Solar cell: Difference between revisions
Revert 2 edits by 74.108.233.243 |
No edit summary |
||
| Line 239: | Line 239: | ||
==External links== |
==External links== |
||
* [http://iranBattery.ir Battery is our business] |
|||
{{Commons category|Photovoltaics}} |
{{Commons category|Photovoltaics}} |
||
{{Commons|solar cell}} |
{{Commons|solar cell}} |
||
*[http://science.howstuffworks.com/solar-cell.htm Howstuffworks.com: How Solar Cells Work] |
*[http://science.howstuffworks.com/solar-cell.htm Howstuffworks.com: How Solar Cells Work] |
||
*[http://www.pveducation.org/pvcdrom Photovoltaics CDROM online] |
*[http://www.pveducation.org/pvcdrom Photovoltaics CDROM online] |
||
* [http://iranBattery.ir Battery is our business] |
|||
*[http://www.madehow.com/Volume-1/Solar-Cell.html Solar cell manufacturing techniques] |
*[http://www.madehow.com/Volume-1/Solar-Cell.html Solar cell manufacturing techniques] |
||
*{{dmoz|Science/Technology/Energy/Renewable/Solar/|Renewable Energy: Solar}} |
*{{dmoz|Science/Technology/Energy/Renewable/Solar/|Renewable Energy: Solar}} |
||
Revision as of 06:11, 16 January 2011


A solar cell (also called photovoltaic cell) is a solid state device that converts the energy of sunlight directly into electricity by the photovoltaic effect. Assemblies of cells are used to make solar modules, also known as solar panels. The energy generated from these solar modules, referred to as solar power, is an example of solar energy.
Photovoltaics is the field of technology and research related to the practical application of photovoltaic cells in producing electricity from light, though it is often used specifically to refer to the generation of electricity from sunlight.
Cells are described as photovoltaic cells when the light source is not necesssarily sunlight. These are used for detecting light or other electromagnetic radiation near the visible range, for example infrared detectors[1], or measurement of light intensity.
History of solar cells
The term "photovoltaic" comes from the Greek φῶς (phōs) meaning "light", and "voltaic", meaning electric, from the name of the Italian physicist Volta, after whom a unit of electro-motive force, the volt, is named. The term "photo-voltaic" has been in use in English since 1849.[2]
The photovoltaic effect was first recognized in 1839 by French physicist A. E. Becquerel. However, it was not until 1883 that the first solar cell was built, by Charles Fritts, who coated the semiconductor selenium with an extremely thin layer of gold to form the junctions. The device was only around 1% efficient. In 1888 Russian physicist Aleksandr Stoletov built the first photoelectric cell (based on the outer photoelectric effect discovered by Heinrich Hertz earlier in 1887). Albert Einstein explained the photoelectric effect in 1905 for which he received the Nobel prize in Physics in 1921. Russell Ohl patented the modern junction semiconductor solar cell in 1946,[3] which was discovered while working on the series of advances that would lead to the transistor. The photovoltaic cell was developed in 1954 at Bell Laboratories.[4] The highly efficient solar cell was first developed by Daryl Chapin, Calvin Souther Fuller and Gerald Pearson in 1954 using a diffused silicon p-n junction.[5] In the past four decades, remarkable progress has been made, with Megawatt solar power generating plants having now been built.[6]
Applications


Solar cells are often electrically connected and encapsulated as a module. Photovoltaic modules often have a sheet of glass on the front (sun up) side, allowing light to pass while protecting the semiconductor wafers from abrasion and impact due to wind-driven debris, rain, hail, et cetera. Solar cells are also usually connected in series in modules, creating an additive voltage. Connecting cells in parallel will yield a higher current. Modules are then interconnected, in series or parallel, or both, to create an array with the desired peak DC voltage and current.
To make practical use of the solar-generated energy, the electricity is most often fed into the electricity grid using inverters (grid-connected photovoltaic systems); in stand-alone systems, batteries are used to store the energy that is not needed immediately. Solar panels can be used to power or recharge portable devices.
