)". National Pesticide Information Center. 2010. Retrieved September 1, 2015.
- ^ a b c d "The agronomic benefits of glyphosate in Europe" (PDF). Monsanto Europe SA. February 2010. Retrieved June 2, 2013.
- ^ a b c d e Cressey, Daniel (March 25, 2015). "Widely used herbicide linked to cancer". Nature. doi:10.1038/nature.2015.17181.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ a b c d e Renewal Assessment Report: Glyphosate. Volume 1. Report and Proposed Decision. December 18, 2013. German Institute for Risk Assessment, page 65. Downloaded from http://dar.efsa.europa.eu/dar-web/provision (registration required)
- ^ a b c Schinasi L, Leon ME (April 2014). "Non-Hodgkin lymphoma and occupational exposure to agricultural pesticide chemical groups and active ingredients: a systematic review and meta-analysis". International Journal of Environmental Research and Public Health. 11 (4): 4449–527. doi:10.3390/ijerph110404449. PMC 4025008. PMID 24762670.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c d Guyton KZ, Loomis D, Grosse Y, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Scoccianti C, Mattock H, Straif K (May 2015). "Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate". The Lancet. Oncology. 16 (5): 490–1. doi:10.1016/S1470-2045(15)70134-8. PMID 25801782.
- ^ a b c "Press release: IARC Monographs Volume 112: evaluation of five organophosphate insecticides and herbicides" (PDF). International Agency for Research on Cancer, World Health Organization. March 20, 2015.
- ^ "European Food Safety Authority - Glyphosate report" (PDF). EFSA. Retrieved May 23, 2016.
- ^ "Glyphosate: EFSA updates toxicological profile | European Food Safety Authority". www.efsa.europa.eu. Retrieved May 23, 2016.
- ^ "JOINT FAO/WHO MEETING ON PESTICIDE RESIDUES" (PDF). May 2016. Retrieved October 3, 2016.
- ^ a b c d e f g h Dill, Gerald M.; Sammons, R. Douglas; Feng, Paul C.; Kohn, Frank; Kretzmer, Keith; Mehrsheikh, Akbar; Bleeke, Marion; Honegger, Joy L.; Farmer, Donna; Wright, Dan; Haupfear, Eric A. (2010). "Glyphosate: Discovery, Development, Applications, and Properties" (PDF). In Nandula, Vijay K. (ed.). Glyphosate Resistance in Crops and Weeds: History, Development, and Management. Hoboken, N.J.: Wiley. ISBN 978-0-470-41031-8.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ United States Patent 3,160,632 (1964) Stauffer Chemical
- ^ Alibhai MF, Stallings WC (March 2001). "Closing down on glyphosate inhibition — with a new structure for drug discovery". Proceedings of the National Academy of Sciences of the United States of America. 98 (6): 2944–6. Bibcode:2001PNAS...98.2944A. doi:10.1073/pnas.061025898. JSTOR 3055165. PMC 33334. PMID 11248008.
- ^ Monsanto's John E. Franz Wins 1990 Perkin Medal Chem. Eng. News, 1990, 68 (11), pp. 29–30 doi:10.1021/cen-v068n011.p029
- ^ Pesticide Action Network UK.Glyphosate fact sheet Pesticides News No.33, September 1996, pp. 28-29 PAN-UK says it is "a welcome move away from chemicals which are highly toxic to humans and other nontarget organisms, and from chemicals which cause direct and lasting damage to the environment" and of course cautions against overuse.
- ^ "The National Medal of Technology and Innovation Recipients - 1987". The United States Patent and Trademark Office. Retrieved November 29, 2012.
- ^ Stong C (May 1990). "People: Monsanto Scientist John E. Franz Wins 1990 Perkin Medal For Applied Chemistry". The Scientist. 4 (10): 28.
- ^ a b c Duke, Stephen O.; Powles, Stephen B. (2008), "Glyphosate: a once-in-a-century herbicide: Mini-review" (PDF), Pest Management Science, vol. 64, pp. 319–325
{{citation}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Powles, Stephen B. (January 19, 2010). "Gene amplification delivers glyphosate-resistant weed evolution". Proceedings of the National Academy of Sciences. pp. 955–956. doi:10.1073/pnas.0913433107. Retrieved March 24, 2016.
- ^ Chenier, Philip J. (2012). Survey of Industrial Chemistry (3rd ed.). Springer Science+Business Media. p. 384. ISBN 9781461506034.
- ^ Schuette J. "Environmental Fate of Glyphosate" (PDF). Department of Pesticide Regulation, State of California.
- ^ Zhou J, Li J, An R, Yuan H, Yu F (2012). "Study on a New Synthesis Approach of Glyphosate". J. Agric. Food Chem. 60 (25): 6279–6285. doi:10.1021/jf301025p.
