Wikipedia talk:WikiProject Elements: Difference between revisions
m →References: new error messages: funny typo |
Parcly Taxel (talk | contribs) →References: new error messages: An update here. |
||
| Line 2,819: | Line 2,819: | ||
* Please change this overview table: set a page {{code|1=aq=B}} (to green) when you have checked & cleared any ref errors.<br/> |
* Please change this overview table: set a page {{code|1=aq=B}} (to green) when you have checked & cleared any ref errors.<br/> |
||
*''Note'': errors may be suppressed, depending on your skin setting. See [[Help:CS1_errors#Controlling_error_message_display]]. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 15:08, 21 April 2013 (UTC) |
*''Note'': errors may be suppressed, depending on your skin setting. See [[Help:CS1_errors#Controlling_error_message_display]]. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 15:08, 21 April 2013 (UTC) |
||
* |
* blue: see below |
||
{|cellpadding="0" cellspacing="0" style="font-size:90%; line-height:120%; background:{{Element color/Table|background}}; border:1px solid {{Element color/Table|border}}; padding:0; width:100%; {{{style|{{{1|}}}}}}" | Articles checked for reference errors |
{|cellpadding="0" cellspacing="0" style="font-size:90%; line-height:120%; background:{{Element color/Table|background}}; border:1px solid {{Element color/Table|border}}; padding:0; width:100%; {{{style|{{{1|}}}}}}" | Articles checked for reference errors |
||
|- |
|- |
||
| Line 2,881: | Line 2,881: | ||
|- |
|- |
||
! {{element cell AQ||Period 4 element|4|aq=B}} |
! {{element cell AQ||Period 4 element|4|aq=B}} |
||
| {{element cell AQ|19|potassium |K |aq= |
| {{element cell AQ|19|potassium |K |aq=FA}} |
||
| {{element cell AQ|20|calcium |Ca|aq=B}} |
| {{element cell AQ|20|calcium |Ca|aq=B}} |
||
| {{element cell AQ|21|scandium |Sc|aq=B}} |
| {{element cell AQ|21|scandium |Sc|aq=B}} |
||
| Line 2,901: | Line 2,901: | ||
|- |
|- |
||
! {{element cell AQ||Period 5 element|5|aq=B}} |
! {{element cell AQ||Period 5 element|5|aq=B}} |
||
| {{element cell AQ| |
| {{element cell AQ|37|rubidium |Rb|aq=FA}} |
||
| {{element cell AQ|38|strontium |Sr|aq=start}} |
| {{element cell AQ|38|strontium |Sr|aq=start}} |
||
| {{element cell AQ|39|yttrium |Y |aq=B}} |
| {{element cell AQ|39|yttrium |Y |aq=B}} |
||
| Line 2,925: | Line 2,925: | ||
| title="Lanthanides (see below)" style="text-align:center;" | * |
| title="Lanthanides (see below)" style="text-align:center;" | * |
||
| {{element cell AQ|72|hafnium |Hf|aq=B}} |
| {{element cell AQ|72|hafnium |Hf|aq=B}} |
||
| {{element cell AQ|73|tantalum |Ta|aq= |
| {{element cell AQ|73|tantalum |Ta|aq=B}} |
||
| {{element cell AQ|74|tungsten |W |aq=B}} |
| {{element cell AQ|74|tungsten |W |aq=B}} |
||
| {{element cell AQ|75|rhenium |Re|aq= |
| {{element cell AQ|75|rhenium |Re|aq=B}} |
||
| {{element cell AQ|76|osmium |Os|aq= |
| {{element cell AQ|76|osmium |Os|aq=B}} |
||
| {{element cell AQ|77|iridium |Ir|aq=B}} |
| {{element cell AQ|77|iridium |Ir|aq=B}} |
||
| {{element cell AQ|78|platinum |Pt|aq=B}} |
| {{element cell AQ|78|platinum |Pt|aq=B}} |
||
| {{element cell AQ|79|gold |Au|aq=B}} |
| {{element cell AQ|79|gold |Au|aq=B}} |
||
| {{element cell AQ|80|mercury (element)|Hg|aq=B}} |
| {{element cell AQ|80|mercury (element)|Hg|aq=B}} |
||
| {{element cell AQ|81|thallium |Tl|aq= |
| {{element cell AQ|81|thallium |Tl|aq=B}} |
||
| {{element cell AQ|82|lead |Pb|aq= |
| {{element cell AQ|82|lead |Pb|aq=B}} |
||
| {{element cell AQ|83|bismuth |Bi|aq=start}} |
| {{element cell AQ|83|bismuth |Bi|aq=start}} |
||
| {{element cell AQ|84|polonium |Po|aq=start}} |
| {{element cell AQ|84|polonium |Po|aq=start}} |
||
| Line 3,060: | Line 3,060: | ||
I must say, I liked it better in the past when the code was more forgiving. [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 10:12, 22 April 2013 (UTC) |
I must say, I liked it better in the past when the code was more forgiving. [[User:Double sharp|Double sharp]] ([[User talk:Double sharp|talk]]) 10:12, 22 April 2013 (UTC) |
||
:These errors mostly are ambiguous info, if not plain wrong. Esp. for outside connections (doi, authors, COinS, ISBN, ...) these corrections are an improvement. And solving them helps me understand the cite-templates & how to cite correctly. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 12:41, 22 April 2013 (UTC) |
:These errors mostly are ambiguous info, if not plain wrong. Esp. for outside connections (doi, authors, COinS, ISBN, ...) these corrections are an improvement. And solving them helps me understand the cite-templates & how to cite correctly. -[[User:DePiep|DePiep]] ([[User talk:DePiep|talk]]) 12:41, 22 April 2013 (UTC) |
||