Theory
The solar cell works in three steps:
- Photons in sunlight hit the solar panel and are absorbed by semiconducting materials, such as silicon.
- Electrons (negatively charged) are knocked loose from their atoms, allowing them to flow through the material to produce electricity. Due to the special composition of solar cells, the electrons are only allowed to move in a single direction.
- An array of solar cells converts solar energy into a usable amount of direct current (DC) electricity.
Efficiency
The efficiency of a solar cell may be broken down into reflectance efficiency, thermodynamic efficiency, charge carrier separation efficiency and conductive efficiency. The overall efficiency is the product of each of these individual efficiencies.
Due to the difficulty in measuring these parameters directly, other parameters are measured instead: thermodynamic efficiency, quantum efficiency, VOC ratio, and fill factor. Reflectance losses are a portion of the quantum efficiency under "external quantum efficiency". Recombination losses make up a portion of the quantum efficiency, VOC ratio, and fill factor. Resistive losses are predominantly categorized under fill factor, but also make up minor portions of the quantum efficiency, VOC ratio.
Crystalline silicon devices are now approaching the theoretical limiting efficiency of 29%.
Cost
The cost of a solar cell is given per unit of peak electrical power. Manufacturing costs necessarily including the cost of energy required for manufacture. Solar-specific feed in tariffs vary worldwide, and even state by state within various countries.[7] Such feed-in tariffs can be highly effective in encouraging the development of solar power projects.
High-efficiency solar cells are of interest to decrease the cost of solar energy. Many of the costs of a solar power plant are proportional to the area of the plant; a higher efficiency cell may reduce area and plant cost, even if the cells themselves are more costly. Efficiencies of bare cells, to be useful in evaluating solar power plant economics, must be evaluated under realistic conditions. The basic parameters that need to be evaluated are the short circuit current, open circuit voltage.[8]
The chart at the right illustrates the best laboratory efficiencies obtained for various materials and technologies, generally this is done on very small, i.e. one square cm, cells. Commercial efficiencies are significantly lower.
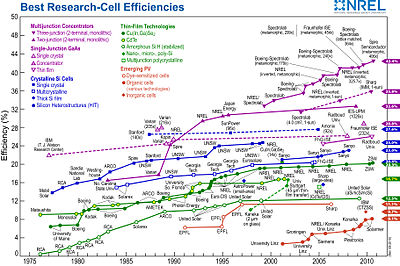
A low-cost photovoltaic cell is a thin-film cell intended to produce electrical energy at a price competitive with traditional (fossil fuels and nuclear power) energy sources. This includes second and third generation photovoltaic cells, that is cheaper than first generation (crystalline silicon cells, also called wafer or bulk cells).
Grid parity, the point at which photovoltaic electricity is equal to or cheaper than grid power, can be reached using low cost solar cells. It is achieved first in areas with abundant sun and high costs for electricity such as in California and Japan.[9] Grid parity has been reached in Hawaii and other islands that otherwise use diesel fuel to produce electricity. George W. Bush had set 2015 as the date for grid parity in the USA.[10][11] Speaking at a conference in 2007, General Electric's Chief Engineer predicted grid parity without subsidies in sunny parts of the United States by around 2015.[12]
The price of solar panels fell steadily for 40 years, until 2004 when high subsidies in Germany drastically increased demand there and greatly increased the price of purified silicon (which is used in computer chips as well as solar panels). One research firm predicted that new manufacturing capacity began coming on-line in 2008 (projected to double by 2009) which was expected to lower prices by 70% in 2015. Other analysts warned that capacity may be slowed by economic issues, but that demand may fall because of lessening subsidies. Other potential bottlenecks which have been suggested are the capacity of ingot shaping and wafer slicing industries, and the supply of specialist chemicals used to coat the cells.[13]
Materials
Different materials display different efficiencies and have different costs. Materials for efficient solar cells must have characteristics matched to the spectrum of available light. Some cells are designed to efficiently convert wavelengths of solar light that reach the Earth surface. However, some solar cells are optimized for light absorption beyond Earth's atmosphere as well. Light absorbing materials can often be used in multiple physical configurations to take advantage of different light absorption and charge separation mechanisms.