- ^ Steinrücken HC, Amrhein N (June 1980). "The herbicide glyphosate is a potent inhibitor of 5-enolpyruvyl-shikimic acid-3-phosphate synthase". Biochemical and Biophysical Research Communications. 94 (4): 1207–12. doi:10.1016/0006-291X(80)90547-1. PMID 7396959.
- ^ Hock, Bertold; Elstner, Erich F. (September 28, 2004). Plant Toxicology, Fourth Edition. CRC Press. pp. 292–296. ISBN 978-0-203-02388-4.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Purdue University, Department of Horticulture and Landscape Architecture, Metabolic Plant Physiology Lecture notes, Aromatic amino acid biosynthesis, The shikimate pathway – synthesis of chorismate.
- ^ Schönbrunn E, Eschenburg S, Shuttleworth WA, Schloss JV, Amrhein N, Evans JN, Kabsch W (February 2001). "Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail". Proceedings of the National Academy of Sciences of the United States of America. 98 (4): 1376–80. Bibcode:2001PNAS...98.1376S. doi:10.1073/pnas.98.4.1376. PMC 29264. PMID 11171958.
- ^ Glyphosate bound to proteins in the Protein Data Bank
- ^ Schulz, A.; Krüper, A.; Amrhein, N. (1985). "Differential sensitivity of bacterial 5-enolpyruvylshikimate-3-phosphate synthases to the herbicide glyphosate". FEMS Microbiology Letters. 28 (3): 297–301. doi:10.1111/j.1574-6968.1985.tb00809.x.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ a b Pollegioni L, Schonbrunn E, Siehl D (August 2011). "Molecular basis of glyphosate resistance-different approaches through protein engineering". The FEBS Journal. 278 (16): 2753–66. doi:10.1111/j.1742-4658.2011.08214.x. PMC 3145815. PMID 21668647.
- ^ Funke T, Han H, Healy-Fried ML, Fischer M, Schönbrunn E (August 2006). "Molecular basis for the herbicide resistance of Roundup Ready crops". Proceedings of the National Academy of Sciences of the United States of America. 103 (35): 13010–5. Bibcode:2006PNAS..10313010F. doi:10.1073/pnas.0603638103. JSTOR 30050705. PMC 1559744. PMID 16916934.
- ^ Maeda H, Dudareva N (2012). "The shikimate pathway and aromatic amino Acid biosynthesis in plants". Annual Review of Plant Biology. 63: 73–105. doi:10.1146/annurev-arplant-042811-105439. PMID 22554242.
The AAA pathways consist of the shikimate pathway (the prechorismate pathway) and individual postchorismate pathways leading to Trp, Phe, and Tyr.... These pathways are found in bacteria, fungi, plants, and some protists, but are absent in animals. Therefore, AAAs and some of their derivatives (vitamins) are essential nutrients in the human diet, although in animals Tyr can be synthesized from Phe by Phe hydroxylase....The absence of the AAA pathways in animals also makes these pathways attractive targets for antimicrobial agents and herbicides.
- ^ Borggaard, OK and AL Gimsing. 2008. Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: a review. Pest Management Sci. 64:441–456.
- ^ Botta, F , G Lavisonb, G Couturier, F Alliot, E Moreau-Guigon, N Fauchon, B Guery, M Chevreuil, and H Blanchoud. 2009. Transfer of glyphosate and its degradate AMPA to surface waters through urban sewerage systems. Chemosphere 77 (1): 133–139.
- ^ Muniraa, S , A Farenhorsta, D Flatena, and C Grant. 2016. Phosphate fertilizer impacts on glyphosate sorption by soil. Chemosphere 153: 471–477.
- ^ Kanissery, RG, A Welsh, and GK Sims. 2014. Effect of soil aeration and phosphate addition on the microbial bioavailability of 14C-glyphosate. J. Environ. Quality 44:137-144. doi:10.2134/jeq2014.08.0331
- ^ a b c d e "Registration Decision Fact Sheet for Glyphosate (EPA-738-F-93-011)" (PDF). R.E.D. FACTS. United States Environmental Protection Agency. 1993.
- ^ Borggaard OK, Gimsing AL (April 2008). "Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: a review". Pest Management Science. 64 (4): 441–56. doi:10.1002/ps.1512. PMID 18161065.
- ^ a b Andréa MM, Peres TB, Luchini LC, Bazarin S, Papini S, Matallo MB, Savoy VL (2003). "Influence of repeated applications of glyphosate on its persistence and soil bioactivity". Pesquisa Agropecuária Brasileira. 38 (11): 1329–1335. doi:10.1590/S0100-204X2003001100012.
- ^ Torstensson NT, Lundgren LN, Stenström J (October 1989). "Influence of climatic and edaphic factors on persistence of glyphosate and 2,4-D in forest soils". Ecotoxicology and Environmental Safety. 18 (2): 230–9. doi:10.1016/0147-6513(89)90084-5. PMID 2806176.
