Amoxapine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | A-mox-a-peen[1] |
| Trade names | Asendin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682202 |
| License data | |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >60%[4] |
| Protein binding | 90%[5] |
| Metabolism | Hepatic (cytochrome P450 system)[4] |
| Elimination half-life | 8–10 hours (30 hours for chief active metabolite)[5] |
| Excretion | Renal (60%), feces (18%)[4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.034.411 |
| Chemical and physical data | |
| Formula | C17H16ClN3O |
| Molar mass | 313.79 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Amoxapine, sold under the brand name Asendin among others, is a tricyclic antidepressant (TCA). It is the N-demethylated metabolite of loxapine. Amoxapine first received marketing approval in the United States in 1980, approximately 10 to 20 years after most of the other TCAs were introduced in the United States.[6]
Medical uses
[edit]Amoxapine is used in the treatment of major depressive disorder. Compared to other antidepressants it is believed to have a faster onset of action, with therapeutic effects seen within four to seven days.[7][8] In excess of 80% of patients that do respond to amoxapine are reported to respond within two weeks of the beginning of treatment.[9] It also has properties similar to those of the atypical antipsychotics,[10][11][12] and may behave as one[13][14] and thus may be used in the treatment of schizophrenia off-label. Despite its apparent lack of extrapyramidal side effects in patients with schizophrenia it has been found to worsen motor function in a study of three patients with Parkinson's disease and psychosis.[15]
Contraindications
[edit]As with all FDA-approved antidepressants it carries a black-box warning about the potential of an increase in suicidal thoughts or behaviour in children, adolescents and young adults under the age of 25.[4] Its use is also advised against in individuals with known hypersensitivities to either amoxapine or other ingredients in its oral formulations.[4] Its use is also recommended against in the following disease states:[4]
- Severe cardiovascular disorders (potential of cardiotoxic adverse effects such as QT interval prolongation)
- Uncorrected narrow angle glaucoma
- Acute recovery post-myocardial infarction
Its use is also advised against in individuals concurrently on monoamine oxidase inhibitors or if they have been on one in the past 14 days and in individuals on drugs that are known to prolong the QT interval (e.g. ondansetron, citalopram, pimozide, sertindole, ziprasidone, haloperidol, chlorpromazine, thioridazine, etc.).[4]
Lactation
[edit]Its use in breastfeeding mothers not recommended as it is excreted in breast milk and the concentration found in breast milk is approximately a quarter that of the maternal serum level.[7][16]
Side effects
[edit]Adverse effects by incidence:[4][17]
Note: Serious (that is, those that can either result in permanent injury or are irreversible or are potentially life-threatening) are written in bold text.
Very common (>10% incidence) adverse effects include:
- Constipation
- Dry mouth
- Sedation
Common (1–10% incidence) adverse effects include:
- Anxiety
- Ataxia
- Blurred vision
- Confusion
- Dizziness
- Headache
- Fatigue
- Nausea
- Nervousness/restlessness
- Excessive appetite
- Rash
- Increased perspiration (sweating)
- Tremor
- Palpitations
- Nightmares
- Excitement
- Weakness
- ECG changes
- Oedema. An abnormal accumulation of fluids in the tissues of the body leading to swelling.
- Prolactin levels increased. Prolactin is a hormone that regulates the generation of breast milk. Prolactin elevation is not as significant as with risperidone or haloperidol.
