Edmond H. Fischer
Edmond H. Fischer | |
|---|---|
 Fischer in 2016 | |
| Born | Edmond Henri Fischer April 6, 1920 |
| Died | August 27, 2021 (aged 101) Seattle, Washington, U.S. |
| Alma mater | University of Geneva |
| Known for | Protein phosphorylation |
| Spouses | Nelly Gagnaux (died); Beverly Bullock |
| Awards |
|
| Scientific career | |
| Institutions | University of Washington |
| Thesis | La purification et l'isolement de l'alpha-amylase de pancréas (1947) |
| Doctoral advisor | Kurt Heinrich Meyer |
| Website | depts |
Edmond Henri Fischer (April 6, 1920 – August 27, 2021) was a Swiss-American biochemist. He and his collaborator Edwin G. Krebs were awarded the Nobel Prize in Physiology or Medicine in 1992 for describing how reversible phosphorylation works as a switch to activate proteins and regulate various cellular processes.[3] From 2007 until 2014, he was the Honorary President of the World Cultural Council.[4][5][6][7] At the time of his death at age 101 in 2021, he was the oldest living Nobel Prize laureate.[8]
Early life[edit]
Fischer was born on April 6, 1920, in the Shanghai International Settlement, China.[9][10] His mother, Renée Tapernoux, was born in France, and his father, Oscar Fischer, was born in Austria.[11][10] His father, who was Jewish,[12] practiced as a lawyer in Shanghai before the various consular courts in the city.[13] Fischer's maternal grandfather founded the Courrier de Chine in Shanghai, the first newspaper published in French in China; he also helped to establish L'Ecole Municipale Française in Shanghai, where Fischer attended primary school.[14]
At age seven, Fischer and his two elder brothers, Raoul and George,[15] were sent to the Swiss boarding school La Châtaigneraie, near his mother's hometown in Vevey. He picked up mountain climbing and skiing during his time at the school.[10] At high school he made a pact with a childhood friend, one of them would become a doctor and the other a scientist and then they could cure the ills of the world.[16] While at high school Fischer was admitted to the Geneva Conservatory of Music as a pianist and he also considered becoming a professional musician.[10]
At the completion of high school, Fischer wanted to study microbiology inspired by the works of French chemist and microbiologist Louis Pasteur and partly driven by his father's death from tuberculosis;[10] however, he was advised to study chemistry. He studied at the University of Geneva during World War II, he enjoyed organic chemistry and also studied biology. He completed a PhD in organic chemistry under the supervision of Kurt Heinrich Meyer, who worked on the structure of polysaccharides, and the enzymes needed for their synthesis and breakdown. Fischer worked on α-amylase.[17][18]
Career and research[edit]
After his PhD, Fischer went to the United States in 1950 for postdoctoral research. He was supposed to take up a position at Caltech, but he was also, unexpectedly, offered a position at the University of Washington in Seattle. Seattle reminded Fischer and his wife of Switzerland so they chose to settle there.[19]
Protein phosphorylation/hydrolysis cycle[edit]
Six months after his arrival in Seattle, Fischer learnt of fellow biochemist from the same university, Edwin G. Krebs, who was also trying to answer a similar question on where muscles received the energy that they needed to contract. Fischer began collaborating with Krebs, and the duo began their investigations on glycogen phosphorylase, an enzyme that had been discovered by the husband and wife pair of Gerty Cori and Carl Ferdinand Cori who had won the Nobel Prize for the discovery in 1947.[10] Krebs had earlier studied the enzyme in the muscle tissue, while Fischer had studied the enzyme in a potato. The duo realized a discrepancy in that the enzyme in the muscle required an additional chemical to function, while the potato did not require that additional chemical.[10] Krebs and Fischer defined a series of reactions leading to the activation/inactivation of this enzyme as triggered by hormones and calcium, and in the process discovering the cycle of protein phosphorylation and hydrolysis.
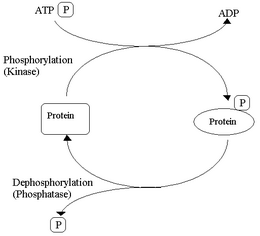
Explained simply, the cycle works like this: a protein kinase moves a phosphate group from adenosine triphosphate (ATP) to a protein, converting it to adenosine diphosphate (ADP). The shape and the function of this protein is thus altered enabling it to take part in converting glycogen into glucose which is used for fuel for muscular contractions. When the protein has completed its role a different enzyme, phosphatase, catalyses hydrolysis of the phosphorylated protein, which reverts to its original state. This cycle takes place to control many metabolic processes.[20][21]
While the importance of the discovery was not fully recognised in 1955, the discovery became core to explaining one of the fundamental mechanisms that cells use to communicate with one another.[10] Phosphorylation determines how a cell grows, divides, differentiates and eventually dies. The reaction also regulates hormones and proliferation of cancerous cells.[10] The regulation of phosphorylation was determined to be key to understanding diseases such as cancer, diabetes, and heart disease. Many modern drugs build on the work done by Fischer and Krebs including attempting to manipulate the process. For the discovery of the cycle of phosphorylation and hydrolysis Fischer and Krebs were awarded the Nobel Prize for Physiology or Medicine in 1992, for explaining how the reaction acted as a switch to activate proteins and regulate various biochemical cellular processes.[9][16]
Throughout his career, Fischer's research continued to look at the role of similar cycling in a variety of cellular processes.[10]
Personal life[edit]
Fischer married his first wife, Nelly Gagnaux, in 1948 and they remained married until her death in 1961. He married Beverly Bullock in 1963 who died in 2006.[10][16] Fischer played the piano and often performed sonatas by Beethoven and Mozart for his friends.[10] He also held a private pilots license and enjoyed flying.[16]
Fischer died on August 27, 2021, in Seattle, Washington. He was aged 101.[10][9]
Awards and honors[edit]
Fischer won numerous awards including the Nobel Prize in Physiology or Medicine in 1992. He was elected a Foreign Member of the Royal Society (ForMemRS) in 2010.[2] He was awarded the Werner Prize from the Swiss Chemical Society, the Lederle Medical Faculty Award, the Prix Jaubert from the University of Geneva, the Senior Passano Award and the Steven C. Beering Award from Indiana University. He received Doctorates Honoris Causa from the University of Montpellier, France and the University of Basel, Switzerland.[14] He was elected a Fellow of the American Academy of Arts and Sciences in 1972[2] and a Member of the National Academy of Sciences in 1973.[1] Fischer was a member of the St. George's University-based Windward Islands Research and Education Foundation (WINDREF) Scientific Advisory Board from 1994 to 2021.