- ^ Albers CN, Banta GT, Hansen PE, Jacobsen OS (October 2009). "The influence of organic matter on sorption and fate of glyphosate in soil--comparing different soils and humic substances". Environmental Pollution. 157 (10): 2865–70. doi:10.1016/j.envpol.2009.04.004. PMID 19447533.
- ^ Ole K, Borggaard OK (2011). "Does phosphate affect soil sorption and degradation of glyphosate? - A review". Trends in Soil Science and Plant Nutrition. 2 (1): 17–27.
- ^ Sparling DW, Matson C, Bickham J, Doelling-Brown P (2006). "Toxicity of glyphosate as Glypro® and LI700 to red‐eared slider (Trachemys scripta elegans) embryos and early hatchlings". Environmental Toxicology and Chemistry. 25 (10): 2768–2774. doi:10.1897/05-152.1.
- ^ Carey Gillam (Nov. 2016). "Alarming Levels of Glyphosate Found in Popular American Foods". EcoWatch.com. Retrieved 14 November 2016.
- ^ Amrein, Thomas M (December 21, 2012). "Analysis of pesticides in food" (PDF). ETH Zurich. p. 15. Retrieved June 2, 2013.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Knezevic, Stevan Z. (February 2010). "Use of Herbicide-Tolerant Crops as Part of an Integrated Weed Management Program". University of Nebraska Extension Integrated Weed Management Specialist. Archived from the original on June 15, 2010.
{{cite web}}: Unknown parameter|dead-url=ignored (|url-status=suggested) (help); Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Nyamai, Priscilla A.; Prather, Timothy S.; Wallace, John M. (2011). "Evaluating Restoration Methods across a Range of Plant Communities Dominated by Invasive Annual Grasses to Native Perennial Grasses". Invasive Plant Science and Management. 4: 306–316. doi:10.1614/IPSM-D-09-00048.1.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Luijendijk CD, Beltman WH, Smidt RA, van der Pas LJ, Kempenaar C (May 2005). "Measures to reduce glyphosate runoff from hard surfaces" (PDF). Plant Research International B.V. Wageningen.
- ^ Botta F, Lavison G, Couturier G, Alliot F, Moreau-Guigon E, Fauchon N, Guery B, Chevreuil M, Blanchoud H (September 2009). "Transfer of glyphosate and its degradate AMPA to surface waters through urban sewerage systems". Chemosphere. 77 (1): 133–9. doi:10.1016/j.chemosphere.2009.05.008. PMID 19482331.
- ^ BBC. May 10, 2015. Colombia to ban coca spraying herbicide glyphosate
- ^ "Sugarcane Ripener Recommendations - Glyphosate". Louisiana State University Agricultural Extension Office. September 3, 2014. Retrieved September 3, 2014.
- ^ Green JM, Owen MD (June 2011). "Herbicide-resistant crops: utilities and limitations for herbicide-resistant weed management". Journal of Agricultural and Food Chemistry. 59 (11): 5819–29. doi:10.1021/jf101286h. PMC 3105486. PMID 20586458.
- ^ Rashid A (2009). Introduction to Genetic Engineering of Crop Plants: Aims and Achievements. I K International. p. 259. ISBN 978-93-80026-16-9.
- ^ "Company History". Web Site. Monsanto Company.
- ^ "Adoption of Genetically Engineered Crops in the U.S." Economic Research Service. USDA. Retrieved August 26, 2015.
- ^ Farm Chemicals International Glyphosate entry in Crop Protection Database
- ^ a b Alberta Agriculture and Rural Development. April 26, 2006. Quick Guide to Glyphosate Products - Frequently Asked Questions
- ^ Hartzler, Bob. "Glyphosate: a Review". ISU Weed Science Online. Iowa State University Extension.
- ^ Tu M, Hurd C, Robison R, Randall JM (November 1, 2001). "Glyphosate" (PDF). Weed Control Methods Handbook. The Nature Conservancy.
- ^ National Pesticide Information Center. Last updated September 2010 Glyphosate General Fact Sheet
- ^ a b c Reuters. Apr 30, 2014. Press Release: Research and Markets: Global Glyphosate Market for Genetically Modified and Conventional Crops 2013 - 2019
- ^ China Research & Intelligence, June 5, 2013. Research Report on Global and China Glyphosate Industry, 2013-2017
- ^ Tu M, Randall JM (June 1, 2003). "Glyphosate" (PDF). Weed Control Methods Handbook. The Nature Conservancy.
- ^ Curran WS, McGlamery MD, Liebl RA, Lingenfelter DD (1999). "Adjuvants for Enhancing Herbicide Performance". Penn State Extension.
- ^ VanGessel M. "Glyphosate Formulations". Control Methods Handbook, Chapter 8, Adjuvants: Weekly Crop Update. University of Delaware Cooperative Extension.
- ^ Sprague C, Hager A (May 12, 2000). "Principles of Postemergence Herbicides". University of Illinois Extension Service. Retrieved November 29, 2012.
