Multiple sclerosis: Difference between revisions
| Line 410: | Line 410: | ||
As of 2009, five disease-modifying treatments have been approved by regulatory agencies of different countries for MS. [[Interferon beta-1a]] (trade names ''Avonex'', ''[[CinnoVex]]'', ''[[ReciGen]]'' and ''Rebif'') and [[interferon beta-1b]] (U.S. trade name ''Betaseron'', in Europe and Japan ''Betaferon''). A third medication is [[glatiramer acetate]] (''Copaxone''), a non-interferon, non-steroidal immunomodulator. The fourth medication, [[mitoxantrone]], is an [[immunosuppressant]] also used in [[chemotherapy|cancer chemotherapy]]. The fifth is [[natalizumab]] (marketed as ''Tysabri'').<ref name="pmid18970977"/> The interferons and glatiramer acetate are delivered by frequent injections, varying from once-per-day for glatiramer acetate to once-per-week (but intra-muscular) for ''[[Avonex]]''. [[Natalizumab]] and mitoxantrone are given by IV infusion at monthly intervals. |
As of 2009, five disease-modifying treatments have been approved by regulatory agencies of different countries for MS. [[Interferon beta-1a]] (trade names ''Avonex'', ''[[CinnoVex]]'', ''[[ReciGen]]'' and ''Rebif'') and [[interferon beta-1b]] (U.S. trade name ''Betaseron'', in Europe and Japan ''Betaferon''). A third medication is [[glatiramer acetate]] (''Copaxone''), a non-interferon, non-steroidal immunomodulator. The fourth medication, [[mitoxantrone]], is an [[immunosuppressant]] also used in [[chemotherapy|cancer chemotherapy]]. The fifth is [[natalizumab]] (marketed as ''Tysabri'').<ref name="pmid18970977"/> The interferons and glatiramer acetate are delivered by frequent injections, varying from once-per-day for glatiramer acetate to once-per-week (but intra-muscular) for ''[[Avonex]]''. [[Natalizumab]] and mitoxantrone are given by IV infusion at monthly intervals. |
||
All five kinds of medications are modestly effective at decreasing the number of attacks in relapsing-remitting MS (RRMS) while the capacity of interferons and glatiramer acetate is more controversial. Studies of their long-term effects are still lacking.<ref name="pmid18970977"/><ref name="pmid19882365"/> Comparisons between immunomodulators (all but mitoxantrone) show that the most effective is natalizumab, both in terms of relapse rate reduction and halting disability progression.<ref name="pmid17350652">{{cite journal |author=Johnson KP |title=Control of multiple sclerosis relapses with immunomodulating agents |journal=J. Neurol. Sci. |volume=256 |issue=Suppl 1 |pages=S23–8 |year=2007 |pmid=17350652 |doi=10.1016/j.jns.2007.01.060}}</ref> Mitoxantrone may be the most effective of them all, however, it is generally not considered as a long-term therapy, as its use is limited by severe |
All five kinds of medications are modestly effective at decreasing the number of attacks in relapsing-remitting MS (RRMS) while the capacity of interferons and glatiramer acetate is more controversial. Studies of their long-term effects are still lacking.<ref name="pmid18970977"/><ref name="pmid19882365"/> Comparisons between immunomodulators (all but mitoxantrone) show that the most effective is natalizumab, both in terms of relapse rate reduction and halting disability progression.<ref name="pmid17350652">{{cite journal |author=Johnson KP |title=Control of multiple sclerosis relapses with immunomodulating agents |journal=J. Neurol. Sci. |volume=256 |issue=Suppl 1 |pages=S23–8 |year=2007 |pmid=17350652 |doi=10.1016/j.jns.2007.01.060}}</ref> Mitoxantrone may be the most effective of them all, however, it is generally not considered as a long-term therapy, as its use is limited by severe severe secondary effects.<ref name="pmid18970977"/><ref name="pmid19882365">{{cite journal |author=Comi G |title=Treatment of multiple sclerosis: role of natalizumab |journal=Neurol. Sci. |volume=30 Suppl 2 |issue= |pages=S155–8 |year=2009 |month=October |pmid=19882365 |doi=10.1007/s10072-009-0147-2 |url=}}</ref> The earliest clinical presentation of RRMS is the clinically isolated syndrome (CIS). Treatment with [[interferon]]s during an initial attack can decrease the chance that a patient will develop clinical MS.<ref name="pmid18970977"/> |
||
Treatment of progressive MS is more difficult than relapsing-remitting MS. Mitoxantrone has shown positive effects in patients with secondary progressive and progressive relapsing courses. It is moderately effective in reducing the progression of the disease and the frequency of relapses in patients in short-term follow-up.<ref name="pmid16235298">{{cite journal |author=Martinelli Boneschi F, Rovaris M, Capra R, Comi G |title=Mitoxantrone for multiple sclerosis |journal=Cochrane database of systematic reviews (Online) |volume= |issue=4 |pages=CD002127 |year=2005 |pmid=16235298 |doi=10.1002/14651858.CD002127.pub2}}</ref> No treatment has been proven to modify the course of primary progressive MS.<ref name="pmid15907149">{{cite journal |author=Leary SM, Thompson AJ |title=Primary progressive multiple sclerosis: current and future treatment options |journal=CNS drugs |volume=19 |issue=5 |pages=369–76 |year=2005 |pmid=15907149 |doi=10.2165/00023210-200519050-00001}}</ref> |
Treatment of progressive MS is more difficult than relapsing-remitting MS. Mitoxantrone has shown positive effects in patients with secondary progressive and progressive relapsing courses. It is moderately effective in reducing the progression of the disease and the frequency of relapses in patients in short-term follow-up.<ref name="pmid16235298">{{cite journal |author=Martinelli Boneschi F, Rovaris M, Capra R, Comi G |title=Mitoxantrone for multiple sclerosis |journal=Cochrane database of systematic reviews (Online) |volume= |issue=4 |pages=CD002127 |year=2005 |pmid=16235298 |doi=10.1002/14651858.CD002127.pub2}}</ref> No treatment has been proven to modify the course of primary progressive MS.<ref name="pmid15907149">{{cite journal |author=Leary SM, Thompson AJ |title=Primary progressive multiple sclerosis: current and future treatment options |journal=CNS drugs |volume=19 |issue=5 |pages=369–76 |year=2005 |pmid=15907149 |doi=10.2165/00023210-200519050-00001}}</ref> |
||
As with any medical treatment, these treatments have several adverse effects. One of the most common is irritation at the injection site for glatiramer acetate and the interferon treatments. Over time, a visible dent at the injection site, due to the local destruction of fat tissue, known as [[lipoatrophy]], may develop. Interferons produce symptoms similar to influenza;<ref name="pmid17131933">{{cite journal |author=Sládková T, Kostolanský F |title=The role of cytokines in the immune response to influenza A virus infection |journal=Acta Virol. |volume=50 |issue=3 |pages=151–62 |year=2006 |pmid=17131933 }}</ref> some patients taking glatiramer experience a post-injection reaction manifested by flushing, chest tightness, heart palpitations, breathlessness, and anxiety, which usually lasts less than thirty minutes.<ref name="pmid14974077">{{cite journal |author=Munari L, Lovati R, Boiko A |title=Therapy with glatiramer acetate for multiple sclerosis |journal=Cochrane database of systematic reviews (Online) |volume= |issue=1 |pages=CD004678 |year=2004 |pmid=14974077 |doi=10.1002/14651858.CD004678}}</ref> More dangerous are [[hepatotoxicity|liver damage]] from interferons |
As with any medical treatment, these treatments have several adverse effects. One of the most common is irritation at the injection site for glatiramer acetate and the interferon treatments. Over time, a visible dent at the injection site, due to the local destruction of fat tissue, known as [[lipoatrophy]], may develop. Interferons produce symptoms similar to influenza;<ref name="pmid17131933">{{cite journal |author=Sládková T, Kostolanský F |title=The role of cytokines in the immune response to influenza A virus infection |journal=Acta Virol. |volume=50 |issue=3 |pages=151–62 |year=2006 |pmid=17131933 }}</ref> some patients taking glatiramer experience a post-injection reaction manifested by flushing, chest tightness, heart palpitations, breathlessness, and anxiety, which usually lasts less than thirty minutes.<ref name="pmid14974077">{{cite journal |author=Munari L, Lovati R, Boiko A |title=Therapy with glatiramer acetate for multiple sclerosis |journal=Cochrane database of systematic reviews (Online) |volume= |issue=1 |pages=CD004678 |year=2004 |pmid=14974077 |doi=10.1002/14651858.CD004678}}</ref> More dangerous but much less common are [[hepatotoxicity|liver damage]] from interferons,<ref name="pmid15592724">{{cite journal |author=Tremlett H, Oger J |title=Hepatic injury, liver monitoring and the beta-interferons for multiple sclerosis |journal=J. Neurol. |volume=251 |issue=11 |pages=1297–303 |year=2004 |month=November |pmid=15592724 |doi=10.1007/s00415-004-0619-5 |url=}}</ref> severe [[cardiotoxicity]], [[infertility]], and [[acute myeloid leukaemia]] of mitoxantrone,<ref name="pmid18970977"/><ref name="pmid19882365"/> and the putative link between natalizumab and some cases of [[progressive multifocal leukoencephalopathy]].<ref name="pmid18970977"/> |
