11-Deoxycortisol

| |

| |
| Names | |
|---|---|
| IUPAC name
17α,21-Dihydroxypregn-4-ene-3,20-dione
| |
| Systematic IUPAC name
(1R,3aS,3bR,9aR,9bS,11aS)-1-Hydroxy-1-(2-hydroxy-1-oxoethyl)-9a,11a-dimethyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H-cyclopenta[a]phenanthren-7-one | |
| Other names
11-Deoxycortisol; 11-Deoxycortisone; Cortoxelone; 17α,21-Dihydroxyprogesterone; 11-Desoxycortisol; 11-Deoxyhydrocortisone; 11-Desoxyhydrocortisone; 17α-Hydroxy-11-deoxycorticosterone; Reichstein's Substance S; Compound S; Cortodoxone; Cortexolone,
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.279 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H30O4 | |
| Molar mass | 346.467 g·mol−1 |
| Melting point | 215 °C (419 °F; 488 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
11-Deoxycortisol, also known as cortodoxone (INN), cortexolone[1][2][3][4] as well as 17α,21-dihydroxyprogesterone or 17α,21-dihydroxypregn-4-ene-3,20-dione,[5] is an endogenous glucocorticoid steroid hormone, and a metabolic intermediate toward cortisol. It was first described by Tadeusz Reichstein in 1938 as Substance S,[6] thus has also been referred to as Reichstein's Substance S[5] or Compound S.[7][8]
Function
[edit]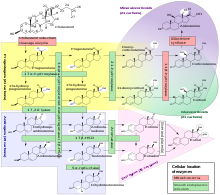
11-Deoxycortisol acts as a glucocorticoid, though is less potent than cortisol.[10]
11-Deoxycortisol is synthesized from 17α-hydroxyprogesterone by 21-hydroxylase and is converted to cortisol by 11β-hydroxylase.
11-Deoxycortisol in mammals has limited biological activity and mainly acts as metabolic intermediate within the glucocorticoid pathway, leading to cortisol.[11] However, in sea lampreys, an early jawless fish species that originated over 500 million years ago, 11-deoxycortisol plays a crucial role as the primary and ultimate glucocorticoid hormone with mineralocorticoid properties; 11-deoxycortisol also takes part, by binding to specific corticosteroid receptors, in intestinal osmoregulation in sea lamprey at metamorphosis, during which they develop seawater tolerance before downstream migration.[12] Sea lampreys do not possess the 11β-hydroxylase enzyme (CYP11B1) responsible for converting 11-deoxycortisol to cortisol and 11-deoxycorticosterone to corticosterone, as observed in mammals. The absence of this enzyme in sea lampreys indicates the existence of a complex and highly specific corticosteroid signaling pathway that emerged at least 500 million years ago with the advent of early vertebrates. The lack of cortisol and corticosterone in sea lampreys suggests that the presence of the 11β-hydroxylase enzyme may have been absent during the early stages of vertebrate evolution.[13] The absence of cortisol and corticosterone in sea lampreys suggests that the 11β-hydroxylase enzyme may not have been present early in vertebrate evolution.[14]
Clinical significance
[edit]11-Deoxycortisol in mammals has limited glucocorticoid activity, but it is the direct precursor of the major mammalian glucocorticoid, cortisol.[15] As a result, the level of 11-deoxycortisol is measured to diagnose impaired cortisol synthesis, to find out the enzyme deficiency that causes impairment along the pathway to cortisol, and to differentiate adrenal disorders.[16]
In 11β-hydroxylase deficiency, 11-deoxycortisol and 11-deoxycorticosterone levels increase, and excess of 11-deoxycorticosterone leads to mineralocorticoid-based hypertension[17] (as opposed to 21-hydroxylase deficiency, in which patients have low blood pressure from a lack of mineralocorticoids). Low levels of cortisol can affect blood pressure by causing a decrease in sodium retention and volume expansion. This is because cortisol plays a role in regulating the balance of water and electrolytes in the body. When cortisol levels are low, there is less sodium reabsorption by the kidneys, leading to increased excretion of sodium through urine. This ultimately reduces blood volume and lowers blood pressure. On the other hand, high levels of cortisol can also affect blood pressure by causing an increase in sodium retention and volume expansion. Cortisol-induced hypertension is accompanied by significant sodium retention, leading to an increase in extracellular fluid volume and exchangeable sodium. This results in an increase in blood volume and subsequently increases blood pressure. The underlying mechanisms for these effects involve various factors such as suppression of the nitric oxide system, alterations in vascular responsiveness to