Materials presently used for photovoltaic solar cells include monocrystalline silicon, polycrystalline silicon, amorphous silicon, cadmium telluride, and copper indium selenide/sulfide.[14]
Many currently available solar cells are made from bulk material that are cut into wafers between 180 to 240 micrometers thick that are then processed like other semiconductors.
Other materials are made as thin-films layers, organic dyes, and organic polymers that are deposited on supporting substrates. A third group are made from nanocrystals and used as quantum dots (electron-confined nanoparticles). Silicon remains the only material that is well-researched in both bulk and thin-film forms.
Crystalline silicon

By far, the most prevalent bulk material for solar cells is crystalline silicon (abbreviated as a group as c-Si), also known as "solar grade silicon". Bulk silicon is separated into multiple categories according to crystallinity and crystal size in the resulting ingot, ribbon, or wafer.
- monocrystalline silicon (c-Si): often made using the Czochralski process. Single-crystal wafer cells tend to be expensive, and because they are cut from cylindrical ingots, do not completely cover a square solar cell module without a substantial waste of refined silicon. Hence most c-Si panels have uncovered gaps at the four corners of the cells.
- Poly- or multicrystalline silicon (poly-Si or mc-Si): made from cast square ingots — large blocks of molten silicon carefully cooled and solidified. Poly-Si cells are less expensive to produce than single crystal silicon cells, but are less efficient. US DOE data shows that there were a higher number of multicrystalline sales than monocrystalline silicon sales.
- Ribbon silicon[15] is a type of multicrystalline silicon: it is formed by drawing flat thin films from molten silicon and results in a multicrystalline structure. These cells have lower efficiencies than poly-Si, but save on production costs due to a great reduction in silicon waste, as this approach does not require sawing from ingots.
Analysts have predicted that prices of polycrystalline silicon will drop as companies build additional polysilicon capacity quicker than the industry’s projected demand. On the other hand, the cost of producing upgraded metallurgical-grade silicon, also known as UMG Si, can potentially be one-sixth that of making polysilicon.[16]
Manufacturers of wafer-based cells have responded to thin-film lower prices with rapid reductions in silicon consumption. According to Jef Poortmans, director of IMEC's organic and solar department,[17] current cells use between eight and nine grams of silicon per watt of power generation, with wafer thicknesses in the neighborhood of 0.200 mm. At 2008 spring's IEEE Photovoltaic Specialists' Conference (PVS'08),[18] John Wohlgemuth, staff scientist at BP Solar, reported that his company has qualified modules based on 0.180 mm thick wafers and is testing processes for 0.16 mm wafers cut with 0.1 mm wire. IMEC's roadmap, presented at the organization's recent annual research review meeting, envisions use of 0.08 mm wafers by 2015.[19]
Thin films
Thin-film technologies reduce the amount of material required in creating a solar cell. Though this reduces material cost, it may also reduce energy conversion efficiency. Thin-film silicon cells have become popular due to cost, flexibility, lighter weight, and ease of integration, compared to wafer silicon cells.
Cadmium telluride solar cell
A cadmium telluride solar cell use a cadmium telluride (CdTe) thin film, a semiconductor layer to absorb and convert sunlight into electricity. Solarbuzz[20] has reported that the lowest quoted thin-film module price stands at US$1.76 per watt-peak, with the lowest crystalline silicon (c-Si) module at $2.48 per watt-peak.
The cadmium present in the cells would be toxic if released. However, release is impossible during normal operation of the cells and is unlikely during fires in residential roofs.[21] A square meter of CdTe contains approximately the same amount of Cd as a single C cell Nickel-cadmium battery, in a more stable and less soluble form.[21]
Copper-Indium Selenide
Copper indium gallium selenide (CIGS) is a direct-bandgap material. It has the highest efficiency (~20%) among thin film materials (see CIGS solar cells). Traditional methods of fabrication involve vacuum processes including co-evaporation and sputtering. Recent developments at IBM and Nanosolar have been targeting to lower the cost by using non-vacuum solution processes.