||
=== Management of the effects of MS === |
=== Management of the effects of MS === |
||
Revision as of 11:54, 27 January 2010
| Multiple sclerosis | |
|---|---|
| Specialty | Neurology |
| Frequency | 0.032% (world) |
Multiple sclerosis (abbreviated MS, also known as disseminated sclerosis or encephalomyelitis disseminata) is a disease in which the fatty myelin sheaths around the axons of the brain and spinal cord are damaged, leading to demyelination and scarring as well as a broad spectrum of signs and symptoms.[1] Disease onset usually occurs in young adults, and it is more common in females.[1] It has a prevalence that ranges between 2 and 150 per 100,000.[2] MS was first described in 1868 by Jean-Martin Charcot.[3]
MS affects the ability of nerve cells in the brain and spinal cord to communicate with each other. Nerve cells communicate by sending electrical signals called action potentials down long fibers called axons, which are wrapped in an insulating substance called myelin. In MS, the body's own immune system attacks and damages the myelin. When myelin is lost, the axons can no longer effectively conduct signals.[4] The name multiple sclerosis refers to scars (scleroses—better known as plaques or lesions) in the white matter of the brain and spinal cord, which is mainly composed of myelin.[3] Although much is known about the mechanisms involved in the disease process, the cause remains unknown. Theories include genetics or infections. Different environmental risk factors have also been found.[4][5]
Almost any neurological symptom can appear with the disease, and often progresses to physical and cognitive disability[4] and neuropsychiatric disorder.[6] MS takes several forms, with new symptoms occurring either in discrete attacks (relapsing forms) or slowly accumulating over time (progressive forms).[7] Between attacks, symptoms may go away completely, but permanent neurological problems often occur, especially as the disease advances.[7]
There is no known cure for MS. Treatments attempt to return function after an attack, prevent new attacks, and prevent disability.[4] MS medications can have adverse effects or be poorly tolerated, and many patients pursue alternative treatments, despite the lack of supporting scientific study. The prognosis is difficult to predict; it depends on the subtype of the disease, the individual patient's disease characteristics, the initial symptoms and the degree of disability the person experiences as time advances.[8] Life expectancy of patients is nearly the same as that of the unaffected population.[8]
Classification

Several subtypes, or patterns of progression, have been described. Subtypes use the past course of the disease in an attempt to predict the future course. They are important not only for prognosis but also for therapeutic decisions. In 1996 the United States National Multiple Sclerosis Society standardized four subtype definitions:[7]
- relapsing remitting,
- secondary progressive,
- primary progressive, and
- progressive relapsing.
The relapsing-remitting subtype is characterized by unpredictable relapses followed by periods of months to years of relative quiet (remission) with no new signs of disease activity. Deficits suffered during attacks may either resolve or leave sequelae, the latter being more common as a function of time.[1] This describes the initial course of 85–90% of individuals with MS.[7] When deficits always resolve between attacks, this is sometimes referred to as benign MS.[9] The relapsing-remitting subtype usually begins with a clinically isolated syndrome (CIS). In CIS, a patient has an attack suggestive of demyelination, but does not fulfill the criteria for multiple sclerosis.[1][10] However only 30 to 70% of persons experiencing CIS later develop MS.[10]
Secondary progressive MS (sometimes called "galloping MS") describes around 65 % of those with an initial relapsing-remitting MS, who then begin to have progressive neurologic decline between acute attacks without any definite periods of remission.[1][7] Occasional relapses and minor remissions may appear.[7] The median time between disease onset and conversion from relapsing-remitting to secondary progressive MS is 19 years.[11]
The primary progressive subtype describes the approximately 10–15% of individuals who never have remission after their initial MS symptoms.[12] It is characterized by progression of disability from onset, with no, or only occasional and minor, remissions and improvements.[7] The age of onset for the primary progressive subtype is later than for the relapsing-remitting, but similar to mean age of progression between the relapsing-remitting and the secondary progressive. In both cases it is around 40 years of age.[1]
Progressive relapsing MS describes those individuals who, from onset, have a steady neurologic decline but also suffer clear superimposed attacks. This is the least common of all subtypes.[7]
Cases with non-standard behavior have also been described. Sometimes referred to as borderline forms of multiple sclerosis, these include Devic's disease, Balo concentric sclerosis, Schilder's diffuse sclerosis and Marburg multiple sclerosis.[13] There is debate whether these are atypical variants of MS or different diseases.[14] Multiple sclerosis also behaves differently in children, taking them more time to reach the progressive stage.[1] Nevertheless they still reach it at a lower mean age than adults.[1]
Signs and symptoms
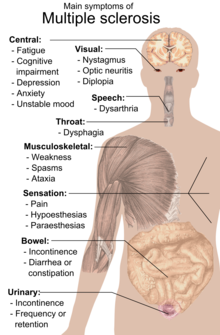
The person with MS can suffer almost any neurological symptom or sign, including changes in sensation (hypoesthesia and paraesthesia), muscle weakness, muscle spasms, or difficulty in moving; difficulties with coordination and balance (ataxia); problems in speech (dysarthria) or swallowing (dysphagia), visual problems (nystagmus, optic neuritis, or diplopia), fatigue, acute or chronic pain, and bladder and bowel difficulties.[1] Cognitive impairment of varying degrees and emotional symptoms of depression or unstable mood are also common.[1] Uhthoff's phenomenon, an exacerbation of extant symptoms due to an exposure to higher than usual ambient temperatures, and Lhermitte's sign, an electrical sensation that runs down the back when bending the neck, are particularly characteristic of MS although not specific.[1] The main clinical measure of disability progression and symptom severity is the Expanded Disability Status Scale or EDSS.[15]
Symptoms of MS usually appear in episodic acute periods of worsening (called relapses, exacerbations, bouts, attacks, or "flare-ups"), in a gradually progressive deterioration of neurologic function, or in a combination of both.[7] Multiple sclerosis relapses are often unpredictable, occurring without warning and without obvious inciting factors with a rate rarely above 1 and a half per year.[1] Some attacks, however, are preceded by common triggers. Relapses occur more frequently during spring and summer.[16] Viral infections such as the common cold, influenza, or gastroenteritis increase the risk of relapse.[1] Stress may also trigger an attack.[17] Pregnancy affects the susceptibility to relapse, with a lower relapse rate at each trimester of gestation. During the first few months after delivery, however, the risk of relapse is increased.[1] Overall, pregnancy does not seem to influence long-term disability. Many potential triggers have been examined and found not to influence MS relapse rates. There is no evidence that vaccination and breast feeding,[1] physical trauma,[18] or Uhthoff's phenomenon[16] are relapse triggers.