pressor agonists like adrenaline, increased cardiac output or stroke volume due to plasma volume expansion, and potential dysregulation of glucocorticoid receptors or 11β-hydroxylase enzyme activity. It's important to note that these mechanisms may be relevant not only for cortisol-induced hypertension but also for conditions such as Cushing's syndrome (excess cortisol production), apparent mineralocorticoid excess (related to defects in 11β-hydroxylase enzymes), licorice abuse (glycyrrhetinic acid affecting glycyrrhetinic acid receptor),[18] chronic renal failure (prolonged half-life of cortisol due to reduced 11β-hydroxylase activity), and even essential hypertension where there may be abnormalities with 11β-hydroxylase activity or glucocorticoid receptor variations.[19][20][21] Low levels of cortisol lead to reduced vascular tone as cortisol helps maintain normal vascular tone by promoting vasoconstriction. Low levels of cortisol can lead to decreased vasoconstriction, resulting in relaxed blood vessels and lower overall blood pressure. Also, low cortisol levels lead to impaired fluid balance, as cortisol affects fluid balance by influencing sodium and water reabsorption in the kidneys. When cortisol levels are low, sodium absorption may be reduced, leading to increased excretion of sodium in the urine and subsequent lowering of blood volume and blood pressure. Additionally, low levels of cortisol cause a dysregulated renin-angiotensin system, as cortisol interacts with the renin-angiotensin system, which regulates blood pressure through vasoconstriction and fluid balance. Low cortisol levels can disrupt this system, leading to altered angiotensin production, reduced aldosterone secretion, and subsequently lower blood pressure. Conversely, high levels of cortisol lead to increased vascular tone, enhanced sodium retention, and increased sympathetic activity. Stress-induced release of high-level glucocorticoids such as cortisol activates the sympathetic nervous system (SNS). The SNS controls heart rate, cardiac output, and vasomotor tone, causing constriction, and thereby increasing peripheral arterial resistance, resulting in an increase in blood pressure.[22] In 11β-hydroxylase deficiency, 11-deoxycortisol can also be converted to androstenedione in a pathway that could explain the increase in androstenedione levels this condition.[23]
In 21-hydroxylase deficiency, 11-deoxycortisol levels are low.[24]
History
[edit]In 1934, biochemist Tadeus Reichstein, working in Switzerland, began research on extracts from animal adrenal glands in order to isolate physiologically active compounds.[25] He was publishing results of his findings along the way. By 1944, he already isolated and elucidated the chemical structure of 29 pure substances.[26] He was assigning names that consisted of the word "Substance" and a letter from the Latin alphabet to the newly found substances. In 1938, he published an article about "Substance R" and "Substance S" describing their chemical structures and properties.[6] The Substance S since about 1955 became known as 11-Deoxycortisol.[27]
In 1949, American research chemist Percy Lavon Julian, in looking for ways to produce cortisone, announced the synthesis of the Compound S, from the cheap and readily available pregnenolone (synthesized from the soybean oil sterol stigmasterol).[28][29]
On 5 April 1952, biochemist Durey Peterson and microbiologist Herbert Murray at Upjohn, published the first report of a breakthrough fermentation process for the microbial 11α-oxygenation of steroids (e.g. progesterone) in a single step by common molds of the order Mucorales.[30] 11α-oxygenation of Compound S produces 11α-hydrocortisone, which can be chemically oxidized to cortisone, or converted by further chemical steps to 11β-hydrocortisone (cortisol).
See also
[edit]References
[edit]- ^ Rosette C, Agan FJ, Rosette N, Mazzetti A, Moro L, Gerloni M (August 2020). "The dual androgen receptor and glucocorticoid receptor antagonist CB-03-10 as a potential treatment for tumors that have acquired GR-mediated resistance to AR blockade". Molecular Cancer Therapeutics. 19 (11): 2256–2266. doi:10.1158/1535-7163.MCT-19-1137. PMID 32847976. S2CID 221345484.
- ^ Iaderets VV, Andriushina VA, Bartoshevich IE, Domracheva AG, Novak MI, Stytsenko TS, Voĭshvillo NE (2007). "[A study of steroid hydroxylation activity of Curvularia lunata mycelium]". Prikladnaia Biokhimiia I Mikrobiologiia (in Russian). 43 (6): 695–700. PMID 18173113.