Uncommon/Rare (<1% incidence) adverse effects include:
- Diarrhoea
- Flatulence
- Hypertension (high blood pressure)
- Hypotension (low blood pressure)
- Syncope (fainting)
- Tachycardia (high heart rate)
- Menstrual irregularity
- Disturbance of accommodation
- Mydriasis (pupil dilation)
- Orthostatic hypotension (a drop in blood pressure that occurs upon standing up)
- Seizure
- Urinary retention (being unable to pass urine)
- Urticaria (hives)
- Vomiting
- Nasal congestion
- Photosensitization
- Hypomania (a dangerously elated/irritable mood)
- Tingling
- Paresthesias of the extremities
- Tinnitus
- Disorientation
- Numbness
- Incoordination
- Disturbed concentration
- Epigastric distress
- Peculiar taste in the mouth
- Increased or decreased libido
- Impotence (difficulty achieving an erection)
- Painful ejaculation
- Lacrimation (crying without an emotional cause)
- Weight gain
- Altered liver function
- Breast enlargement
- Drug fever
- Pruritus (itchiness)
- Vasculitis a disorder where blood vessels are destroyed by inflammation. Can be life-threatening if it affects the right blood vessels.
- Galactorrhoea (lactation that is not associated with pregnancy or breast feeding)
- Delayed micturition (that is, delays in urination from when a conscious effort to urinate is made)
- Hyperthermia (elevation of body temperature; its seriousness depends on the extent of the hyperthermia)
- Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) this is basically when the body's level of the hormone, antidiuretic hormone, which regulates the conservation of water and the restriction of blood vessels, is elevated. This is potentially fatal as it can cause electrolyte abnormalities including hyponatraemia (low blood sodium), hypokalaemia (low blood potassium) and hypocalcaemia (low blood calcium) which can be life-threatening.
- Agranulocytosis a drop in white blood cell counts. The white blood cells are the cells of the immune system that fight off foreign invaders. Hence agranulocytosis leaves an individual open to life-threatening infections.
- Leukopaenia the same as agranulocytosis but less severe.
- Neuroleptic malignant syndrome (a potentially fatal reaction to antidopaminergic agents, most often antipsychotics. It is characterised by hyperthermia, diarrhoea, tachycardia, mental status changes [e.g. confusion], rigidity, extrapyramidal side effects)
- Tardive dyskinesia a most often irreversible neurologic reaction to antidopaminergic treatment, characterised by involuntary movements of facial muscles, tongue, lips, and other muscles. It develops most often only after prolonged (months, years or even decades) exposure to antidopaminergics.
- Extrapyramidal side effects. Motor symptoms such as tremor, parkinsonism, involuntary movements, reduced ability to move one's voluntary muscles, etc.
Unknown incidence or relationship to drug treatment adverse effects include:
- Paralytic ileus (paralysed bowel)
- Atrial arrhythmias including atrial fibrillation
- Myocardial infarction (heart attack)
- Stroke
- Heart block
- Hallucinations
- Purpura
- Petechiae
- Parotid swelling
- Changes in blood glucose levels
- Pancreatitis swelling of the pancreas
- Hepatitis swelling of the liver
- Urinary frequency
- Testicular swelling
- Anorexia (weight loss)
- Alopecia (hair loss)
- Thrombocytopenia a significant drop in platelet count that leaves one open to life-threatening bleeds.
- Eosinophilia an elevated level of the eosinophils of the body. Eosinophils are the type of immune cell that's job is to fight off parasitic invaders.
- Jaundice yellowing of the skin, eyes and mucous membranes due to an impaired ability of the body to clear the by product of haem breakdown, bilirubin, most often the result of liver damage as it is the liver's responsibility to clear bilirubin.