Causes
Most likely MS occurs as a result of some combination of genetic, environmental and infectious factors.[1] Epidemiological studies of MS have provided hints on possible causes for the disease. Theories try to combine the known data into plausible explanations, but none has proved definitive.
Genetics

MS is not considered a hereditary disease. However, a number of genetic variations have been shown to increase the risk of developing the disease.[19]
The risk of acquiring MS is higher in relatives of a person with the disease than in the general population, especially in the case of siblings, parents, and children.[4] The disease has an overall familial recurrence rate of 20%.[1] In the case of monozygotic twins, concordance occurs only in about 35% of cases, while it goes down to around 5% in the case of siblings and even lower in half-siblings. This indicates susceptibility is partly polygenically driven.[1][4]
It seems to be more common in some ethnic groups than others.
Apart from familial studies, specific genes have been linked with MS. Differences in the human leukocyte antigen (HLA) system—a group of genes in chromosome 6 that serves as the major histocompatibility complex (MHC) in humans—increase the probability of suffering MS.[1] The most consistent finding is the association between multiple sclerosis and alleles of the MHC defined as DR15 and DQ6.[1] Other loci have shown a protective effect, such as HLA-C554 and HLA-DRB1*11.[1]
Environmental factors
Different environmental factors, both of infectious and non infectious origin have been proposed to as risk factors for MS. Although some are partly modifiable, only further research—especially clinical trials—will reveal whether their elimination can help prevent MS.[20]
MS is more common in people who live farther from the equator, although many exceptions exist.[1] Decreased sunlight exposure has been linked with a higher risk of MS.[20] Decreased vitamin D production and intake has been the main biological mechanism used to explain the higher risk among those less exposed to sun.[20][21]
Severe stress may also be a risk factor although evidence is weak.[20] Smoking has also been shown to be an independent risk factor for developing MS.[21] Association with occupational exposures and toxins—mainly solvents—has been evaluated, but no clear conclusions have been reached.[20] Vaccinations were also considered as causal factors for the disease; however, most studies show no association between MS and vaccines.[20] Several other possible risk factors, such as diet and hormone intake, have been investigated; however, more evidence is needed to confirm or refute their relation with the disease.[21]
Gout occurs less than would statistically be expected in people with MS, and low levels of uric acid have been found in MS patients as compared to normal individuals. This led to the theory that uric acid protects against MS, although its exact importance remains unknown.[22]
Infections
Genetic susceptibility can explain some of the geographic and epidemiological variations in MS incidence, like the high appearance of the disease among some families or the risk decline with genetic distance, but does not account for other phenomena, such as the changes in risk that occur with migration at an early age.[5]
An explanation for this epidemiology finding could be that some kind of infection, produced by a widespread microbe rather than a rare pathogen, is the origin of the disease.[5] Different hypotheses have elaborated on the mechanism by which this may occur. The hygiene hypothesis proposes that exposure to several infectious agents early in life is protective against MS, being the disease a response to a later encounter with such agents.[1] The prevalence hypothesis proposes that the disease is due to a pathogen more common in regions of high MS prevalence. This pathogen is very common, causing in most individuals an asymptomatic persistent infection. Only in a few cases, and after many years since the original infection, does it bring demyelination.[5][23] The hygiene hypothesis has received more support than the prevalence hypothesis.[5]
Evidence for viruses as a cause includes the presence of oligoclonal bands in the brain and cerebrospinal fluid of most patients, the association of several viruses with human demyelinating encephalomyelitis, and induction of demyelination in animals through viral infection.[24] Human herpes viruses are a candidate group of viruses linked to MS. Individuals who have never been infected by the Epstein-Barr virus have a reduced risk of having the disease and among the infected ones those infected as young adults have a greater risk than those who had it at a younger age.[5][1] Although some consider that this goes against the hygiene hypothesis, since the non-infected have probably experienced a more hygienic upbringing,[5] others believe that there is no contradiction since it is the encounter at a later moment with the virus the trigger for the disease.[1] Other diseases that have also been related with MS are measles, mumps and rubella.[1]
Pathophysiology
Blood-brain barrier breakdown

The blood–brain barrier is a capillary system that should prevent entrance of T cells into the nervous system.[4] The blood–brain barrier is normally not permeable to these types of cells, unless triggered by infection or a virus, which decreases the integrity of the tight junctions forming the barrier.[4] When the blood–brain barrier regains its integrity, usually after infection or virus has cleared, the T cells are trapped inside the brain.[4]
Autoimmunology
MS is currently believed to be an immune-mediated disorder with an initial trigger, which may have a viral etiology,[4] although this concept has been debated for years and some still oppose it. Damage is believed to be caused by the patient's own immune system. The immune system attacks the nervous system, possibly as a result of exposure to a molecule with a similar structure to one of its own.[4]
- Lesions
The name multiple sclerosis refers to the scars (scleroses – better known as plaques or lesions) that form in the nervous system. MS lesions most commonly involve white matter areas close to the ventricles of the cerebellum, brain stem, basal ganglia and spinal cord; and the optic nerve. The function of white matter cells is to carry signals between grey matter areas, where the processing is done, and the rest of the body. The peripheral nervous system is rarely involved.[4]
More specifically, MS destroys oligodendrocytes, the cells responsible for creating and maintaining a fatty layer—known as the myelin sheath—which helps the neurons carry electrical signals.[4] MS results in a thinning or complete loss of myelin and, as the disease advances, the cutting (transection) of the neuron's extensions or axons.[25] When the myelin is lost, a neuron can no longer effectively conduct electrical signals.[4] A repair process, called remyelination, takes place in early phases of the disease, but the oligodendrocytes cannot completely rebuild the cell's myelin sheath.[26] Repeated attacks lead to successively fewer effective remyelinations, until a scar-like plaque is built up around the damaged axons.[26] Four different lesion patterns have been described.[27]
- Inflammation
Apart from demyelination, the other pathologic hallmark of the disease is inflammation. According to a strictly immunological explanation of MS, the inflammatory process is caused by T cells, a kind of lymphocyte. Lymphocytes are cells that play an important role in the body's defenses.[4] In MS, T cells gain entry into the brain via the previously described blood–brain barrier. Recent evidence from animal models also point to a role of B cells in addition to T cells in development of the disease.[28][29]
The T cells recognize myelin as foreign and attack it as if it were an invading virus. This triggers inflammatory processes, stimulating other immune cells and soluble factors like cytokines and antibodies. Leaks form in the blood–brain barrier, which in turn cause a number of other damaging effects such as swelling, activation of macrophages, and more activation of cytokines and other destructive proteins.[4]
Diagnosis
Multiple sclerosis can be difficult to diagnose since its signs and symptoms may be similar to other medical problems.[1][30] Medical organizations have created diagnostic criteria to ease and standardize the diagnostic process for practicing physicians, specially in the first stages of the disease.[1] Historically, the Schumacher and Poser criteria were both popular.[31] Currently, the McDonald criteria focus on a demonstration with clinical, laboratory and radiologic data of the dissemination of MS lesions in time and space.[1][32][33]