- ^ Young B. "Adjuvant Products by Manufacturer, Compendium of Herbicide Adjuvants". Southern Illinois University.
- ^ e-phy: Le catalogue des produits phytopharmaceutiques et de leurs usages des matières fertilisantes et des supports de culture homologués en France
- ^ "History of Monsanto's Glyphosate Herbicides" (PDF). Monsanto. Retrieved December 20, 2015.
- ^ US application 3799758, John E. Franz, "N-phosphonomethyl-glycine phytotoxicant compositions", published 1974-03-26, assigned to Monsanto Company
- ^ US application 4405531, John E. Franz, "Salts of N-phosphonomethylglycine", published 1983-09-20, assigned to Monsanto Company
- ^ Fernandez, Ivan (May 15, 2002). "The Glyphosate Market: A 'Roundup'". Frost & Sullivan. Archived from the original on March 4, 2016. Retrieved March 10, 2015.
- ^ "The debate over whether Monsanto is a corporate sinner or saint". The Economist. November 19, 2009. Retrieved November 20, 2009.
- ^ Cavallaro M (June 26, 2009). "The Seeds Of A Monsanto Short Play". Forbes. Retrieved July 11, 2009.
- ^ Mink PJ, Mandel JS, Lundin JI, Sceurman BK (November 2011). "Epidemiologic studies of glyphosate and non-cancer health outcomes: a review". Regulatory Toxicology and Pharmacology. 61 (2): 172–84. doi:10.1016/j.yrtph.2011.07.006. PMID 21798302.
- ^ a b Mink PJ, Mandel JS, Sceurman BK, Lundin JI (August 2012). "Epidemiologic studies of glyphosate and cancer: a review". Regulatory Toxicology and Pharmacology. 63 (3): 440–52. doi:10.1016/j.yrtph.2012.05.012. PMID 22683395.
- ^ Williams AL, Watson RE, DeSesso JM (2012). "Developmental and reproductive outcomes in humans and animals after glyphosate exposure: a critical analysis". Journal of Toxicology and Environmental Health. Part B, Critical Reviews. 15 (1): 39–96. doi:10.1080/10937404.2012.632361. PMID 22202229.
- ^ a b Williams GM, Kroes R, Munro IC (April 2000). "Safety evaluation and risk assessment of the herbicide Roundup and its active ingredient, glyphosate, for humans". Regulatory Toxicology and Pharmacology. 31 (2 Pt 1): 117–65. doi:10.1006/rtph.1999.1371. PMID 10854122.
- ^ a b "Review report for the active substance glyphosate" (PDF). Commission working document. European Commission, Health and Protection Directorate-General: Directorate E – Food Safety: plant health, animal health and welfare, international questions: E1 - Plant Health. January 21, 2002.
- ^ Kimmel GL, Kimmel CA, Williams AL, DeSesso JM (February 2013). "Evaluation of developmental toxicity studies of glyphosate with attention to cardiovascular development". Critical Reviews in Toxicology. 43 (2): 79–95. doi:10.3109/10408444.2012.749834. PMC 3581053. PMID 23286529.
- ^ Kier LD, Kirkland DJ (April 2013). "Review of genotoxicity studies of glyphosate and glyphosate-based formulations". Critical Reviews in Toxicology. 43 (4): 283–315. doi:10.3109/10408444.2013.770820. PMID 23480780.
- ^ "JOINT FAO/WHO MEETING ON PESTICIDE RESIDUES" (PDF). May 2016. Retrieved June 2016.
{{cite web}}: Check date values in:|access-date=(help) - ^ Kimmel, Gary (2013). "Evaluation of developmental toxicity studies of glyphosate with attention to cardiovascular development". Critical Reviews in Toxicology. 43 (2): 79–95. doi:10.3109/10408444.2012.749834. PMC 3581053. PMID 23286529.
- ^ Gress S, Lemoine S, Séralini GE, Puddu PE (April 2015). "Glyphosate-based herbicides potently affect cardiovascular system in mammals: review of the literature". Cardiovascular Toxicology. 15 (2): 117–26. doi:10.1007/s12012-014-9282-y. PMID 25245870.
- ^ Abraham William (Wildwood. Glyphosate formulations and their use for inhibition of 5-enolpyrovylshikimate-3-phosphate synthase, US Patent 7, 771, 736 B2; 2010
- ^ Roberts CW, Roberts F, Lyons RE, Kirisits MJ, Mui EJ, Finnerty J, Johnson JJ, Ferguson DJ, Coggins JR, Krell T, Coombs GH, Milhous WK, Kyle DE, Tzipori S, Barnwell J, Dame JB, Carlton J, McLeod R (February 2002). "The shikimate pathway and its branches in apicomplexan parasites". The Journal of Infectious Diseases. 185 (Suppl 1): S25–36. doi:10.1086/338004. PMID 11865437.