Gallium arsenide multijunction
High-efficiency multijunction cells were originally developed for special applications such as satellites and space exploration, but at present, their use in terrestrial concentrators might be the lowest cost alternative in terms of $/kWh and $/W.[22] These multijunction cells consist of multiple thin films produced using metalorganic vapour phase epitaxy. A triple-junction cell, for example, may consist of the semiconductors: GaAs, Ge, and GaInP2.[23] Each type of semiconductor will have a characteristic band gap energy which, loosely speaking, causes it to absorb light most efficiently at a certain color, or more precisely, to absorb electromagnetic radiation over a portion of the spectrum. The semiconductors are carefully chosen to absorb nearly all of the solar spectrum, thus generating electricity from as much of the solar energy as possible.
GaAs based multijunction devices are the most efficient solar cells to date, reaching a record high of 40.7% efficiency under "500-sun" solar concentration and laboratory conditions.[24] This was surpassed in October 2010 with a 42.3% triple junction metamorphic cell.[25]
This technology is currently being utilized in the Mars Exploration Rover missions which have run far past their 90 day design life.
Tandem solar cells based on monolithic, series connected, gallium indium phosphide (GaInP), gallium arsenide GaAs, and germanium Ge pn junctions, are seeing demand rapidly rise. In just the past 12 months (12/2006 - 12/2007), the cost of 4N gallium metal has risen from about $350 per kg to $680 per kg. Additionally, germanium metal prices have risen substantially to $1000–$1200 per kg this year. Those materials include gallium (4N, 6N and 7N Ga), arsenic (4N, 6N and 7N) and germanium, pyrolitic boron nitride (pBN) crucibles for growing crystals, and boron oxide, these products are critical to the entire substrate manufacturing industry.
Triple-junction GaAs solar cells were also being used as the power source of the Dutch four-time World Solar Challenge winners Nuna in 2003, 2005 and 2007, and also by the Dutch solar cars Solutra (2005), Twente One (2007) and 21Revolution (2009).
The Dutch Radboud University Nijmegen set the record for thin film solar cell efficiency using a single junction GaAs to 25.8% in August 2008 using only 4 µm thick GaAs layer which can be transferred from a wafer base to glass or plastic film.[26]
Light-absorbing dyes (DSSC)
Dye-sensitized solar cells (DSSCs) are made of low-cost materials and do not need elaborate equipment to manufacture, so they can be made in a DIY fashion, possibly allowing players to produce more of this type of solar cell than others. In bulk it should be significantly less expensive than older solid-state cell designs. DSSC's can be engineered into flexible sheets, and although its conversion efficiency is less than the best thin film cells, its price/performance ratio should be high enough to allow them to compete with fossil fuel electrical generation. The DSSC has been developed by Prof. Michael Grätzel in 1991 at the Swiss Federal Institute of Technology (EPFL) in Lausanne (CH).
Typically a ruthenium metalorganic dye (Ru-centered) is used as a monolayer of light-absorbing material. The dye-sensitized solar cell depends on a mesoporous layer of nanoparticulate titanium dioxide to greatly amplify the surface area (200–300 m2/g TiO2, as compared to approximately 10 m2/g of flat single crystal). The photogenerated electrons from the light absorbing dye are passed on to the n-type TiO2, and the holes are absorbed by an electrolyte on the other side of the dye. The circuit is completed by a redox couple in the electrolyte, which can be liquid or solid. This type of cell allows a more flexible use of materials, and is typically manufactured by screen printing and/or use of Ultrasonic Nozzles, with the potential for lower processing costs than those used for bulk solar cells. However, the dyes in these cells also suffer from degradation under heat and UV light, and the cell casing is difficult to seal due to the solvents used in assembly. In spite of the above, this is a popular emerging technology with some commercial impact forecast within this decade. The first commercial shipment of DSSC solar modules occurred in July 2009 from G24i Innovations (www.g24i.com).