Clinical data alone may be sufficient for a diagnosis of MS if an individual has suffered separate episodes of neurologic symptoms characteristic of MS.[32] Since some people seek medical attention after only one attack, other testing may hasten and ease the diagnosis. The most commonly used diagnostic tools are neuroimaging, analysis of cerebrospinal fluid and evoked potentials. Magnetic resonance imaging of the brain and spine shows areas of demyelination (lesions or plaques). Gadolinium can be administered intravenously as a contrast to highlight active plaques and, by elimination, demonstrate the existence of historical lesions not associated with symptoms at the moment of the evaluation.[32][34]
Testing of cerebrospinal fluid obtained from a lumbar puncture can provide evidence of chronic inflammation of the central nervous system. The cerebrospinal fluid is tested for oligoclonal bands, which are an inflammation marker found in 75–85% of people with MS.[32].[35] Since no known test is perfectly specific to MS, only biopsies or post-mortem examinations can yield an absolutely certain diagnosis.
The nervous system of a person with MS often responds less actively to stimulation of the optic nerve and sensory nerves due to demyelination of such pathways. These brain responses can be examined using visual and sensory evoked potentials.[36]
Management
Although there is no known cure for multiple sclerosis, several therapies have proven helpful. The primary aims of therapy are returning function after an attack, preventing new attacks, and preventing disability. As with any medical treatment, medications used in the management of MS have several adverse effects. Alternative treatments are pursued by some patients, despite the shortage of supporting, comparable, replicated scientific study.
Acute attacks
During symptomatic attacks, administration of high doses of intravenous corticosteroids, such as methylprednisolone, is the routine therapy for acute relapses.[1] Although generally effective in the short term for relieving symptoms, corticosteroid treatments do not appear to have a significant impact on long-term recovery.[37] Oral and intravenous administration seem to have similar efficacy.[38] Consequences of severe attacks which do not respond to corticosteroids might be treated by plasmapheresis.[1]
Disease-modifying treatments
As of 2009, five disease-modifying treatments have been approved by regulatory agencies of different countries for MS. Interferon beta-1a (trade names Avonex, CinnoVex, ReciGen and Rebif) and interferon beta-1b (U.S. trade name Betaseron, in Europe and Japan Betaferon). A third medication is glatiramer acetate (Copaxone), a non-interferon, non-steroidal immunomodulator. The fourth medication, mitoxantrone, is an immunosuppressant also used in cancer chemotherapy. The fifth is natalizumab (marketed as Tysabri).[1] The interferons and glatiramer acetate are delivered by frequent injections, varying from once-per-day for glatiramer acetate to once-per-week (but intra-muscular) for Avonex. Natalizumab and mitoxantrone are given by IV infusion at monthly intervals.
All five kinds of medications are modestly effective at decreasing the number of attacks in relapsing-remitting MS (RRMS) while the capacity of interferons and glatiramer acetate is more controversial. Studies of their long-term effects are still lacking.[1][39] Comparisons between immunomodulators (all but mitoxantrone) show that the most effective is natalizumab, both in terms of relapse rate reduction and halting disability progression.[40] Mitoxantrone may be the most effective of them all, however, it is generally not considered as a long-term therapy, as its use is limited by severe severe secondary effects.[1][39] The earliest clinical presentation of RRMS is the clinically isolated syndrome (CIS). Treatment with interferons during an initial attack can decrease the chance that a patient will develop clinical MS.[1]
Treatment of progressive MS is more difficult than relapsing-remitting MS. Mitoxantrone has shown positive effects in patients with secondary progressive and progressive relapsing courses. It is moderately effective in reducing the progression of the disease and the frequency of relapses in patients in short-term follow-up.[41] No treatment has been proven to modify the course of primary progressive MS.[42]
As with any medical treatment, these treatments have several adverse effects. One of the most common is irritation at the injection site for glatiramer acetate and the interferon treatments. Over time, a visible dent at the injection site, due to the local destruction of fat tissue, known as lipoatrophy, may develop. Interferons produce symptoms similar to influenza;[43] some patients taking glatiramer experience a post-injection reaction manifested by flushing, chest tightness, heart palpitations, breathlessness, and anxiety, which usually lasts less than thirty minutes.[44] More dangerous but much less common are liver damage from interferons,[45] severe cardiotoxicity, infertility, and acute myeloid leukaemia of mitoxantrone,[1][39] and the putative link between natalizumab and some cases of progressive multifocal leukoencephalopathy.[1]
Management of the effects of MS
Disease-modifying treatments reduce the progression rate of the disease, but do not stop it. As multiple sclerosis progresses, the symptomatology tends to increase. The disease is associated with a variety of symptoms and functional deficits that result in a range of progressive impairments and disability. Management of these deficits is therefore very important. Both drug therapy and neurorehabilitation have shown to ease the burden of some symptoms, though neither influences disease progression.[46][1] Some symptoms have a good response to medication, such as unstable bladder and spasticity, while management of many others is much more complicated.[1] As for any patient with neurologic deficits, a multidisciplinary approach is key to improving quality of life; however, there are particular difficulties in specifying a ‘core team’ because people with MS may need help from almost any health profession or service at some point.[1] Multidisciplinary rehabilitation programmes increase activity and participation of patients but do not influence impairment level.[47] On January 22, 2010, the U.S. Food and Drug Administration (FDA) approved the oral drug dalfampridine (Ampyra) to improve walking ability for people with multiple sclerosis.[48] Ampyra will be available in March 2010.[49]
Alternative treatments
As with most chronic diseases, alternative treatments are pursued by some patients, despite the shortage of supporting, comparable, replicated scientific study. Examples are dietary regimens,[50] herbal medicine, including the use of medical cannabis to help alleviate symptoms,[51][52] and hyperbaric oxygenation.[53] The therapeutic practice of martial arts such as tai chi, relaxation disciplines such as yoga, or general exercise seems to mitigate fatigue, but has no effect on cognitive function.[54]
Prognosis

The prognosis (the expected future course of the disease) for a person with multiple sclerosis depends on the subtype of the disease; the individual's sex, age, and initial symptoms; and the degree of disability the person experiences.[8] The disease evolves and advances over decades, 30 being the mean years to death since onset.[1]
Female sex, relapsing-remitting subtype, optic neuritis or sensory symptoms at onset, few attacks in the initial years and especially early age at onset, are associated with a better course.[8][55]
The life expectancy of people with MS is 5 to 10 years lower to that of unaffected people.[1] Almost 40% of patients reach the seventh decade of life.[55] Nevertheless, two thirds of the deaths in people with MS are directly related to the consequences of the disease.[1] Suicide is also a much more important risk of death than in the healthy population, while infections and complications are specially hazardous for the more disabled ones.[1]
Although most patients lose the ability to walk prior to death, 90% are still capable of independent walking at 10 years from onset, and 75% at 15 years.[55][56]
Epidemiology
Two main measures are used in epidemiological studies: incidence and prevalence. Incidence is the number of new cases per unit of person–time at risk (usually number of new cases per thousand person–years); while prevalence is the total number of cases of the disease in the population at a given time. Prevalence is known to depend not only on incidence, but also on survival rate and migrations of affected people. MS has a prevalence that ranges between 2 and 150 per 100,000 depending on the country or specific population.[2] Studies on populational and geographical patterns of epidemiological measures have been very common in MS,[23] and have led to the proposal of different etiological (causal) theories.[5][20][21][23]