- ^ Zablotowicz RM, Reddy KN (2004). "Impact of glyphosate on the Bradyrhizobium japonicum symbiosis with glyphosate-resistant transgenic soybean: a minireview". Journal of Environmental Quality. 33 (3): 825–31. doi:10.2134/jeq2004.0825. PMID 15224916.
- ^ a b Giesy JP, Dobson S, Solomon KR (2000). "Ecotoxicological risk assessment for Roundup® herbicide". Reviews of Environmental Contamination and Toxicology. 167: 35–120. doi:10.1007/978-1-4612-1156-3_2. ISBN 978-0-387-95102-7.
- ^ Cerdeira AL, Duke SO (January 2010). "Effects of glyphosate-resistant crop cultivation on soil and water quality". GM Crops. 1 (1): 16–24. doi:10.4161/gmcr.1.1.9404. PMID 21912208.
- ^ Santos A, Flores M (1995). "Effects of glyphosate on nitrogen fixation of free-living heterotrophic bacteria". Letters in Applied Microbiology. 20 (6): 349–52. doi:10.1111/j.1472-765X.1995.tb01318.x.
- ^ Yamada, T; Kremer, RJ; de Camargo e Castro, PR; Wood, BW (2009). "Glyphosate interactions with physiology, nutrition, and diseases of plants: Threat to agricultural sustainability?". European Journal of Agronomy. 31 (3). Elsevier: 111–113. doi:10.1016/j.eja.2009.07.004.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Paoletti MG (1999). "The role of earthworms for assessment of sustainability and as bioindicators". In Paoletti MG (ed.). Invertebrate Biodiversity as Bioindicators of Sustainable Landscapes: Practical Use of Invertebrates to Assess Sustainable Land Use. Elsevier. pp. 137–156.
- ^ Yasmin S, D'Souza D (2010). "Effects of pesticides on the growth and reproduction of earthworm: a review". Applied and Environmental Soil Science. 2010: 1–9. doi:10.1155/2010/678360.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Pesticide Registration Manual | Pesticide Registration | US EPA".
- ^ "Measuring POEA, a Surfactant Mixture in Herbicide Formulations". U.S. Geological Survey.
- ^ a b c d Gary L. Diamond and Patrick R. Durkin February 6, 1997, under contract from the United States Department of Agriculture. Effects of Surfactants on the Toxicity of Glyphosate, with Specific Reference to RODEO
- ^ a b c d Mann RM, Hyne RV, Choung CB, Wilson SP (2009). "Amphibians and agricultural chemicals: Review of the risks in a complex environment". Environmental Pollution. 157 (11): 2903–2927. doi:10.1016/j.envpol.2009.05.015.
- ^ a b c d Bradberry SM, Proudfoot AT, Vale JA (2004). "Glyphosate poisoning". Toxicological Reviews. 23 (3): 159–67. doi:10.2165/00139709-200423030-00003. PMID 15862083.
- ^ Sribanditmongkol P, Jutavijittum P, Pongraveevongsa P, Wunnapuk K, Durongkadech P (September 2012). "Pathological and toxicological findings in glyphosate-surfactant herbicide fatality: a case report". The American Journal of Forensic Medicine and Pathology. 33 (3): 234–7. doi:10.1097/PAF.0b013e31824b936c. PMID 22835958.
- ^ Talbot AR, Shiaw MH, Huang JS, Yang SF, Goo TS, Wang SH, Chen CL, Sanford TR (January 1991). "Acute poisoning with a glyphosate-surfactant herbicide ('Roundup'): a review of 93 cases". Human & Experimental Toxicology. 10 (1): 1–8. doi:10.1177/096032719101000101. PMID 1673618.
- ^ "Response to "The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities"" (PDF). Backgrounder. Monsanto Company. April 1, 2005.
- ^ "Aquatic Use of Glyphosate Herbicides in Australia" (PDF). Backgrounder. Monsanto Company. May 1, 2003.
- ^ Mesnage R, Defarge N, Spiroux de Vendômois J, Séralini GE (2015). "Potential toxic effects of glyphosate and its commercial formulations below regulatory limits". Food Chem. Toxicol. 84: 133–53. doi:10.1016/j.fct.2015.08.012. PMID 26282372.
- ^ Govindarajulu PP (2008). "Literature Review of Impacts of Glyphosate Herbicide on Amphibians: What Risks can the Silvicultural Use of this Herbicide Pose for Amphibians in BC?". British Columbia, Ecosystems Branch, Ministry of Environment. Retrieved December 12, 2015.
- ^ Solomon KR, Thompson DG (2003). "Ecological risk assessment for aquatic organisms from over-water uses of glyphosate". Journal of Toxicology and Environmental Health. Part B, Critical Reviews. 6 (3): 289–324. doi:10.1080/10937400306468. PMID 12746143.