- ^ Manosroi J, Chisti Y, Manosroi A (2006). "Biotransformation of cortexolone to hydrocortisone by molds using a rapid color development assay". Prikladnaia Biokhimiia I Mikrobiologiia. 42 (5): 547–51. PMID 17066954.
- ^ Lisowska K, Długoński J (May 2003). "Concurrent corticosteroid and phenanthrene transformation by filamentous fungus Cunninghamella elegans". The Journal of Steroid Biochemistry and Molecular Biology. 85 (1): 63–9. doi:10.1016/s0960-0760(03)00136-5. PMID 12798358. S2CID 384672.
- ^ a b R.A. Hill, H.L.J. Makin, D.N. Kirk, G.M. Murphy (23 May 1991). Dictionary of Steroids. CRC Press. pp. 338–. ISBN 978-0-412-27060-4.
- ^ a b Reichstein T (1938). "Über Bestandteile der Nebennierenrinde. 21. Mitteilung. Die Konstitution der Substanzen R und S". Helvetica Chimica Acta. 21: 1490–1497. doi:10.1002/hlca.193802101183. Archived from the original on 10 October 2020. Retrieved 5 October 2020.
- ^ Wudy SA, Hartmann M, Homoki J (September 2002). "Determination of 11-deoxycortisol (Reichstein's compound S) in human plasma by clinical isotope dilution mass spectrometry using benchtop gas chromatography-mass selective detection". Steroids. 67 (10): 851–7. doi:10.1016/s0039-128x(02)00052-1. PMID 12231120. S2CID 29970823.
- ^ Berneis K, Staub JJ, Gessler A, Meier C, Girard J, Müller B (December 2002). "Combined stimulation of adrenocorticotropin and compound-S by single dose metyrapone test as an outpatient procedure to assess hypothalamic-pituitary-adrenal function". The Journal of Clinical Endocrinology and Metabolism. 87 (12): 5470–5. doi:10.1210/jc.2001-011959. PMID 12466339.
- ^ Häggström M, Richfield D (2014). "Diagram of the pathways of human steroidogenesis". WikiJournal of Medicine. 1 (1). doi:10.15347/wjm/2014.005. ISSN 2002-4436.
- ^ Engels M, Pijnenburg-Kleizen KJ, Utari A, Faradz SM, Oude-Alink S, van Herwaarden AE, Span PN, Sweep FC, Claahsen-van der Grinten HL (November 2019). "Glucocorticoid Activity of Adrenal Steroid Precursors in Untreated Patients With Congenital Adrenal Hyperplasia". The Journal of Clinical Endocrinology and Metabolism. 104 (11): 5065–5072. doi:10.1210/jc.2019-00547. PMID 31090904.
- ^ "Congenital Adrenal Hyperplasia Caused by 11Beta-Hydroxylase Deficiency". Merck Manuals Professional Edition. Archived from the original on 25 September 2020. Retrieved 3 October 2020.
- ^ Barany A, Shaughnessy CA, McCormick SD (March 2021). "Corticosteroid control of Na+/K+-ATPase in the intestine of the sea lamprey (Petromyzon marinus)". General and Comparative Endocrinology. 307: 113756. doi:10.1016/j.ygcen.2021.113756. PMID 33741310. S2CID 232296805.
- ^ Shaughnessy CA, Barany A, McCormick SD (July 2020). "11-Deoxycortisol controls hydromineral balance in the most basal osmoregulating vertebrate, sea lamprey (Petromyzon marinus)". Scientific Reports. 10 (1): 12148. Bibcode:2020NatSR..1012148S. doi:10.1038/s41598-020-69061-4. PMC 7376053. PMID 32699304.
- ^ Close DA, Yun SS, McCormick SD, Wildbill AJ, Li W (August 2010). "11-deoxycortisol is a corticosteroid hormone in the lamprey". Proceedings of the National Academy of Sciences of the United States of America. 107 (31): 13942–7. Bibcode:2010PNAS..10713942C. doi:10.1073/pnas.0914026107. PMC 2922276. PMID 20643930.
- ^ Zöllner A, Kagawa N, Waterman MR, Nonaka Y, Takio K, Shiro Y, Hannemann F, Bernhardt R (February 2008). "Purification and functional characterization of human 11beta hydroxylase expressed in Escherichia coli". The FEBS Journal. 275 (4): 799–810. doi:10.1111/j.1742-4658.2008.06253.x. PMID 18215163. S2CID 45997341.
- ^ "Serum steroid levels can help differentiate adrenal disorders". Archived from the original on 7 November 2020. Retrieved 3 October 2020.