MS usually appears in adults in their thirties but it can also appear in children.[1] The primary progressive subtype is more common in people in their fifties.[12] As with many autoimmune disorders, the disease is more common in women, and the trend may be increasing.[1][57] In children, the sex ratio difference is higher,[1] while in people over fifty, MS affects males and females almost equally.[12]
There is a north-to-south gradient in the northern hemisphere and a south-to-north gradient in the southern hemisphere, with MS being much less common in people living near the equator.[1][57] Climate, sunlight and intake of vitamin D have been investigated as possible causes of the disease that could explain this latitude gradient.[21] However, there are important exceptions to the north-south pattern and changes in prevalence rates over time;[1] in general, this trend might be disappearing.[57] This indicates that other factors such as environment or genetics have to be taken into account to explain the origin of MS.[1] MS is also more common in regions with northern Europe populations.[1] But even in regions where MS is common, some ethnic groups are at low risk of developing the disease, including the Samis, Turkmen, Amerindians, Canadian Hutterites, Africans, and New Zealand Māori.[20]
Environmental factors during childhood may play an important role in the development of MS later in life. Several studies of migrants show that if migration occurs before the age of 15, the migrant acquires the new region's susceptibility to MS. If migration takes place after age 15, the migrant retains the susceptibility of his home country.[1][20] However, the age–geographical risk for developing multiple sclerosis may span a larger timescale.[1] A relationship between season of birth and MS has also been found which lends support to an association with sunlight and vitamin D. For example fewer people with MS are born in November as compared to May.[58]
History
Medical discovery

The French neurologist Jean-Martin Charcot (1825–1893) was the first person to recognize multiple sclerosis as a distinct disease in 1868.[59] Summarizing previous reports and adding his own clinical and pathological observations, Charcot called the disease sclerose en plaques. The three signs of MS now known as Charcot's triad 1 are nystagmus, intention tremor, and telegraphic speech, though these are not unique to MS. Charcot also observed cognition changes, describing his patients as having a "marked enfeeblement of the memory" and "conceptions that formed slowly".[3]
Prior to Charcot, Robert Carswell (1793–1857), a British professor of pathology, and Jean Cruveilhier (1791–1873), a French professor of pathologic anatomy, had described and illustrated many of the disease's clinical details, but did not identify it as a separate disease.[60] Specifically, Carswell described the injuries he found as "a remarkable lesion of the spinal cord accompanied with atrophy".[1]
After Charcot's description, Eugène Devic (1858–1930), Jozsef Balo (1895–1979), Paul Ferdinand Schilder (1886–1940), and Otto Marburg (1874–1948) described special cases of the disease. The disease did not receive its current name until 1955.[1] During all the 20th century there was an important development on the theories about the cause and pathogenesis of MS while efficacious treatments began to appear in 1990.[1]
Historical cases
There are several historical accounts of people who lived before or shortly after the disease was described by Charcot and probably had MS.
A young woman called Halldora, who lived in Iceland around the year 1200, suddenly lost her vision and mobility, but after praying to the saints, recovered them seven days after. Saint Lidwina of Schiedam (1380–1433), a Dutch nun, may be one of the first clearly identifiable MS patients. From the age of 16 until her death at 53, she suffered intermittent pain, weakness of the legs, and vision loss—symptoms typical of MS.[61] Both cases have led to the proposal of a 'Viking gene' hypothesis for the dissemination of the disease.[62]
Augustus Frederick d'Este (1794–1848), son of Prince Augustus Frederick, Duke of Sussex and Lady Augusta Murray and the grandson of George III of the United Kingdom, almost certainly suffered from MS. D'Este left a detailed diary describing his 22 years living with the disease. His diary began in 1822 and ended in 1846, although it remained unknown until 1948. His symptoms began at age 28 with a sudden transient visual loss after the funeral of a friend. During the course of his disease, he developed weakness of the legs, clumsiness of the hands, numbness, dizziness, bladder disturbances, and erectile dysfunction. In 1844, he began to use a wheelchair. Despite his illness, he kept an optimistic view of life.[63][64]
Another early account of MS was kept by the British diarist W. N. P. Barbellion, nom-de-plume of Bruce Frederick Cummings (1889–1919), who maintained a detailed log of his diagnosis and struggle with MS.[64] His diary was published in 1919 as The Journal of a Disappointed Man.[65]
Research directions
Therapies

A number of treatments that may curtail attacks or improve function are under investigation. Some of these treatments involve the combination of drugs that are already in use for multiple sclerosis, such as the joint administration of mitoxantrone and glatiramer acetate (Copaxone).[66] However, most treatments already in clinical trials involve drugs that are used in other diseases. These are alemtuzumab (trade name Campath),[67] daclizumab (trade name Zenapax),[68] inosine,[69] BG00012,[70] fingolimod,[71] and teriflunomide, the active metabolite of the DMARD leflunomide. Alemtuzumab performed better than interferon beta-1a in relapsing-remitting MS reducing disability, imaging abnormalities and frequence of relapses, at the cost of increased autoimmunity problems. These included three cases of thrombocytopenic purpura which led to the suspension of the therapy.[72]
Other drugs in clinical trials have been designed specifically for MS, such as laquinimod,[73] and Neurovax.[74]
Low dose naltrexone has been prescribed off-label for certain autoimmune disorders, including MS, and there is anecdotal evidence of benefit,[75][76] but only two small clinical trials have been conducted (as on December 2008),[77] one of them being for primary progressive MS.[78]
Diagnostic methods
New diagnostic and evolution evaluation methods are also being investigated. The measurement of antibodies against myelin proteins such as myelin oligodendrocyte glycoprotein and myelin basic protein could be useful for diagnosis. Optical coherence tomography of the eye's retina could be used as a measure of response to medication, axonal degeneration and brain atrophy.[79][80] A test over serum autoantibodies has been proposed to make a safer and more confident diagnosis.[81]
Evolution prediction
Currently there are no clinically established laboratory investigations available that can predict prognosis. However, several promising approaches have been proposed, such as the measurement of a lipid-specific immunoglobulin M as predictor of long-term outcomes.[82]
Chronic cerebrospinal venous insufficiency
Investigator Paolo Zamboni has conducted research suggesting that MS is not an autoimmune condition, but a vascular disease, chronic cerebrospinal venous insufficiency, in which the brain's veins are constricted. He theorises that this leads to a buildup of iron in the brain, which causes MS symptoms. He has performed a surgical procedure to correct what he believes to be venous insufficiency and claims that some patients have shown significant improvement in 73% of cases.[83] The surgeries were non controlled and non randomized.[84] The neurology community has been skeptical. Lily Jung, speaking on behalf of the American Academy of Neurology, felt that some of the claims in the report are not supported by the data. Jung also said. "We would welcome some randomized, controlled, double-blinded studies to look at the issue, but before then would not be encouraging our patients to jump in with both feet to do this procedure, which has significant risks and has not been proven to be safe."[84][85]
See also
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au av aw ax ay az ba bb bc bd Compston A, Coles A (2008). "Multiple sclerosis". Lancet. 372 (9648): 1502–17. doi:10.1016/S0140-6736(08)61620-7. PMID 18970977.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b Rosati G (2001). "The prevalence of multiple sclerosis in the world: an update". Neurol. Sci. 22 (2): 117–39. doi:10.1007/s100720170011. PMID 11603614.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c Charcot, J. (1868). "Histologie de la sclerose en plaques". Gazette des hopitaux, Paris. 41: 554–5.