It tends to produce less anticholinergic effects, sedation and weight gain than some of the earlier TCAs (e.g. amitriptyline, clomipramine, doxepin, imipramine, trimipramine).[18] It may also be less cardiotoxic than its predecessors.[19]
Overdose
[edit]It is considered particularly toxic in overdose,[20] with a high rate of renal failure (which usually takes 2–5 days), rhabdomyolysis, coma, seizures and even status epilepticus.[19] Some believe it to be less cardiotoxic than other TCAs in overdose, although reports of cardiotoxic overdoses have been made.[7][17]
Pharmacology
[edit]Pharmacodynamics
[edit]| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | 58 | Human | [22] |
| NET | 16 | Human | [22] |
| DAT | 4,310 | Human | [22] |
| 5-HT2A | 0.5 | Human | [23] |
| 5-HT2C | 2.0 | Monkey | [24] |
| 5-HT6 | 6.0–50 | Human | [24][25] |
| 5-HT7 | 41 | Monkey | [24] |
| α1 | 50 | Human | [26] |
| α2 | 2,600 | Human | [26] |
| D2 | 3.6–160 | Human | [27][23][26] |
| D3 | 11 | Human | [23] |
| D4 | 2.0–40 | Human | [23] |
| H1 | 7.9–25 | Human | [28][26] |
| H2 | ND | ND | ND |
| H3 | >100,000 | Human | [28] |
| H4 | 6,310 | Human | [28] |
| mACh | 1,000 | Human | [26] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
Amoxapine possesses a wide array of pharmacological effects. It is a moderate and strong reuptake inhibitor of serotonin and norepinephrine, respectively,[22] and binds to the 5-HT2A,[29] 5-HT2B,[30] 5-HT2C,[29] 5-HT3,[31] 5-HT6,[24] 5-HT7,[24] D2,[26] α1-adrenergic,[26] D3,[27] D4,[27] and H1 receptors[26] with varying but significant affinity, where it acts as an antagonist (or inverse agonist depending on the receptor in question) at all sites. It has weak but negligible affinity for the dopamine transporter and the 5-HT1A,[31] 5-HT1B,[31] D1,[32] α2-adrenergic,[26] H4,[33] mACh,[26] and GABAA receptors,[32] and no affinity for the β-adrenergic receptors or the allosteric benzodiazepine site on the GABAA receptor.[32] Amoxapine is also a weak GlyT2 blocker,[34] as well as a weak (Ki = 2.5 μM, EC50 = 0.98 μM) δ-opioid receptor partial agonist.[35]
7-Hydroxyamoxapine, a major active metabolite of amoxapine, is a more potent dopamine receptor antagonist and contributes to its neuroleptic efficacy,[10] whereas 8-Hydroxyamoxapine is a norepinephrine reuptake inhibitor but a stronger serotonin reuptake inhibitor and helps to balance amoxapine's ratio of serotonin to norepinephrine transporter blockade.[36]
Pharmacokinetics
[edit]Amoxapine is metabolised into two main active metabolites: 7-hydroxyamoxapine and 8-hydroxyamoxapine.[37]
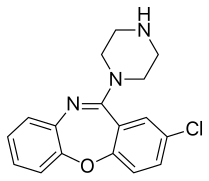 |
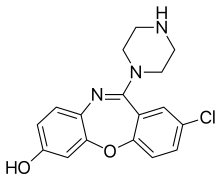 |
 |
| Compound[37][38][39] | t1/2 (hr)[40] | tmax (hr) | CSS (ng/mL) | Protein binding[4] | Vd[4] | Excretion[4] |
|---|---|---|---|---|---|---|
| Amoxapine | 8 | 1-2 | 17-93 ng/mL (divided dosing), 13-209 ng/mL (single daily dosing) | 90% | 0.9-1.2 L/kg | Urine (60%), feces (18%) |
| 8-hydroxyamoxapine | 30 | 5.3 (single dosing) | 158-512 ng/mL (divided dosing), 143-593 ng/mL (single dose) | ? | ? | ? |
| 7-hydroxyamoxapine | 6.5 | 2.6-5.4 (single dosing) | ? | ? | ? | ? |
Where:
- t1/2 is the elimination half life of the compound.
- tmax is the time to peak plasma levels after oral administration of amoxapine.
- CSS is the steady state plasma concentration.
- protein binding is the extent of plasma protein binding.
- Vd is the volume of distribution of the compound.
