Chlorambucil
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Leukeran |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682899 |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hepatic |
| Elimination half-life | 1.5 hours |
| Excretion | N/A |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.603 |
| Chemical and physical data | |
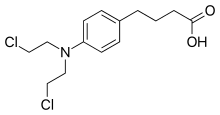
| Formula | C14H19Cl2NO2 |
| Molar mass | 304.212 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Chlorambucil (marketed as Leukeran by GlaxoSmithKline) is a chemotherapy drug that has been mainly used in the treatment of chronic lymphocytic leukemia. It is a nitrogen mustard alkylating agent[2] and can be given orally.
It is on the World Health Organization's List of Essential Medicines, a list of the most important medication needed in a basic health system.[3]
Medical uses
Chlorambucil's current use is mainly in chronic lymphocytic leukemia, as it is well tolerated by most patients, though chlorambucil has been largely replaced by fludarabine as first-line treatment in younger patients.[4] It can be used for treating some types of non-Hodgkin lymphoma, Waldenström macroglobulinemia, polycythemia vera, trophoblastic neoplasms, and ovarian carcinoma. Moreover, it also has been used as an immunosuppressive drug for various autoimmune and inflammatory conditions, such as nephrotic syndrome.
Side effects
Bone marrow suppression (anemia, neutropenia, thrombocytopenia) is the most commonly occurring side effect of chlorambucil. Withdrawn from the drug, this side effect is typically reversible. Like many alkylating agents, chlorambucil has been associated with the development of other forms of cancer.
Less commonly occurring side effects include:
- Gastrointestinal Distress (nausea, vomiting, diarrhea, and oral ulcerations).
- Central Nervous System: Seizures, tremors, muscular twitching, confusion, agitation, ataxia, and hallucinations.
- Skin reactions
- Hepatotoxicity
- Infertility
- Hair Loss
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ Takimoto CH, Calvo E. "Principles of Oncologic Pharmacotherapy" in Pazdur R, Wagman LD, Camphausen KA, Hoskins WJ (Eds) Cancer Management: A Multidisciplinary Approach. 11 ed. 2008.
- ^ "WHO Model List of EssentialMedicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ^ Rai KR, Peterson BL, Appelbaum FR, Kolitz J, Elias L, Shepherd L, Hines J, Threatte GA, Larson RA, Cheson BD, Schiffer CA (2000). "Fludarabine compared with chlorambucil as primary therapy for chronic lymphocytic leukemia". N Engl J Med. 343 (24): 1750–7. doi:10.1056/NEJM200012143432402. PMID 11114313.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
External links
- Leukeran (manufacturer's website)
- MedlinePlus's Drug Information
