Phosphofructokinase
| Phosphofructokinase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||
| Identifiers | |||||||||||
| Symbol | Ppfruckinase | ||||||||||
| Pfam | PF00365 | ||||||||||
| InterPro | IPR000023 | ||||||||||
| PROSITE | PDOC00336 | ||||||||||
| |||||||||||
Phosphofructokinase (PFK) is a kinase enzyme that phosphorylates fructose 6-phosphate in glycolysis.
Function[edit]
The enzyme-catalysed transfer of a phosphoryl group from ATP is an important reaction in a wide variety of biological processes.[1] Phosphofructokinase catalyses the phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate, a key regulatory step in the glycolytic pathway.[2][3] It is allosterically inhibited by ATP and allosterically activated by AMP, thus indicating the cell's energetic needs when it undergoes the glycolytic pathway.[4] PFK exists as a homotetramer in bacteria and mammals (where each monomer possesses 2 similar domains) and as an octomer in yeast (where there are 4 alpha- (PFK1) and 4 beta-chains (PFK2), the latter, like the mammalian monomers, possessing 2 similar domains[3]). This protein may use the morpheein model of allosteric regulation.[5]
PFK is about 300 amino acids in length, and structural studies of the bacterial enzyme have shown it comprises two similar (alpha/beta) lobes: one involved in ATP binding and the other housing both the substrate-binding site and the allosteric site (a regulatory binding site distinct from the active site, but that affects enzyme activity). The identical tetramer subunits adopt 2 different conformations: in a 'closed' state, the bound magnesium ion bridges the phosphoryl groups of the enzyme products (ADP and fructose-1,6-bisphosphate); and in an 'open' state, the magnesium ion binds only the ADP,[6] as the 2 products are now further apart. These conformations are thought to be successive stages of a reaction pathway that requires subunit closure to bring the 2 molecules sufficiently close to react.[6]
The reverse reaction is catalyzed by the enzyme Fructose-1,6-bisphosphatase.[citation needed]
Phosphofructokinase family[edit]
PFK belongs to the phosphofructokinase B (PfkB) family of sugar kinases.[7] Other members of this family (also known as the Ribokinase family) include ribokinase (RK), adenosine kinase (AK), inosine kinase, and 1-phosphofructokinase.[7][8][9] The members of the PfkB/RK family are identified by the presence of three conserved sequence motifs.[7][8][10] The structures of several PfK family of proteins have been determined from a number of organisms and the enzymatic activity of this family of protein shows a dependence on the presence of pentavalent ions.[11][7][10] PFK is found in isoform versions in skeletal muscle (PFKM), in the liver (PFKL), and from platelets (PFKP), allowing for tissue-specific expression and function. It is still speculated that the isoforms may play a role in specific glycolytic rates in the tissue-specific environments they are in. It has been found in humans that some human tumor cell lines had increased glycolytic productivity and correlated with the increased amount of PFKL.[12][13]
Clinical significance[edit]
Deficiency in PFK leads to glycogenosis type VII (Tarui's disease), an autosomal recessive disorder characterised by severe nausea, vomiting, muscle cramps and myoglobinuria in response to bursts of intense or vigorous exercise.[3] Sufferers are usually able to lead a reasonably ordinary life by learning to adjust activity levels.[3]
Regulation[edit]
There are two different phosphofructokinase enzymes in humans:
| Type | Synonyms | EC number | Substrate | Product | Paralog genes |
|---|---|---|---|---|---|
| Phosphofructokinase 1 | 6-phosphofructokinase phosphohexokinase |
EC 2.7.1.11 |  Fructose 6-phosphate |
 Fructose-1,6-bisphosphate |
PFKL, PFKM, PFKP |
| Phosphofructokinase 2 | 6-phosphofructo-2-kinase | EC 2.7.1.105 | 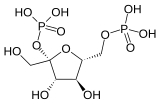 Fructose-2,6-bisphosphate |
PFKFB1, PFKFB2, PFKFB3, PFKFB4 |
See also[edit]
- Phosphofructokinase deficiency (GSD type VII, Tarui's disease)
References[edit]
- ^ Hellinga HW, Evans PR (1987). "Mutations in the active site of Escherichia coli phosphofructokinase". Nature. 327 (6121): 437–439. Bibcode:1987Natur.327..437H. doi:10.1038/327437a0. PMID 2953977. S2CID 4357039.