- ^ Wagner N, Reichenbecher W, Teichmann H, Tappeser B, Lötters S (August 2013). "Questions concerning the potential impact of glyphosate-based herbicides on amphibians". Environmental Toxicology and Chemistry / SETAC. 32 (8): 1688–700. doi:10.1002/etc.2268. PMID 23637092.
- ^ a b "IARC monograph on glyphosate" (PDF). IARC. Archived from the original (PDF) on September 5, 2015. Retrieved September 27, 2015.
- ^ Fernandez MR, Selles F, Gehl D, Depauw RM, Zentner RP (2005). "Crop Production Factors Associated with Fusarium Head Blight in Spring Wheat in Eastern Saskatchewan". Crop Science. 45 (5): 1908–16. doi:10.2135/cropsci2004.0197.
- ^ Duke SO, Wedge DE, Cerdeira AL, Matallo MB (2007). "Interactions of Synthetic Herbicides with Plant Disease and Microbial Herbicides". In Vurro M, Gressel J (eds.). Novel Biotechnologies for Biocontrol Agent Enhancement and Management. NATO Security through Science Series. pp. 277–96. doi:10.1007/978-1-4020-5799-1_15. ISBN 978-1-4020-5797-7.
{{cite book}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Rosenblueth M, Martínez-Romero E (August 2006). "Bacterial endophytes and their interactions with hosts". Molecular Plant-Microbe Interactions. 19 (8): 827–37. doi:10.1094/MPMI-19-0827. PMID 16903349.
- ^ United States Environmental Protection Agency (June 18, 2007). "Draft List of Initial Pesticide Active Ingredients and Pesticide Inerts to be Considered for Screening under the Federal Food, Drug, and Cosmetic Act" (PDF). Federal Register. 72 (116): 33486–503.
- ^ United States Environmental Protection Agency (June 29, 2015). "Memorandum: EDSP Weight of Evidence Conclusions on the Tier 1 Screening Assays for the List 1 Chemicals" (PDF).
- ^ ToxNet. Glyposate. National Library of Medicine.
- ^ a b András Székács and Béla Darvas. Forty years with glyphosate. In: Herbicides - Properties, Synthesis and Control of Weeds", Ed. Mohammed Naguib Abd El-Ghany Hasaneen, ISBN 978-953-307-803-8, Published: January 13, 2012.
- ^ http://www.bfr.bund.de/en/press_information/2014/03/glyphosate__no_more_poisonous_than_previously_assumed__although_a_critical_view_should_be_taken_of_certain_co_formulants-188898.html
- ^ "Glyphosate RAR 01 Volume 1 2013-12-18 San". Renewal Assessment Report. Hungry4Pesticides. December 18, 2013. Retrieved March 27, 2015.
- ^ Bundesinstitut für Risikobewertung. Updated 15 January 2014 Frequently asked questions on the health assessment of glyphosate
- ^ "Conclusion on the peer review of the pesticide risk assessment of the active substance glyphosate" (PDF). EFSA Journal. 13 (11): 4302. 2015. doi:10.2903/j.efsa.2015.4302.
- ^ "Independent scientists warn over Monsanto herbicide". DW. December 1, 2015. Retrieved December 9, 2015.
- ^ a b Portier, C. J.; et al. (November 27, 2015). "Open letter: Review of the Carcinogenicity of Glyphosate by EFSA and BfR" (PDF). Retrieved December 9, 2015.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ "Open letter. Subject: Plant protection products - transparency in the context of the decision-making process on glyphosate" (PDF). European Commission. April 4, 2016. Retrieved May 16, 2016.
- ^ Charles, Dan (September 17, 2016). "EPA Weighs In On Glyphosate, Says It Doesn't Cause Cancer". NPR. Retrieved September 19, 2016.
- ^ Specter, Michael (April 10, 2015). "Roundup and Risk Assessment". New Yorker.
'Probable' means that there was enough evidence to say it is more than possible, but not enough evidence to say it is a carcinogen," Aaron Blair, a lead researcher on the IARC's study, said. Blair, a scientist emeritus at the National Cancer Institute, has studied the effects of pesticides for years. "It means you ought to be a little concerned about" glyphosate, he said.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Pollack, Andrew (March 27, 2015). "Weed Killer, Long Cleared, Is Doubted". New York Times.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ "Löst glyphosat Krebs aus? (announcement 007/2015)" (PDF) (in German). German Institute for Risk Assessment. March 23, 2015.
- ^ Gillam, Carey (March 24, 2015). "Monsanto seeks retraction for report linking herbicide to cancer". Reuters.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Reuters (January 21, 2016). "Monsanto Sues California Over Herbicide Classification". The New York Times. ISSN 0362-4331. Retrieved January 25, 2016.
{{cite news}}:|last=has generic name (help) - ^ James, Clive (1996). "Global Review of the Field Testing and Commercialization of Transgenic Plants: 1986 to 1995" (PDF). The International Service for the Acquisition of Agri-biotech Applications. Retrieved July 17, 2010.