Organic/polymer solar cells
Organic solar cells are a relatively novel technology, yet hold the promise of a substantial price reduction (over thin-film silicon) and a faster return on investment. These cells can be processed from solution, hence the possibility of a simple roll-to-roll printing process, leading to inexpensive, large scale production.
Organic solar cells and polymer solar cells are built from thin films (typically 100 nm) of organic semiconductors including polymers, such as polyphenylene vinylene and small-molecule compounds like copper phthalocyanine (a blue or green organic pigment) and carbon fullerenes and fullerene derivatives such as PCBM. Energy conversion efficiencies achieved to date using conductive polymers are low compared to inorganic materials. However, it improved quickly in the last few years and the highest NREL (National Renewable Energy Laboratory) certified efficiency has reached 6.77%.[27] In addition, these cells could be beneficial for some applications where mechanical flexibility and disposability are important.
These devices differ from inorganic semiconductor solar cells in that they do not rely on the large built-in electric field of a PN junction to separate the electrons and holes created when photons are absorbed. The active region of an organic device consists of two materials, one which acts as an electron donor and the other as an acceptor. When a photon is converted into an electron hole pair, typically in the donor material, the charges tend to remain bound in the form of an exciton, and are separated when the exciton diffuses to the donor-acceptor interface. The short exciton diffusion lengths of most polymer systems tend to limit the efficiency of such devices. Nanostructured interfaces, sometimes in the form of bulk heterojunctions, can improve performance.[28]
Silicon thin films
Silicon thin-film cells are mainly deposited by chemical vapor deposition (typically plasma-enhanced (PE-CVD)) from silane gas and hydrogen gas. Depending on the deposition parameters, this can yield:[29]
- Amorphous silicon (a-Si or a-Si:H)
- Protocrystalline silicon or
- Nanocrystalline silicon (nc-Si or nc-Si:H), also called microcrystalline silicon.
It has been found that protocrystalline silicon with a low volume fraction of nanocrystalline silicon is optimal for high open circuit voltage.[30] These types of silicon present dangling and twisted bonds, which results in deep defects (energy levels in the bandgap) as well as deformation of the valence and conduction bands (band tails). The solar cells made from these materials tend to have lower energy conversion efficiency than bulk silicon, but are also less expensive to produce. The quantum efficiency of thin film solar cells is also lower due to reduced number of collected charge carriers per incident photon.
An amorphous silicon (a-Si) solar cell is made of amorphous or microcrystalline silicon and its basic electronic structure is the p-i-n junction. As the amorphous structure has a higher absorption rate of light than crystalline cells, the complete light spectrum can be absorbed with a very thin layer of photo-electrically active material. A film only 1 micron thick can absorb 90% of the usable solar energy.[31] The production of a-Si thin film solar cells uses glass as a substrate and deposits a very thin layer of silicon by plasma-enhanced chemical vapor deposition (PECVD). A-Si manufacturers are working towards lower costs per watt and higher conversion efficiency with continuous research and development on Multijunction solar cells for solar panels. Anwell Technologies Limited recently announced its target for multi-substrate-multi-chamber PECVD, to lower the cost to USD0.5 per watt.[32]
Amorphous silicon has a higher bandgap (1.7 eV) than crystalline silicon (c-Si) (1.1 eV), which means it absorbs the visible part of the solar spectrum more strongly than the infrared portion of the spectrum. As nc-Si has about the same bandgap as c-Si, the nc-Si and a-Si can advantageously be combined in thin layers, creating a layered cell called a tandem cell. The top cell in a-Si absorbs the visible light and leaves the infrared part of the spectrum for the bottom cell in nc-Si.
Recently, solutions to overcome the limitations of thin-film crystalline silicon have been developed. Light trapping schemes where the weakly absorbed long wavelength light is obliquely coupled into the silicon and traverses the film several times can significantly enhance the absorption of sunlight in the thin silicon films.[33] Thermal processing techniques can significantly enhance the crystal quality of the silicon and thereby lead to higher efficiencies of the final solar cells.[34]
Manufacture
Because solar cells are semiconductor devices, they share many of the same processing and manufacturing techniques as other semiconductor devices such as computer and memory chips. However, the stringent requirements for cleanliness and quality control of semiconductor fabrication are a little more relaxed for solar cells. Most large-scale commercial solar cell factories today make screen printed poly-crystalline silicon solar cells. Single crystalline wafers which are used in the semiconductor industry can be made into excellent high efficiency solar cells, but they are generally considered to be too expensive for large-scale mass production.