- ^ Gupta V (October 2011). "Mineralocorticoid hypertension". Indian Journal of Endocrinology and Metabolism. 15 (Suppl 4): S298-312. doi:10.4103/2230-8210.86972. PMC 3230101. PMID 22145132.
- ^ Li YL, Zhu XM, Liang H, Orvig C, Chen ZF (2021). "Recent Advances in Asialoglycoprotein Receptor and Glycyrrhetinic Acid Receptor-Mediated and/or pH-Responsive Hepatocellular Carcinoma- Targeted Drug Delivery". Curr Med Chem. 28 (8): 1508–1534. doi:10.2174/0929867327666200505085756. PMID 32368967.
- ^ Kelly JJ, Mangos G, Williamson PM, Whitworth JA (November 1998). "Cortisol and hypertension". Clin Exp Pharmacol Physiol Suppl. 25: S51–6. doi:10.1111/j.1440-1681.1998.tb02301.x. PMID 9809193. S2CID 13428727.
- ^ van Uum SH, Lenders JW, Hermus AR (May 2004). "Cortisol, 11beta-hydroxysteroid dehydrogenases, and hypertension". Semin Vasc Med. 4 (2): 121–8. doi:10.1055/s-2004-835369. PMID 15478032. S2CID 260371999.
- ^ Stewart PM (1998). "Cortisol, hypertension and obesity: the role of 11 beta-hydroxysteroid dehydrogenase". J R Coll Physicians Lond. 32 (2): 154–9. PMC 9663025. PMID 9597634.
- ^ Lewandowski KC, Tadros-Zins M, Horzelski W, Krekora M, Lewinski A (April 2023). "Renin, Aldosterone, and Cortisol in Pregnancy-Induced Hypertension". Exp Clin Endocrinol Diabetes. 131 (4): 222–227. doi:10.1055/a-2025-0510. PMC 10101736. PMID 36807213.
- ^ Auzéby A, Bogdan A, Touitou Y (January 1991). "Evidence for a new biologic pathway of androstenedione synthesis from 11-deoxycortisol". Steroids. 56 (1): 33–6. doi:10.1016/0039-128X(91)90112-9. PMID 2028480. S2CID 6261965.
- ^ Essential Endocrinology: A Primer for Nonspecialists. Springer Science & Business Media. 2013. p. 263. ISBN 978-1-4899-1692-1.
- ^ "Chemistry of the Adrenal Cortex Hormones". Archived from the original on 10 October 2020. Retrieved 5 October 2020.
- ^ Grzybowski A, Pietrzak K (2012). "Tadeusz Reichstein (1897-1996): a cofounder of modern steroid treatment in dermatology". Clinics in Dermatology. 30 (2): 243–7. doi:10.1016/j.clindermatol.2011.09.003. PMID 22435119.
- ^ Forchielli E, Rosenkrantz H, Dorfman RI (1 August 1955). "Metabolism of 11-deoxycortisol in vitro". The Journal of Biological Chemistry. 215 (2): 713–22. doi:10.1016/S0021-9258(18)65996-4. PMID 13242571. Archived from the original on 17 November 2020. Retrieved 9 November 2020.
- ^ "Science gets synthetic key to rare drug; discovery is made in Chicago". Chicago Tribune. 30 September 1949. p. 1. Archived from the original on 31 January 2013.
Dr. Julian's new method for synthesizing the anti-arthritis compound, cortisone, is less costly than present methods, because it eliminates the need for utilizing osmium tetroxide, a rare and expensive chemical, the Glidden company declared....But whether has Dr. Julian has also synthesized cortisone from soybeans neither he nor the Glidden company would reveal.
- ^ "News of the week: New cortisone synthesis". Chemical & Engineering News. 27 (41): 2936–2942. 10 October 1949. doi:10.1021/cen-v027n041.p2936.
Quote: A new synthesis of cortisone, eliminating the need for expensive osmium tetroxide, and the synthesis of three other compounds related to cortisone, which may possible be useful in the treatment of arthritis, have been announced by Percy L. Julian, director of research of the soya products division of the Glidden Co., Chicago. No statement was made as to further details of the new synthesis, but it was revealed that soybean products were not involved...all three [other compounds] were made from soybean sterols.
- ^ Peterson DH, Murray, HC (1952). "Microbiological oxygenation of steroids at carbon 11". J Am Chem Soc. 74 (7): 1871–2. doi:10.1021/ja01127a531.