- ^ "Roundup Ready System". Monsanto.
- ^ a b "Resisting Roundup". The New York Times. May 16, 2010. Retrieved March 24, 2016.
- ^ a b Heap, Ian (April 11, 2014). Herbicide Resistant Weeds. Integrated Pest Management. doi:10.1007/978-94-007-7796-5_12. Retrieved March 24, 2016.
- ^ a b Lori (May 7, 2009). "U of G Researchers Find Suspected Glyphosate-Resistant Weed". Uoguelph.ca. Retrieved August 22, 2010.
- ^ Tarter S (April 6, 2009). "Attack of the Superweeds". Journal Star (Peoria). Retrieved August 22, 2010.
- ^ Heap, Ian Michael (1988). "Resistance to herbicides in annual ryegrass (Lolium Rigidum)". Adelaide: Department of Agronomy, University of Adelaide.
{{cite web}}:|access-date=requires|url=(help); Missing or empty|url=(help) - ^ King, Carolyn (June 2015). "History of herbicide resistance Herbicide resistance: Then, now, and the years to come". Retrieved March 24, 2016.
- ^ Hartzler, Bob (January 29, 2003), "Are RR Weeds in Your Future I", Iowa State University (ISU), Weed Science Online, retrieved March 24, 2016
- ^ Powles SB, Lorraine-Colwill DF, Dellow JJ, Preston C (1998). "Evolved Resistance to Glyphosate in Rigid Ryegrass (Lolium rigidum) in Australia". Weed Science. 46 (5): 604–7. JSTOR 4045968.
- ^ Glyphosate Resistance in Crops and Weeds: History, Development, and Management. Editor, Vijay K. Nandula. John Wiley & Sons, 2010 ISBN 9781118043547
- ^ "Glyphosate resistance is a reality that should scare some cotton growers into changing the way they do business". Southeastfarmpress.com. Retrieved August 22, 2010.
- ^ "Map of Glyphosate-Resistant Weeds Globally". The International Survey of Herbicide Resistant Weeds. 2010. Retrieved January 12, 2013.
- ^ Neuman W, Pollack A (May 4, 2010). "U.S. Farmers Cope With Roundup-Resistant Weeds". New York Times. New York. pp. B1. Retrieved May 4, 2010.
- ^ Benbrook, Charles M (2012). "Impacts of genetically engineered crops on pesticide use in the U.S. - the first sixteen years". Environmental Sciences Europe. 24: 24. doi:10.1186/2190-4715-24-24.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help)CS1 maint: unflagged free DOI (link) - ^ Heap I. "Resistance by Active Ingredient (select "glyphosate" from the pulldown menu)". The International Survey of Herbicide Resistant Weeds. Retrieved April 13, 2014.
- ^ "With BioDirect, Monsanto Hopes RNA Sprays Can Someday Deliver Drought Tolerance and Other Traits to Plants on Demand". MIT Technology Review. Retrieved August 31, 2015.
- ^ Culpepper AS, Grey TL, Vencill WK, Kichler JM, Webster TM, Brown SM, York AC, Davis JW, Hanna WW (2006). "Glyphosate-resistant Palmer amaranth (Amaranthus palmeri) confirmed in Georgia". Weed Science. 54 (4): 620–6. doi:10.1614/WS-06-001R.1. JSTOR 4539441.
- ^ a b Hampton N. "Cotton versus the monster weed". Retrieved July 19, 2009.
- ^ a b Smith JT (March 2009). "Resistance a growing problem" (PDF). The Farmer Stockman. Retrieved July 19, 2009.
- ^ Taylor O (July 16, 2009). "Peanuts: variable insects, variable weather, Roundup resistant Palmer in new state". PeanutFax. AgFax Media. Retrieved July 19, 2009.
- ^ Vargas L, Bianchi MA, Rizzardi MA, Agostinetto D, Dal Magro T (2007). "Buva (Conyza bonariensis) resistente ao glyphosate na região sul do Brasil". Planta Daninha (in Portuguese). 25 (3): 573–8. doi:10.1590/S0100-83582007000300017.
{{cite journal}}: Unknown parameter|trans_title=ignored (|trans-title=suggested) (help) - ^ Koger CH, Shaner DL, Henry WB, Nadler-Hassar T, Thomas WE, Wilcut JW (2005). "Assessment of two nondestructive assays for detecting glyphosate resistance in horseweed (Conyza canadensis)". Weed Science. 53 (4): 438–45. doi:10.1614/WS-05-010R. JSTOR 4047050.
- ^ Ge X, d'Avignon DA, Ackerman JJ, Sammons RD (April 2010). "Rapid vacuolar sequestration: the horseweed glyphosate resistance mechanism". Pest Management Science. 66 (4): 345–8. doi:10.1002/ps.1911. PMC 3080097. PMID 20063320.