- ^ Wegener G, Krause U (April 2002). "Different modes of activating phosphofructokinase, a key regulatory enzyme of glycolysis, in working vertebrate muscle". Biochemical Society Transactions. 30 (2): 264–270. doi:10.1042/bst0300264. PMID 12023862.
- ^ a b c d Raben N, Exelbert R, Spiegel R, Sherman JB, Nakajima H, Plotz P, Heinisch J (January 1995). "Functional expression of human mutant phosphofructokinase in yeast: genetic defects in French Canadian and Swiss patients with phosphofructokinase deficiency". American Journal of Human Genetics. 56 (1): 131–141. PMC 1801305. PMID 7825568.
- ^ Garrett R, Grisham R (2012). Biochemistry. Cengage Learning. p. 585. ISBN 978-1133106296.
- ^ Selwood T, Jaffe EK (March 2012). "Dynamic dissociating homo-oligomers and the control of protein function". Archives of Biochemistry and Biophysics. 519 (2): 131–143. doi:10.1016/j.abb.2011.11.020. PMC 3298769. PMID 22182754.
- ^ a b Shirakihara Y, Evans PR (December 1988). "Crystal structure of the complex of phosphofructokinase from Escherichia coli with its reaction products". Journal of Molecular Biology. 204 (4): 973–994. doi:10.1016/0022-2836(88)90056-3. PMID 2975709.
- ^ a b c d Park J, Gupta RS (September 2008). "Adenosine kinase and ribokinase--the RK family of proteins". Cellular and Molecular Life Sciences. 65 (18): 2875–96. doi:10.1007/s00018-008-8123-1. PMID 18560757. S2CID 11439854.
- ^ a b Bork P, Sander C, Valencia A (January 1993). "Convergent evolution of similar enzymatic function on different protein folds: the hexokinase, ribokinase, and galactokinase families of sugar kinases". Protein Science. 2 (1): 31–40. doi:10.1002/pro.5560020104. PMC 2142297. PMID 8382990.
- ^ Spychala J, Datta NS, Takabayashi K, Datta M, Fox IH, Gribbin T, Mitchell BS (February 1996). "Cloning of human adenosine kinase cDNA: sequence similarity to microbial ribokinases and fructokinases". Proceedings of the National Academy of Sciences of the United States of America. 93 (3): 1232–7. Bibcode:1996PNAS...93.1232S. doi:10.1073/pnas.93.3.1232. PMC 40062. PMID 8577746.
- ^ a b Maj MC, Singh B, Gupta RS (March 2002). "Pentavalent ions dependency is a conserved property of adenosine kinase from diverse sources: identification of a novel motif implicated in phosphate and magnesium ion binding and substrate inhibition". Biochemistry. 41 (12): 4059–69. doi:10.1021/bi0119161. PMID 11900549.
- ^ Sigrell JA, Cameron AD, Jones TA, Mowbray SL (February 1998). "Structure of Escherichia coli ribokinase in complex with ribose and dinucleotide determined to 1.8 A resolution: insights into a new family of kinase structures". Structure. 6 (2): 183–93. doi:10.1016/s0969-2126(98)00020-3. PMID 9519409.
- ^ Sola-Penna M, Da Silva D, Coelho WS, Marinho-Carvalho MM, Zancan P (November 2010). "Regulation of mammalian muscle type 6-phosphofructo-1-kinase and its implication for the control of the metabolism". IUBMB Life. 62 (11): 791–796. doi:10.1002/iub.393. PMID 21117169.
- ^ Ausina P, Da Silva D, Majerowicz D, Zancan P, Sola-Penna M (July 2018). "Insulin specifically regulates expression of liver and muscle phosphofructokinase isoforms". Biomedicine & Pharmacotherapy. 103: 228–233. doi:10.1016/j.biopha.2018.04.033. PMID 29655163. S2CID 4874742.
External links[edit]
- Phosphofructokinases at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