Society and culture
[edit]Brand names
[edit]Brand names for amoxapine include (where † denotes discontinued brands):[7][41]
- Adisen (KR)
- Amolife (IN)
- Amoxan (JP)
- Asendin† (previously marketed in CA, NZ, US)
- Asendis† (previously marketed in IE, UK)
- Défanyl (FR)
- Demolox (DK†, IN, ES†)
- Oxamine (IN)
- Oxcap (IN)
See also
[edit]References
[edit]- ^ "Amoxapine: Indications, Side Effects, Warnings -Drugs.com". Drugs.com. 6 November 2013. Retrieved 26 November 2013.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d e f g h i j k "Asendin, (amoxapine) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 26 November 2013.
- ^ a b Kinney JL, Evans RL (September–October 1982). "Evaluation of amoxapine". Clinical Pharmacy. 1 (5): 417–424. PMID 6764165.
- ^ National Center for Drugs and Biologics (U.S.) (1984). Approved Prescription Drug Products with Therapeutic Equivalence Evaluations (5th, Cumulative Supplement 3 ed.). Rockville, MD: U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration. p. IV-9.
- ^ a b c d Amoxapine. London, UK: Pharmaceutical Press. 30 January 2013. Retrieved 26 November 2013.
{{cite book}}:|work=ignored (help) - ^ Ban TA, Fujimori M, Petrie WM, Ragheb M, Wilson WH (1982). "Systematic studies with amoxapine, a new antidepressant". International Pharmacopsychiatry. 17 (1): 18–27. doi:10.1159/000468553. PMID 7045016.
- ^ Product Information: Asendin(R), amoxapine tablets. Physicians' Desk Reference (electronic version), MICROMEDEX, Inc, Englewood, CO, USA, 1999.
- ^ a b Cohen BM, Harris PQ, Altesman RI, Cole JO (September 1982). "Amoxapine: neuroleptic as well as antidepressant?". The American Journal of Psychiatry. 139 (9): 1165–1167. doi:10.1176/ajp.139.9.1165. PMID 6126130.
- ^ Kapur S, Cho R, Jones C, McKay G, Zipursky RB (May 1999). "Is amoxapine an atypical antipsychotic? Positron-emission tomography investigation of its dopamine2 and serotonin2 occupancy". Biological Psychiatry. 45 (9): 1217–1220. doi:10.1016/S0006-3223(98)00204-2. PMID 10331115. S2CID 21407817.
- ^ Wadenberg MG, Sills TL, Fletcher PJ, Kapur S (April 2000). "Antipsychoticlike effects of amoxapine, without catalepsy, using the prepulse inhibition of the acoustic startle reflex test in rats". Biological Psychiatry. 47 (7): 670–676. doi:10.1016/S0006-3223(99)00267-X. PMID 10745061. S2CID 27619436.
- ^ Apiquian R, Fresan A, Ulloa RE, de la Fuente-Sandoval C, Herrera-Estrella M, Vazquez A, et al. (December 2005). "Amoxapine as an atypical antipsychotic: a comparative study vs risperidone". Neuropsychopharmacology. 30 (12): 2236–2244. doi:10.1038/sj.npp.1300796. PMID 15956984.
- ^ Chaudhry IB, Husain N, Khan S, Badshah S, Deakin B, Kapur S (December 2007). "Amoxapine as an antipsychotic: comparative study versus haloperidol". Journal of Clinical Psychopharmacology. 27 (6): 575–581. doi:10.1097/jcp.0b013e31815a4424. PMID 18004123. S2CID 45880666.
- ^ Sa DS, Kapur S, Lang AE (July–August 2001). "Amoxapine shows an antipsychotic effect but worsens motor function in patients with Parkinson's disease and psychosis". Clinical Neuropharmacology. 24 (4): 242–244. doi:10.1097/00002826-200107000-00010. PMID 11479398.
- ^ Gelenberg AJ, Cooper DS, Doller JC, Maloof F (October 1979). "Galactorrhea and hyperprolactinemia associated with amoxapine therapy. Report of a case". JAMA. 242 (17): 1900–1901. doi:10.1001/jama.1979.03300170046029. PMID 573343.
