Midodrine
 | |
| Clinical data | |
|---|---|
| Trade names | Proamatine |
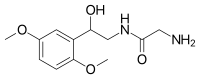
| Other names | 2-amino-N-[2-(2,5-dimethoxyphenyl)-2-hydroxy-ethyl]-acetamide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602023 |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.151.349 100.050.842, 100.151.349 |
| Chemical and physical data | |
| Formula | C12H18N2O4 |
| Molar mass | 254.282 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Midodrine (brand names Amatine, ProAmatine, Gutron) is a vasopressor/antihypotensive agent. Midodrine was approved in the United States by the Food and Drug Administration (FDA) in 1996 for the treatment of dysautonomia and orthostatic hypotension. In August 2010, the FDA proposed withdrawing this approval because the manufacturer, Shire plc, has failed to complete required studies after the medicine reached the market.[1][2]
In September 2010, the FDA reversed its decision to remove Midodrine from the market and has allowed it to remain available to patients while Shire plc collects further data regarding the efficacy and safety of the drug.[3] Shire plc announced on September 27, 2011 that it was continuing the process to work with the FDA towards a final approval of the drug.[4]
Chemical properties
Midodrine is an odorless, white, crystalline powder, soluble in water and sparingly soluble in methanol.
Mechanism of action
Midodrine is a prodrug which forms an active metabolite, desglymidodrine, which is an α1-receptor agonist and exerts its actions via activation of the alpha-adrenergic receptors of the arteriolar and venous vasculature, producing an increase in vascular tone and elevation of blood pressure. Desglymidodrine does not stimulate cardiac beta-adrenergic receptors. Desglymidodrine diffuses poorly across the blood–brain barrier, and is therefore not associated with effects on the central nervous system.

Metabolism
After oral administration, midodrine is rapidly absorbed. The plasma levels of the prodrug peak after about half an hour, and decline with a half-life of approximately 25 minutes, while the metabolite reaches peak blood concentrations about 1 to 2 hours after a dose of midodrine and has a half-life of about 3 to 4 hours. The absolute bioavailability of midodrine (measured as desglymidodrine) is 93%.
Indications
Midodrine hydrochloride tablets are indicated for the treatment of symptomatic orthostatic hypotension. In a critical care setting, it is widely used as an adjunctive therapy to weaning patients off of intravenous vasopressive medications. It has been suggested also as a treatment for chronic fatigue syndrome[5] as well as for hepato-pulmonary syndrome. It is also used with octreotide for hepatorenal syndrome; the proposed mechanism is constriction of splanchnic vessels and dilation of renal vasculature.
Contraindications
Midodrine is contraindicated in patients with severe organic heart disease, acute renal disease, urinary retention, pheochromocytoma or thyrotoxicosis. Midodrine should not be used in patients with persistent and excessive supine hypertension.
Side effects
Headache; feeling of pressure/fullness in the head, uncomfortable 'crawling' sensation on scalp and skin, vasodilation/flushing face, confusion/thinking abnormality, dry mouth; nervousness/anxiety and rash.[citation needed]
See also
References
- ^ U.S. proposes withdrawal of Shire hypotension drug, Aug 16, 2010.
- ^ O'Riordan, Michael. "FDA recommends withdrawal of midodrine". Food and Drug Administration. FDA proposes withdrawal of low blood pressure drug [press release]. August 16, 2010. TheHeart.org. Retrieved 1 April 2011..
- ^ Midodrine (ProAmatine, generic) Proposed Market Withdrawal – Update September 10, 2010.
- ^ Shire Announces Update on ProAmatime September 27, 2011.
- ^ Naschitz J, Dreyfuss D, Yeshurun D, Rosner I (April 2004). "Midodrine treatment for chronic fatigue syndrome". Postgrad Med J. 80 (942): 230–2. doi:10.1136/pgmj.2003.011429. PMC 1742969. PMID 15082846.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
External links
- Midodrine at drugs.com