Poly-crystalline silicon wafers are made by wire-sawing block-cast silicon ingots into very thin (180 to 350 micrometer) slices or wafers. The wafers are usually lightly p-type doped. To make a solar cell from the wafer, a surface diffusion of n-type dopants is performed on the front side of the wafer. This forms a p-n junction a few hundred nanometers below the surface.
Antireflection coatings, to increase the amount of light coupled into the solar cell, are typically next applied. Silicon nitride has gradually replaced titanium dioxide as the antireflection coating because of its excellent surface passivation qualities. It prevents carrier recombination at the surface of the solar cell. It is typically applied in a layer several hundred nanometers thick using plasma-enhanced chemical vapor deposition (PECVD). Some solar cells have textured front surfaces that, like antireflection coatings, serve to increase the amount of light coupled into the cell. Such surfaces can usually only be formed on single-crystal silicon, though in recent years methods of forming them on multicrystalline silicon have been developed.
The wafer then has a full area metal contact made on the back surface, and a grid-like metal contact made up of fine "fingers" and larger "busbars" are screen-printed onto the front surface using a silver paste. The rear contact is also formed by screen-printing a metal paste, typically aluminium. Usually this contact covers the entire rear side of the cell, though in some cell designs it is printed in a grid pattern. The paste is then fired at several hundred degrees celsius to form metal electrodes in ohmic contact with the silicon. Some companies use an additional electro-plating step to increase the cell efficiency. After the metal contacts are made, the solar cells are interconnected in series (and/or parallel) by flat wires or metal ribbons, and assembled into modules or "solar panels". Solar panels have a sheet of tempered glass on the front, and a polymer encapsulation on the back.
Lifespan
Most commercially available solar cells are capable of producing electricity for at least twenty years without a significant decrease in efficiency. The typical warranty given by panel manufacturers is for a period of 25 – 30 years, wherein the output shall not fall below a specified percentage (around 80%) of the rated capacity.[35]
Research topics
There are currently many research groups active in the field of photovoltaics in universities and research institutions around the world. This research can be divided into three areas: making current technology solar cells cheaper and/or more efficient to effectively compete with other energy sources; developing new technologies based on new solar cell architectural designs; and developing new materials to serve as light absorbers and charge carriers.
Manufacturers and certification
National Renewable Energy Laboratory tests and validates solar technologies. There are three reliable certifications of solar equipment: UL and IEEE (both U.S. standards) and IEC.
Solar cells are manufactured primarily in Japan, Germany, Mainland China, Taiwan and United States,[36] though numerous other nations have or are acquiring significant solar cell production capacity. While technologies are constantly evolving toward higher efficiencies, the most effective cells for low cost electrical production are not necessarily those with the highest efficiency, but those with a balance between low-cost production and efficiency high enough to minimize area-related balance of systems cost. Those companies with large scale manufacturing technology for coating inexpensive substrates may, in fact, ultimately be the lowest cost net electricity producers, even with cell efficiencies that are lower than those of single-crystal technologies.