- ^ a b c d e f g h i j k l m n o p Compston A, Coles A (2002). "Multiple sclerosis". Lancet. 359 (9313): 1221–31. doi:10.1016/S0140-6736(02)08220-X. PMID 11955556.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d e f g h Ascherio A, Munger KL (2007). "Environmental risk factors for multiple sclerosis. Part I: the role of infection". Ann. Neurol. 61 (4): 288–99. doi:10.1002/ana.21117. PMID 17444504.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Berrios G E & Quemada J I (1990) Andre G. Ombredane and the psychopathology of multiple sclerosis: a conceptual and statistical history. Comprehensive Psychiatry 31: 438–446
- ^ a b c d e f g h i Lublin FD, Reingold SC (1996). "Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis". Neurology. 46 (4): 907–11. PMID 8780061.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d Weinshenker BG (1994). "Natural history of multiple sclerosis". Ann. Neurol. 36 (Suppl): S6–11. doi:10.1002/ana.410360704. PMID 8017890.
- ^ Pittock SJ, Rodriguez M (2008). "Benign multiple sclerosis: a distinct clinical entity with therapeutic implications". Curr. Top. Microbiol. Immunol. 318: 1–17. doi:10.1007/978-3-540-73677-6_1. PMID 18219812.
- ^ a b Miller D, Barkhof F, Montalban X, Thompson A, Filippi M (2005). "Clinically isolated syndromes suggestive of multiple sclerosis, part I: natural history, pathogenesis, diagnosis, and prognosis". Lancet Neurol. 4 (5): 281–8. doi:10.1016/S1474-4422(05)70071-5. PMID 15847841.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Rovaris M, Confavreux C, Furlan R, Kappos L, Comi G, Filippi M (2006). "Secondary progressive multiple sclerosis: current knowledge and future challenges". Lancet Neurol. 5 (4): 343–54. doi:10.1016/S1474-4422(06)70410-0. PMID 16545751.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c Miller DH, Leary SM (2007). "Primary-progressive multiple sclerosis". Lancet Neurol. 6 (10): 903–12. doi:10.1016/S1474-4422(07)70243-0. PMID 17884680.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ De Seze J (2006). "[Borderlines types of multiple sclerosis]". Rev. Neurol. (Paris) (in French). 162 (1): 137–43. PMID 16446632.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ O'Riordan JI (1997). "Central nervous system white matter diseases other than multiple sclerosis". Curr. Opin. Neurol. 10 (3): 211–4. PMID 9229127.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Kurtzke JF (1983). "Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS)". Neurology. 33 (11): 1444–52. PMID 6685237.
- ^ a b Tataru N, Vidal C, Decavel P, Berger E, Rumbach L (2006). "Limited impact of the summer heat wave in France (2003) on hospital admissions and relapses for multiple sclerosis". Neuroepidemiology. 27 (1): 28–32. doi:10.1159/000094233. PMID 16804331.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Heesen C, Mohr DC, Huitinga I; et al. (2007). "Stress regulation in multiple sclerosis: current issues and concepts". Mult. Scler. 13 (2): 143–8. doi:10.1177/1352458506070772. PMID 17439878.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Martinelli V (2000). "Trauma, stress and multiple sclerosis". Neurol. Sci. 21 (4 Suppl 2): S849–52. doi:10.1007/s100720070024. PMID 11205361.
- ^ Dyment DA, Ebers GC, Sadovnick AD (2004). "Genetics of multiple sclerosis". Lancet Neurol. 3 (92): 104–10. doi:10.1016/S1474-4422(03)00663-X. PMID 14747002.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c d e f g h i Marrie RA (2004). "Environmental risk factors in multiple sclerosis aetiology". Lancet Neurol. 3 (12): 709–18. doi:10.1016/S1474-4422(04)00933-0. PMID 15556803.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d e Ascherio A, Munger KL (2007). "Environmental risk factors for multiple sclerosis. Part II: Noninfectious factors". Ann. Neurol. 61 (6): 504–13. doi:10.1002/ana.21141. PMID 17492755.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Spitsin S, Koprowski H (2008). "Role of uric acid in multiple sclerosis". Curr. Top. Microbiol. Immunol. 318: 325–42. doi:10.1007/978-3-540-73677-6_13. PMID 18219824.
- ^ a b c Kurtzke JF (1993). "Epidemiologic evidence for multiple sclerosis as an infection". Clin. Microbiol. Rev. 6 (4): 382–427. PMC 358295. PMID 8269393.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Gilden DH (2005). "Infectious causes of multiple sclerosis". The Lancet Neurology. 4 (3): 195–202. doi:10.1016/S1474-4422(05)01017-3. PMID 15721830.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Pascual AM, Martínez-Bisbal MC, Boscá I; et al. (2007). "Axonal loss is progressive and partly dissociated from lesion load in early multiple sclerosis". Neurology. 69 (1): 63–7. doi:10.1212/01.wnl.0000265054.08610.12. PMID 17606882.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Chari DM (2007). "Remyelination in multiple sclerosis". Int. Rev. Neurobiol. 79: 589–620. doi:10.1016/S0074-7742(07)79026-8. PMID 17531860.
- ^ Lucchinetti C, Brück W, Parisi J, Scheithauer B, Rodriguez M, Lassmann H (2000). "Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination". Ann. Neurol. 47 (6): 707–17. doi:10.1002/1531-8249(200006)47:6<707::AID-ANA3>3.0.CO;2-Q. PMID 10852536.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Krishnamoorthy G, Saxena A, Mars LT, Domingues HS, Mentele R, Ben-Nun A, Lassmann H, Dornmair K, Kurschus FC, Liblau RS, Wekerle H (2009). "Myelin-specific T cells also recognize neuronal autoantigen in a transgenic mouse model of multiple sclerosis". Nat. Med. 15 (6): 626–32. doi:10.1038/nm.1975. PMID 19483694.