- ^ a b "AMOXAPINE TABLET [WATSON LABORATORIES, INC.]". DailyMed. Watson Laboratories, Inc. July 2010. Retrieved 26 November 2013.
- ^ "Side effects of antidepressant medications". UpToDate. Wolters Kluwer Health. Retrieved 26 November 2013.
- ^ a b Walker, R; Whittlesea, C, eds. (2007) [1994]. Clinical Pharmacy and Therapeutics (4th ed.). Edinburgh: Churchill Livingstone Elsevier. ISBN 978-0-7020-4293-5.
- ^ White N, Litovitz T, Clancy C (December 2008). "Suicidal antidepressant overdoses: a comparative analysis by antidepressant type". Journal of Medical Toxicology. 4 (4): 238–250. doi:10.1007/BF03161207. PMC 3550116. PMID 19031375.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ a b c d Tatsumi M, Groshan K, Blakely RD, Richelson E (December 1997). "Pharmacological profile of antidepressants and related compounds at human monoamine transporters". European Journal of Pharmacology. 340 (2–3): 249–258. doi:10.1016/s0014-2999(97)01393-9. PMID 9537821.
- ^ a b c d Seeman P, Tallerico T (March 1998). "Antipsychotic drugs which elicit little or no parkinsonism bind more loosely than dopamine to brain D2 receptors, yet occupy high levels of these receptors". Molecular Psychiatry. 3 (2): 123–134. doi:10.1038/sj.mp.4000336. PMID 9577836.
- ^ a b c d e Roth BL, Craigo SC, Choudhary MS, Uluer A, Monsma FJ, Shen Y, et al. (March 1994). "Binding of typical and atypical antipsychotic agents to 5-hydroxytryptamine-6 and 5-hydroxytryptamine-7 receptors". The Journal of Pharmacology and Experimental Therapeutics. 268 (3): 1403–1410. PMID 7908055.
- ^ Kohen R, Metcalf MA, Khan N, Druck T, Huebner K, Lachowicz JE, et al. (January 1996). "Cloning, characterization, and chromosomal localization of a human 5-HT6 serotonin receptor". Journal of Neurochemistry. 66 (1): 47–56. doi:10.1046/j.1471-4159.1996.66010047.x. PMID 8522988. S2CID 35874409.
- ^ a b c d e f g h i j Richelson E, Nelson A (July 1984). "Antagonism by antidepressants of neurotransmitter receptors of normal human brain in vitro". The Journal of Pharmacology and Experimental Therapeutics. 230 (1): 94–102. PMID 6086881.
- ^ a b c Burstein ES, Ma J, Wong S, Gao Y, Pham E, Knapp AE, et al. (December 2005). "Intrinsic efficacy of antipsychotics at human D2, D3, and D4 dopamine receptors: identification of the clozapine metabolite N-desmethylclozapine as a D2/D3 partial agonist". The Journal of Pharmacology and Experimental Therapeutics. 315 (3): 1278–1287. doi:10.1124/jpet.105.092155. PMID 16135699. S2CID 2247093.
- ^ a b c Appl H, Holzammer T, Dove S, Haen E, Strasser A, Seifert R (February 2012). "Interactions of recombinant human histamine H₁R, H₂R, H₃R, and H₄R receptors with 34 antidepressants and antipsychotics". Naunyn-Schmiedeberg's Archives of Pharmacology. 385 (2): 145–170. doi:10.1007/s00210-011-0704-0. PMID 22033803. S2CID 253747520.
- ^ a b Pälvimäki EP, Roth BL, Majasuo H, Laakso A, Kuoppamäki M, Syvälahti E, Hietala J (August 1996). "Interactions of selective serotonin reuptake inhibitors with the serotonin 5-HT2c receptor". Psychopharmacology. 126 (3): 234–240. doi:10.1007/BF02246453. PMID 8876023. S2CID 24889381.