China
Backed by Chinese government's unprecedented plan to offer subsidies for utility-scale solar power projects that is likely to spark a new round of investment from Chinese solar panel makers. Chinese companies have already played a more important role in solar panels manufacturing in recent years. China produced solar cells/modules with an output of 1,180 MW in 2007 making it the largest producer in the world, according to statistics from China Photovoltaic Association.[37] Some Chinese companies such as Suntech Power, Yingli, LDK Solar Co, JA Solar and ReneSola have already announced projects in cooperation with regional governments with hundreds of megawatts each after the ‘Golden Sun’ incentive program was announced by the government.[38] The new development of solar module manufacturers with thin-film technology such as Veeco and Anwell Technologies Limited will further help to boost the domestic solar industry.[39][40]
United States
This section needs additional citations for verification. (August 2010) |
New manufacturing facilities for solar cells and modules in Massachusetts, Michigan, New York, Ohio, Oregon, and Texas promise to add enough capacity to produce thousands of megawatts of solar devices per year within the next few years from 2008.[41]
In late September 2008, Sanyo Electric Company, Ltd. announced its decision to build a manufacturing plant for solar ingots and wafers in Salem, Oregon. The plant will begin operating in October 2009 and will reach its full production capacity of 70 megawatts (MW) of solar wafers per year by April 2010.
In early October 2008, First Solar, Inc. broke ground on an expansion of its Perrysburg, Ohio, facility that will add enough capacity to produce another 57 MW per year of solar modules at the facility, bringing its total capacity to roughly 192 MW per year. The company expects to complete construction early next year and reach full production by mid-2010.
In mid-October 2008, SolarWorld AG opened a manufacturing plant in Hillsboro, Oregon, that is expected to produce 500 MW of solar cells per year when it reaches full production in 2011.
In March 2010, SpectraWatt, Inc. began production at its manufacturing plant in Hopewell Junction, NY, which is expected to produce 120 MW of solar cells per year when it reaches full production in 2011.
See also
{{Top}} may refer to:
- {{Collapse top}}
- {{Archive top}}
- {{Hidden archive top}}
- {{Afd top}}
- {{Discussion top}}
- {{Tfd top}}
- {{Top icon}}
- {{Top text}}
- {{Cfd top}}
- {{Rfd top}}
- {{Skip to top}}
{{Template disambiguation}} should never be transcluded in the main namespace.
- Multijunction solar cell
- Thin film solar cell
- Anomalous photovoltaic effect
- Autonomous building
- Black silicon
- Energy development
- Flexible substrate
- Green technology
- Helianthos
- Junction
- List of solar cell manufacturers
- List of Dye Solar Cell (DSC) manufacturers
- Metallurgical grade silicon
- Microgeneration
- Maximum Power Point Tracking
- Nanoflake
- Carbon Nanotubes in photovoltaics
- Photovoltaics
- P-N junction
- Plasmonic solar cell
- Printed electronics
- Quantum efficiency
- Quantum efficiency of a solar cell
- Renewable energy
- Roll-to-roll processing
- Shockley-Queisser limit
- Solar roof
- Solar shingles
- Solar tracker
- Spectrophotometry
- Silicon thin-film cell
- Thermophotovoltaics
References
- ^ "Photovoltaic infrared detectors". Retrieved 2009-10-31.
- ^ Alfred Smee (1849). Elements of Electro-Biology, or The Voltaic Mechanism of Man; of Electro-Pathology, Especially of the Nervous System... London: Longman, Brown, Green, and Longmans.
- ^ "Light sensitive device" U.S. patent 2,402,662
- ^ K. A. Tsokos, Physics for the IB Diploma Fifth edition, Cambridge University Press, Cambridge, 2008
- ^ Perlin, John (2004). "The Silicon Solar Cell Turns 50" (PDF). National Renewable Energy Laboratory. Retrieved 5 October 2010.
- ^ A textbook of Engineering Physics by Navneet Gupta & S. K. Tiwari
- ^ http://www.solarfeedintariff.net
- ^ N. Gupta, G. F. Alapatt, R. Podila, R. Singh, K.F. Poole, (2009). "Prospects of Nanostructure-Based Solar Cells for Manufacturing Future Generations of Photovoltaic Modules". International Journal of Photoenergy. 2009: 1. doi:10.1155/2009/154059.
{{cite journal}}: CS1 maint: extra punctuation (link) CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Going for grid parity 2005 article
- ^ Gaining on the grid
- ^ The Path to Grid Parity (Graphic)
- ^ Wynn, Gerard (2007-10-19). "Solar power edges towards boom time". Reuters. Retrieved 2009-07-29.