{{cite journal}}: Unknown parameter|laysource=ignored (help); Unknown parameter|laysummary=ignored (help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Pöllinger B, Krishnamoorthy G, Berer K, Lassmann H, Bösl MR, Dunn R, Domingues HS, Holz A, Kurschus FC, Wekerle H (2009). "Spontaneous relapsing-remitting EAE in the SJL/J mouse: MOG-reactive transgenic T cells recruit endogenous MOG-specific B cells". J. Exp. Med. 206 (6): 1303–16. doi:10.1084/jem.20090299. PMC 2715069. PMID 19487416.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Trojano M, Paolicelli D (2001). "The differential diagnosis of multiple sclerosis: classification and clinical features of relapsing and progressive neurological syndromes". Neurol. Sci. 22 (Suppl 2): S98–102. doi:10.1007/s100720100044. PMID 11794488.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Poser CM, Brinar VV (2004). "Diagnostic criteria for multiple sclerosis: an historical review". Clin Neurol Neurosurg. 106 (3): 147–58. doi:10.1016/j.clineuro.2004.02.004. PMID 15177763.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d McDonald WI, Compston A, Edan G; et al. (2001). "Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis". Ann. Neurol. 50 (1): 121–7. doi:10.1002/ana.1032. PMID 11456302.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Polman CH, Reingold SC, Edan G; et al. (2005). "Diagnostic criteria for multiple sclerosis: 2005 revisions to the "McDonald Criteria"". Ann. Neurol. 58 (6): 840–6. doi:10.1002/ana.20703. PMID 16283615.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Rashid W, Miller DH (2008). "Recent advances in neuroimaging of multiple sclerosis". Semin Neurol. 28 (1): 46–55. doi:10.1055/s-2007-1019127. PMID 18256986.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Link H, Huang YM (2006). "Oligoclonal bands in multiple sclerosis cerebrospinal fluid: an update on methodology and clinical usefulness". J. Neuroimmunol. 180 (1–2): 17–28. doi:10.1016/j.jneuroim.2006.07.006. PMID 16945427.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Gronseth GS, Ashman EJ (2000). "Practice parameter: the usefulness of evoked potentials in identifying clinically silent lesions in patients with suspected multiple sclerosis (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology". Neurology. 54 (9): 1720–5. PMID 10802774.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ The Royal College of Physicians (2004). Multiple Sclerosis. National clinical guideline for diagnosis and management in primary and secondary care (PDF). Salisbury, Wiltshire: Sarum ColourView Group. pp. 54–57. ISBN 1 86016 182 0. Retrieved on 2010-01-07.
- ^ Burton JM, O'Connor PW, Hohol M, Beyene J (2009). "Oral versus intravenous steroids for treatment of relapses in multiple sclerosis". Cochrane Database Syst Rev (3): CD006921. doi:10.1002/14651858.CD006921.pub2. PMID 19588409.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c Comi G (2009). "Treatment of multiple sclerosis: role of natalizumab". Neurol. Sci. 30 Suppl 2: S155–8. doi:10.1007/s10072-009-0147-2. PMID 19882365.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Johnson KP (2007). "Control of multiple sclerosis relapses with immunomodulating agents". J. Neurol. Sci. 256 (Suppl 1): S23–8. doi:10.1016/j.jns.2007.01.060. PMID 17350652.
- ^ Martinelli Boneschi F, Rovaris M, Capra R, Comi G (2005). "Mitoxantrone for multiple sclerosis". Cochrane database of systematic reviews (Online) (4): CD002127. doi:10.1002/14651858.CD002127.pub2. PMID 16235298.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Leary SM, Thompson AJ (2005). "Primary progressive multiple sclerosis: current and future treatment options". CNS drugs. 19 (5): 369–76. doi:10.2165/00023210-200519050-00001. PMID 15907149.
- ^ Sládková T, Kostolanský F (2006). "The role of cytokines in the immune response to influenza A virus infection". Acta Virol. 50 (3): 151–62. PMID 17131933.
- ^ Munari L, Lovati R, Boiko A (2004). "Therapy with glatiramer acetate for multiple sclerosis". Cochrane database of systematic reviews (Online) (1): CD004678. doi:10.1002/14651858.CD004678. PMID 14974077.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Tremlett H, Oger J (2004). "Hepatic injury, liver monitoring and the beta-interferons for multiple sclerosis". J. Neurol. 251 (11): 1297–303. doi:10.1007/s00415-004-0619-5. PMID 15592724.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Kesselring J, Beer S (2005). "Symptomatic therapy and neurorehabilitation in multiple sclerosis". The Lancet Neurology. 4 (10): 643–52. doi:10.1016/S1474-4422(05)70193-9. PMID 16168933.
- ^ Khan F, Turner-Stokes L, Ng L, Kilpatrick T (2007). "Multidisciplinary rehabilitation for adults with multiple sclerosis". Cochrane Database Syst Rev (2): CD006036. doi:10.1002/14651858.CD006036.pub2. PMID 17443610.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ FDA Approves Ampyra to Improve Walking in Adults with Multiple Sclerosis
- ^ http://www.nationalmssociety.org/news/news-detail/index.aspx?nid=2586
- ^ Farinotti M, Simi S, Di Pietrantonj C; et al. (2007). "Dietary interventions for multiple sclerosis". Cochrane database of systematic reviews (Online) (1): CD004192. doi:10.1002/14651858.CD004192.pub2. PMID 17253500.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Chong MS, Wolff K, Wise K, Tanton C, Winstock A, Silber E (2006). "Cannabis use in patients with multiple sclerosis". Mult. Scler. 12 (5): 646–51. doi:10.1177/1352458506070947. PMID 17086912.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Zajicek JP, Sanders HP, Wright DE, Vickery PJ, Ingram WM, Reilly SM, Nunn AJ, Teare LJ, Fox PJ, Thompson AJ (2005). "Cannabinoids in multiple sclerosis (CAMS) study: safety and efficacy data for 12 months follow up". J. Neurol. Neurosurg. Psychiatr. 76 (12): 1664–9. doi:10.1136/jnnp.2005.070136. PMC 1739436. PMID 16291891.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bennett M, Heard R (2004). "Hyperbaric oxygen therapy for multiple sclerosis". Cochrane database of systematic reviews (Online) (1): CD003057. doi:10.1002/14651858.CD003057.pub2. PMID 14974004.