- ^ Vila-Aiub MM, Vidal RA, Balbi MC, Gundel PE, Trucco F, Ghersa CM (April 2008). "Glyphosate-resistant weeds of South American cropping systems: an overview". Pest Management Science. 64 (4): 366–71. doi:10.1002/ps.1488. PMID 18161884.
- ^ Jhala, Amit (June 4, 2015). "Post-Emergence Herbicide Options for Glyphosate-Resistant Marestail in Corn and Soybean". CropWatch. Nebraska Extension. Retrieved August 17, 2015.
{{cite news}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Preston C, Wakelin AM, Dolman FC, Bostamam Y, Boutsalis P (2009). "A Decade of Glyphosate-Resistant Lolium around the World: Mechanisms, Genes, Fitness, and Agronomic Management". Weed Science. 57 (4): 435–41. doi:10.1614/WS-08-181.1.
- ^ Vila-Aiub MM, Balbi MC, Gundel PE, Ghersa CM, Powles SB (2007). "Evolution of Glyphosate-Resistant Johnsongrass (Sorghum halepense) in Glyphosate-Resistant Soybean". Weed Science. 55 (6): 566–71. doi:10.1614/WS-07-053.1. JSTOR 4539618.
- ^ Kniss, Andrew (February 10, 2014). "Are herbicides responsible for the decline in Monarch butterflies?". Control Freaks. Retrieved June 16, 2016.
The evidence seems clear that the number of milkweed plants through this region has indeed declined. The cause for the milkweed decline, though, is a little less certain.
- ^ Plumer, Brad (January 29, 2014). "Monarch butterflies keep disappearing. Here's why". The Washington Post. Retrieved June 16, 2016.
- ^ Pleasants, John M.; Oberhauser, Karen S. (2012). "Milkweed loss in agricultural fields because of herbicide use: effect on the monarch butterfly population" (PDF). Insect Conservation and Diversity. 6: 135–144. doi:10.1111/j.1752-4598.2012.00196.x.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ Hartzler, Robert G.; Buhler, Douglas D. (2000). "Occurrence of common milkweed (Asclepias syriaca) in cropland and adjacent areas". Crop Protection. 19: 363–366. doi:10.1016/s0261-2194(00)00024-7.
{{cite journal}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ "NRDC Sues EPA Over Demise of Monarch Butterfly Population". NBC. 2015.
- ^ Staff, Centralamericadata.com. September 6, 2013 El Salvador: Use of 53 Chemicals Banned
- ^ Staff, Centralamericadata.com. November 27, 2013 El Salvador: Confirmation to Be Given on Ban of Agrochemicals
- ^ Legislative Assembly of El Salvador. November 26, 2013 Analizan observaciones del Ejecutivo al decreto que contiene la prohibición de 53 agroquímicos que dañan la salud English translation by Google
- ^ Staff, Sustainable Pulse. Apr 4 2014 Dutch Parliament Bans Glyphosate Herbicides for Non-Commercial Use
- ^ Staff, Colombo Page. May 22, 2015 Sri Lankan President orders to ban import of glyphosate with immediate effect
- ^ Sarina Locke for the Australian Broadcasting Corporation. Updated May 27, 2015 Toxicologist critical of 'dodgy science' in glyphosate bans
- ^ "Health Minister: importation of roundup weed spray suspended". Bermuda Today. May 11, 2015.
- ^ "Colombia to ban coca spraying herbicide glyphosate". BBC News. May 10, 2015.
- ^ "UPDATE 2-French minister asks shops to stop selling Monsanto Roundup weedkiller". Reuters.
- ^ Arthur Nelson (March 8, 2016). "Vote on controversial weedkiller's European licence postponed". The Guardian.
- ^ "Recall of Monsanto's Roundup likely as EU refuses limited use of glyphosate". Reuters. June 6, 2016.
- ^ Arthur Nelson (June 29, 2016). "Controversial chemical in Roundup weedkiller escapes immediate ban". The Guardian.
- ^ Charry T (May 29, 1997). "Monsanto recruits the horticulturist of the San Diego Zoo to pitch its popular herbicide". Business Day. New York Times.
- ^ "Monsanto fined in France for 'false' herbicide ads". Terradaily.com (January 26, 2007).
- ^ Europe | Monsanto guilty in 'false ad' row. BBC News (October 15, 2009).
- ^ Piller D (April 1, 2010). "Albaugh accuses Chinese of dumping herbicide". Staff Blogs. Des Moines Register.
- ^ "In the Matter of: GLYPHOSATE FROM CHINA" (PDF). United States International Trade Commission. April 22, 2010.
External links
- Glyphosate in the Pesticide Properties DataBase (PPDB)
- Glyphosate trimesium in the Pesticide Properties DataBase (PPDB)
- Glyphosate, isopropylamine salt in the Pesticide Properties DataBase (PPDB)
- Glyphosate, potassium salt in the Pesticide Properties DataBase (PPDB)