- ^ Solar rally, The Economist, 28 August 2008
- ^ Mark Z. Jacobson (2009). Review of Solutions to Global Warming, Air Pollution, and Energy Security p. 4.
- ^ "String ribbon silicon solar cells with 17.8% efficiency" (PDF).
- ^ Charting a Path to Low-Cost Solar
- ^ Organic solar cells
- ^ PVSC 33 Conference Site
- ^ Katherine Derbyshire (January 9, 2009). "Wafer-based Solar Cells Aren't Done Yet".
- ^ http://www.solarbuzz.com
- ^ a b Fthenakis, Vasilis M. (August 2004). "Life cycle impact analysis of cadmium in CdTe PV production" (PDF). Renewable and Sustainable Energy Reviews. 8: 303–334. doi:10.1016/j.rser.2003.12.001.
- ^ R. M. Swanson, "The Promise of Concentrators," Progress in Photovoltaics: Res. Appl. 8, pp. 93-111 (2000).
- ^ http://www.spectrolab.com/DataSheets/TerCel/tercell.pdf
- ^ Spectrolab - Frequently Asked Questions
- ^ [1]
- ^ New world record for Nijmegen solar cell (dutch)
- ^ http://www.foxbusiness.com/story/markets/industries/finance/solarmer-breaks-world-records-plastic-solar-technology/ Fox Business News
- ^ Mayer, A; et al. (2007). "Polymer-based solar cells". Materials Today. 10 (11): 28. doi:10.1016/S1369-7021(07)70276-6.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ R.W. Collins, A.S. Ferlauto, G.M. Ferreira, C. Chen, J. Koh, R.J. Koval, Y. Lee, J.M. Pearce, and C. R. Wronski, Evolution of microstructure and phase in amorphous, protocrystalline, and microcrystalline silicon studied by real time spectroscopic ellipsometry, Solar Energy Materials and Solar Cells, 78(1-4), pp. 143-180, 2003.
- ^ J. M. Pearce, N. Podraza, R. W. Collins, M.M. Al-Jassim, K.M. Jones, J. Deng, and C. R. Wronski (2007). "Optimization of Open-Circuit Voltage in Amorphous Silicon Solar Cells with Mixed Phase (Amorphous + Nanocrystalline) p-Type Contacts of Low Nanocrystalline Content" (PDF). Journal of Applied Physics. 101: 114301.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Photovoltaics".
- ^ "ANWELL produces its first solar panel". NextInsight. 2009-09-01.
- ^ P. I. Widenborg and A. G. Aberle, "Polycrystalline silicon thin-film solar cells on AIT-textured glass superstrates," Advances in OptoElectronics, vol. 2007, September 2007.
- ^ M. L. Terry, A. Straub, D. Inns, D. Y. Song, and A. G. Aberle, "Large open-circuit voltage improvement by rapid thermal annealing of evaporated solid-phase-crystallized thin-film silicon solar cells on glass," Applied Physics Letters, vol. 86, p. 3, April 2005.
- ^ "Photovoltaic Systems". toolbase.org. Retrieved November 11, 2010.
- ^ Solar Cell Production Jumps 50 Percent in 2007
- ^ "High Growth Reported for the Global Photovoltaic Industry". Reuters. 2009-03-10.
- ^ "First Solar's Gift to China: How to Build a Solar Farm". GreentechMedia. 2009-09-10.
- ^ "Veeco Make Plans to be First CIGS Thin Film Solar Manufacturer in China". AZoNetwork. 2009-09-10.
- ^ "Boost domestic solar industry". China daily. 2009-09-10.
- ^ EERE News: EERE Network News
External links
- Howstuffworks.com: How Solar Cells Work
- Photovoltaics CDROM online
- Battery is our business
- Solar cell manufacturing techniques
- Template:Dmoz
- Solar Energy Laboratory at University of Southampton
- NASA's Photovoltaic Info
- Nanotechnology Based Improvements for Solar Cells
- Chinese Firms Developing Solar Power Plants for Less Than 1 Yuan per kWh? ($0.15 per kWh)