- ^ Glusa E, Pertz HH (June 2000). "Further evidence that 5-HT-induced relaxation of pig pulmonary artery is mediated by endothelial 5-HT(2B) receptors". British Journal of Pharmacology. 130 (3): 692–698. doi:10.1038/sj.bjp.0703341. PMC 1572101. PMID 10821800.
- ^ a b c Gozlan H, Saddiki-Traki F, Merahi N, Laguzzi R, Hamon M (December 1991). "[Preclinical pharmacology of amoxapine and amitriptyline. Implications of serotoninergic and opiodergic systems in their central effect in rats]". L'Encéphale (in French). 17 Spec No 3: 415–422. PMID 1666997.
- ^ a b c Wei HB, Niu XY (1990). "[Comparison of the affinities of amoxapine and loxapine for various receptors in rat brain and the receptor down-regulation after chronic administration]". Yao Xue Xue Bao = Acta Pharmaceutica Sinica (in Chinese). 25 (12): 881–885. PMID 1966571.
- ^ Lim HD, van Rijn RM, Ling P, Bakker RA, Thurmond RL, Leurs R (September 2005). "Evaluation of histamine H1-, H2-, and H3-receptor ligands at the human histamine H4 receptor: identification of 4-methylhistamine as the first potent and selective H4 receptor agonist". The Journal of Pharmacology and Experimental Therapeutics. 314 (3): 1310–1321. doi:10.1124/jpet.105.087965. PMID 15947036. S2CID 24248896.
- ^ Sitte H, Freissmuth M (2 August 2006). Neurotransmitter Transporters. Springer Science & Business Media. pp. 472–. ISBN 978-3-540-29784-0.
- ^ Onali P, Dedoni S, Olianas MC (January 2010). "Direct agonist activity of tricyclic antidepressants at distinct opioid receptor subtypes". The Journal of Pharmacology and Experimental Therapeutics. 332 (1): 255–265. doi:10.1124/jpet.109.159939. PMID 19828880. S2CID 18893305.
- ^ Midha KK, Hubbard JW, McKay G, Rawson MJ, Hsia D (September 1999). "The role of metabolites in a bioequivalence study II: amoxapine, 7-hydroxyamoxapine, and 8-hydroxyamoxapine". International Journal of Clinical Pharmacology and Therapeutics. 37 (9): 428–438. PMID 10507241.
- ^ a b Jue SG, Dawson GW, Brogden RN (July 1982). "Amoxapine: a review of its pharmacology and efficacy in depressed states". Drugs. 24 (1): 1–23. doi:10.2165/00003495-198224010-00001. PMID 7049659. S2CID 7279867.
- ^ Calvo B, García MJ, Pedraz JL, Mariño EL, Domínguez-Gil A (April 1985). "Pharmacokinetics of amoxapine and its active metabolites". International Journal of Clinical Pharmacology, Therapy, and Toxicology. 23 (4): 180–185. PMID 3997304.
- ^ Takeuchi H, Yokota S, Shimada S, Ohtani Y, Miura S, Kubo H (April 1993). "Pharmacokinetics of amoxapine and its active metabolites in healthy subjects". Current Therapeutic Research. 53 (4): 427–434. doi:10.1016/S0011-393X(05)80202-4.
- ^ "Amoxapine Monograph for Professionals - Drugs.com". Drugs.com. Bethesda, MD, USA: American Society of Health-System Pharmacists, Inc. 1 October 2007. Retrieved 30 November 2013.
- ^ "Amoxapine -Drugs.com". Drugs.com. 2013. Retrieved 26 November 2013.
- Alpha-1 blockers
- Atypical antipsychotics
- Chloroarenes
- Delta-opioid receptor agonists
- Dibenzoxazepines
- Dopamine antagonists
- Glycine reuptake inhibitors
- H1 receptor antagonists
- Human drug metabolites
- Muscarinic antagonists
- 1-Piperazinyl compounds
- Serotonin receptor antagonists
- Serotonin–norepinephrine reuptake inhibitors
- Tetracyclic antidepressants
- Tricyclic antidepressants