See also[edit]
References[edit]
- ^ a b "Edmond H. Fischer, University of Washington, Election Year: 1973". nasonline.org. Archived from the original on October 10, 2015.
- ^ a b c "Edmond Fischer: Professor Emeritus of Biochemistry ForMemRS". London: Royal Society. Archived from the original on October 10, 2015.
- ^ "The Nobel Prize in Physiology or Medicine 1992 Press Release". Nobel Assembly at Karolinska Institutet. Retrieved August 21, 2013.
- ^ Almada López, Carlos. "World Cultural Council 24th Award Ceremony, Closing Remarks" (in Spanish). Archived from the original on December 25, 2014. Retrieved August 21, 2013.
- ^ Pacific Northwest Research Institute. About Edmond H. Fischer, Ph.D. Archived September 25, 2006, at the Wayback Machine
- ^ Hughes, R. 1998. After the Prize
- ^ Freeview video 'An Interview with Edmond Fischer' by the Vega Science Trust
- ^ "Nobel Laureate Edmond H. Fischer 1920 – 2021". Archived from the original on August 29, 2021. Retrieved August 29, 2021.
- ^ a b c Schudel, Matt (August 31, 2021), "Edmond Fischer, who won Nobel Prize for discoveries about proteins in cells, dies at 101", The Washington Post
- ^ a b c d e f g h i j k l m Imbler, Sabrina (September 2, 2021). "Edmond H. Fischer, 101, Who Helped Discover How Cells Talk, Dies". The New York Times. ISSN 0362-4331. Retrieved September 3, 2021.
- ^ Edmond H. Fischer Summary.
- ^ "Jewish Nobel Prize Winners in Medicine". www.jinfo.org. Retrieved March 30, 2023.
- ^ Oscar Fischer's biography in Men of Shanghai and North China, p. 129
- ^ a b "Edmond H. Fischer – Biographical". nobelprize.org. Retrieved August 21, 2013.
- ^ Oscar Fischer's biography in Men of Shanghai and North China, p129
- ^ a b c d "In Memoriam: Nobel Laureate Edmond Fischer". newsroom.uw.edu. August 31, 2021. Retrieved September 3, 2021.
- ^ Fischer, Edmond H.; Piguet, Alfred (December 1, 2009). "Macromolecules and Enzymes: The Geneva Heritage from Kurt H. Meyer and Edmond H. Fischer". CHIMIA. 63 (12): 835–838. doi:10.2533/chimia.2009.835. ISSN 0009-4293. PMID 28372606.
- ^ Vasmatkar, Pashupat; Kaur, Kamaljit. Miracles in Biochemistry: The contribution of the Nobel laureates to the field of Biochemistry. Educreation Publishing.
- ^ Haydon, Ian (August 31, 2021). "In Memoriam: Nobel Laureate Edmond Fischer".
- ^ Fischer, E H; Krebs, E G (1966), "Relationship of structure to function of muscle phosphorylase.", Fed. Proc., vol. 25, no. 5, pp. 1511–20, PMID 5332191
- ^ KREBS, E G; FISCHER, E H (1964), "Phosphorylase and related enzymes of Glycogen Metabolism", Vitam. Horm., Vitamins & Hormones, vol. 22, pp. 399–410, doi:10.1016/S0083-6729(08)60345-3, ISBN 9780127098227, PMID 14284112
External links[edit]
- 1920 births
- 2021 deaths
- Alumni of the Conservatoire de Musique de Genève
- American biochemists
- American centenarians
- American Nobel laureates
- American people of Austrian-Jewish descent
- American people of French descent
- American people of Swiss-Jewish descent
- Shanghai Nobel laureates
- Swiss emigrants to the United States
- Swiss people of Austrian descent
- Swiss people of French descent
- Swiss Nobel laureates
- Jewish American scientists
- Men centenarians
- Nobel laureates in Physiology or Medicine
- Members of the European Molecular Biology Organization
- University of Washington faculty
- Foreign Members of the Royal Society
- Members of the United States National Academy of Sciences
- Swiss expatriates in China
- Jewish centenarians