- ^ Oken BS, Kishiyama S, Zajdel D; et al. (2004). "Randomized controlled trial of yoga and exercise in multiple sclerosis". Neurology. 62 (11): 2058–64. PMID 15184614.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ a b c Phadke JG (1987). "Survival pattern and cause of death in patients with multiple sclerosis: results from an epidemiological survey in north east Scotland". J. Neurol. Neurosurg. Psychiatr. 50 (5): 523–31. doi:10.1136/jnnp.50.5.523. PMC 1031962. PMID 3495637.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Myhr KM, Riise T, Vedeler C; et al. (2001). "Disability and prognosis in multiple sclerosis: demographic and clinical variables important for the ability to walk and awarding of disability pension". Mult. Scler. 7 (1): 59–65. PMID 11321195.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c Alonso A, Hernán MA (2008). "Temporal trends in the incidence of multiple sclerosis: a systematic review". Neurology. 71 (2): 129–35. doi:10.1212/01.wnl.0000316802.35974.34. PMID 18606967.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Kulie T, Groff A, Redmer J, Hounshell J, Schrager S (2009). "Vitamin D: an evidence-based review". J Am Board Fam Med. 22 (6): 698–706. doi:10.3122/jabfm.2009.06.090037. PMID 19897699.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Berrios G.E. and Quemada J I (1995) Multiple Sclerosis. In Berrios G.E. and Porter R (eds) The History of Clinical Psychiatry. London, Athlone Press, pp174–192
- ^ Compston A (1988). "The 150th anniversary of the first depiction of the lesions of multiple sclerosis". J. Neurol. Neurosurg. Psychiatr. 51 (10): 1249–52. doi:10.1136/jnnp.51.10.1249. PMC 1032909. PMID 3066846.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Medaer R (1979). "Does the history of multiple sclerosis go back as far as the 14th century?". Acta Neurol. Scand. 60 (3): 189–92. doi:10.1111/j.1600-0447.1979.tb08970.x. PMID 390966.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Holmøy T (2006). "A Norse contribution to the history of neurological diseases". Eur. Neurol. 55 (1): 57–8. doi:10.1159/000091431. PMID 16479124.
- ^ Firth, D (1948). The Case of August D`Esté. Cambridge: Cambridge University Press.
- ^ a b Pearce JM (2005). "Historical descriptions of multiple sclerosis". Eur. Neurol. 54 (1): 49–53. doi:10.1159/000087387. PMID 16103678.
- ^ Barbellion, Wilhelm Nero Pilate (1919). The Journal of a Disappointed Man. New York: George H. Doran.
- ^ United Kingdom early Mitoxantrone Copaxone trial. Onyx Healthcare (2006-01-01). Retrieved on 2007-09-02.
- ^ Genzyme and Bayer HealthCare Announce Detailed Interim Two-Year Alemtuzumab in Multiple Sclerosis Data Presented at AAN. Genzyme (2007-02-01). Retrieved on 2007-09-02.
- ^ Daclizumab. PDL Biopharma (2006-01-01). Retrieved on 2007-09-02.
- ^ Treatment of Multiple Sclerosis Using Over the Counter Inosine. ClinicalTrials.gov (2006-03-16). Retrieved on 2007-09-02.
- ^ Efficacy and Safety of BG00012 in Relapsing-Remitting Multiple Sclerosis. ClinicalTrials.gov (2007-09-01). Retrieved on 2007-11-12.
- ^ Efficacy and Safety of Fingolimod in Patients With Relapsing-Remitting Multiple Sclerosis. ClinicalTrials.gov (2006-02-09). Retrieved on 2007-09-02.
- ^ The CAMMS223 Trial Investigators (2008). "Alemtuzumab vs. Interferon Beta-1a in Early Multiple Sclerosis". N Engl J Med. 359 (17): 1786–1801. doi:10.1056/NEJMoa0802670. PMID 18946064.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ Polman C, Barkhof F, Sandberg-Wollheim M, Linde A, Nordle O, Nederman T (2005). "Treatment with laquinimod reduces development of active MRI lesions in relapsing MS". Neurology. 64 (6): 987–91. PMID 15781813.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Darlington CL (2005). "Technology evaluation: NeuroVax, Immune Response Corp". Curr. Opin. Mol. Ther. 7 (6): 598–603. PMID 16370383.
- ^ Agrawal YP (2005). "Low dose naltrexone therapy in multiple sclerosis". Med. Hypotheses. 64 (4): 721–4. doi:10.1016/j.mehy.2004.09.024. PMID 15694688.
- ^ search of clinicaltrials.gov data-base for Low dose naltrexone Multiple Sclerosis
- ^ 2007 clinical trial using LDN
- ^ Gironi M, Martinelli-Boneschi F, Sacerdote P, Solaro C, Zaffaroni M, Cavarretta R, Moiola L, Bucello S, Radaelli M, Pilato V, Rodegher M, Cursi M, Franchi S, Martinelli V, Nemni R, Comi G, Martino G (2008). "A pilot trial of low-dose naltrexone in primary progressive multiple sclerosis". Multiple Sclerosis. 14 (8): 1076–83. doi:10.1177/1352458508095828. PMID 18728058.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Albrecht P, Fröhlich R, Hartung HP, Kieseier BC, Methner A (2007). "Optical coherence tomography measures axonal loss in multiple sclerosis independently of optic neuritis". J Neurol. Online (11): 1595. doi:10.1007/s00415-007-0538-3. PMID 17987252.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gordon-Lipkin E, Chodkowski B, Reich DS; et al. (2007). "Retinal nerve fiber layer is associated with brain atrophy in multiple sclerosis". Neurology. 69 (16): 1603–09. doi:10.1212/01.wnl.0000295995.46586.ae. PMID 17938370.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Quintana FJ, Farez MF, Viglietta V, Iglesias AH, Merbl Y, Izquierdo G, Lucas M, Basso AS, Khoury SJ, Lucchinetti CF, Cohen IR, Weiner HL. (2008). "Antigen microarrays identify unique serum autoantibody signatures in clinical and pathologic subtypes of multiple sclerosis". Proceeding of the National Academy of Sciences. 105 (48): 18889–94. doi:10.1073/pnas.0806310105. PMC 2596207. PMID 19028871.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Thangarajh M, Gomez-Rial J, Hedström AK; et al. (2008). "Lipid-specific immunoglobulin M in CSF predicts adverse long-term outcome in multiple sclerosis". Mult. Scler. 14 (9): 1208. doi:10.1177/1352458508095729. PMID 18755821.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Zamboni, R; Galeotti, R; Menegatti, E; Malagoni, AM; Tacconi, G; Dall’Ara, S; Bartolomei, I; Salvi, F (2009). "Chronic Cerebrospinal venous insufficiency in patients with multiple sclerosis". Journal of Neurology, Neurosurgery and Psychiatry. 80 (4). BMJ Publishing Group: 392–399. doi:10.1136/jnnp.2008.157164. ISSN 1468-330X. Retrieved 21 December 2009.
{{cite journal}}: More than one of|first1=and|first=specified (help) - ^ a b "Angioplasty for MS? Italian surgeon's claims prompt media circus, but Canadian doctors are skeptical". Medical Post. Retrieved Dec 16, 2009.
- ^ MS Resource Centre http://www.msrc.co.uk/index.cfm?CFID=2797780&CFTOKEN=19383080
Further reading
- Langgartner M, Langgartner I, Drlicek M (2005). "The patient's journey: multiple sclerosis". BMJ. 330 (7496): 885–8. doi:10.1136/bmj.330.7496.885. PMC 556161. PMID 15831874.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)
External links
- Multiple sclerosis at Curlie
- Database for analysis and comparison of global data on the epidemiology of MS
- NIH listing of clinical trials related to MS
- Abstract index of the Cochrane Library
- GeneReview/NCBI/NIH/UW entry on Multiple Sclerosis Overview
