20α-Dihydroprogesterone: Difference between revisions
Appearance
Content deleted Content added
No edit summary |
Lncallahan (talk | contribs) m added unii |
||
| Line 16: | Line 16: | ||
| CASNo = 145-14-2 |
| CASNo = 145-14-2 |
||
| PubChem = 8956 |
| PubChem = 8956 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = 45630A64AC |
|||
| EINECS = 205-649-5 |
| EINECS = 205-649-5 |
||
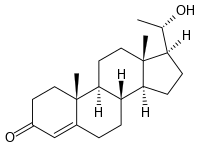
| SMILES = O=C4\C=C2/[C@]([C@H]1CC[C@@]3([C@H](CC[C@H]3[C@@H]1CC2)[C@H](O)C)C)(C)CC4 |
| SMILES = O=C4\C=C2/[C@]([C@H]1CC[C@@]3([C@H](CC[C@H]3[C@@H]1CC2)[C@H](O)C)C)(C)CC4 |
||
Revision as of 00:22, 14 July 2016
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
| MeSH | 20-alpha-Dihydroprogesterone |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H32O2 | |
| Molar mass | 316.478 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
20α-Dihydroprogesterone, or 20α-hydroxyprogesterone, is a naturally-occurring, endogenous progestogen.[1][2][3] It is a metabolite of progesterone, converted by the 20α-hydroxysteroid dehydrogenases AKR1C1 and AKR1C3, and although still active as a progestogen,[4] is much less potent in comparison.[1][2][3]
See also
References
- ^ a b Beranič N, Gobec S, Rižner TL (2011). "Progestins as inhibitors of the human 20-ketosteroid reductases, AKR1C1 and AKR1C3". Chem. Biol. Interact. 191 (1–3): 227–33. doi:10.1016/j.cbi.2010.12.012. PMID 21182831.
- ^ a b Tony M. Plant; Anthony J. Zeleznik (15 November 2014). Knobil and Neill's Physiology of Reproduction: Two-Volume Set. Academic Press. pp. 1–. ISBN 978-0-12-397769-4.
- ^ a b Cynthia L. Darlington (27 April 2009). The Female Brain. CRC Press. pp. 4–. ISBN 978-1-4200-7745-2.
- ^ Marianne J. Legato (29 October 2009). Principles of Gender-Specific Medicine. Academic Press. pp. 617–. ISBN 978-0-08-092150-1.