Much of the sixth period has been fixed. For lead one of the errors was in a {{tl|cite doi}} template. [[Special:Contributions/Parcly Taxel|<span style="color:#6DC6FB;">Princess</span>]] [[User:Parcly Taxel|<span style="color:#961370;">Parcly</span>]] [[User talk:Parcly Taxel|<span style="color:#F3A4D9;">Taxel</span>]] 11:17, 24 April 2013 (UTC) |
|||
Revision as of 11:17, 24 April 2013
| Elements Project‑class | |||||||
| |||||||
| This is the talk page for discussing improvements to the WikiProject Elements page. |
|
| Archives: Index, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62Auto-archiving period: 60 days |
| Table of contents | Article alerts |
|---|---|
|
Redirects for discussion
Featured article candidates
Good article nominees
Good article reassessments
Requests for comments Articles for creation
|
| B | C | Start | Stub | List | Category | Disambig | Draft | File | Portal | Project | Redirect | Template | NA | ??? | Total | ||||
| 28 | 0 | 97 | 103 | 123 | 96 | 34 | 0 | 172 | 305 | 3 | 3 | 116 | 1 | 22 | 3,893 | 227 | 10 | 0 | 5,233 |
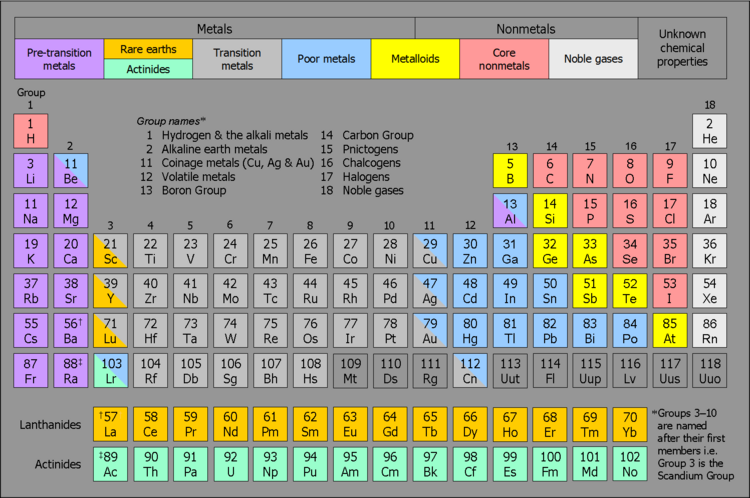
Metalloids
- (I'm aware that the title I originally gave this discussion is now quite misleading, as the discussion now covers a much wider scope than just the metalloids, but I won't change it as that would break quite a few links where I or someone else mentioned this discussion.) Double sharp (talk) 08:07, 7 December 2012 (UTC)
Polonium – metalloid or post-transition metal?
The polonium article states that "Due to its position in the periodic table, polonium is sometimes referred to as a metalloid, however others note that on the basis of its properties and behaviour it is 'unambiguously a metal.'" It has many more metallic properties than non-metallic ones (see Metalloid#Polonium). Shouldn't we be basing our treatment and classification of an element on the basis of its properties and behaviour and not its position in the periodic table? Double sharp (talk) 08:23, 26 June 2012 (UTC)
- Yes, certainly, especially since its position in the periodic table doesn't really imply it to be a metalloid IMO. The question of whether it is a metal "based on properties" was already discussed e.g. on Talk:Polonium#Metal_or_Metalloid, BTW. If the evidence is conclusive, I'd support changing polonium to "metal". (However, note that about half the recent sources in List of metalloid lists do consider it to be a metalloid.) --Roentgenium111 (talk) 12:06, 27 June 2012 (UTC)
- Po has only metallic allotropes, usually forms Po2+ in aqueous solution, requires very reducing conditions to form Po2− in aqueous solution, forms a weakly basic oxide (PoO2), has moderate electrical conductivity (like Bi), and forms many salts (see Template:Polonium compounds). However, its halides do resemble nonmetal halides in some ways, and many metal polonides (involving the Po2− anion) are known. However, only Na, the alkaline earth metals (except Ra), the lanthanides, and the group 12 elements (except Hg) are known to form ionic polonides; Hg, Pb, and Pt form intermetallic polonides. Double sharp (talk) 14:03, 27 June 2012 (UTC)
- Dunno, nonmetal properties don't convince me. Aurides are known, and none calls gold a metalloid. So are bismuthides. And even alkalides. The halides argument can probably be shown in analogy to aluminum. It is also a p-block thing, not only a metal/nonmetal thing, at least to some extent.--R8R Gtrs (talk) 19:27, 27 June 2012 (UTC)
- Yeah, I feel the same way too. I suspect the labelling of Po as a metalloid is because they won't let you play around as much with radioactive stuff now as you could get away with in the 60s and 70s. :-( So people don't know so much about its chemical properties and just give it a default metalloid status as it's next to the "metalloid line". (At, although also next to the metalloid line, isn't defaulted to being a metalloid as much, because it's also in group 17 and tends to instead be defaulted to being a halogen – it's in astatine and metalloid, and you probably know more than me about At anyway. ;-)) Double sharp (talk) 14:36, 28 June 2012 (UTC)
- Dunno, nonmetal properties don't convince me. Aurides are known, and none calls gold a metalloid. So are bismuthides. And even alkalides. The halides argument can probably be shown in analogy to aluminum. It is also a p-block thing, not only a metal/nonmetal thing, at least to some extent.--R8R Gtrs (talk) 19:27, 27 June 2012 (UTC)
- Po has only metallic allotropes, usually forms Po2+ in aqueous solution, requires very reducing conditions to form Po2− in aqueous solution, forms a weakly basic oxide (PoO2), has moderate electrical conductivity (like Bi), and forms many salts (see Template:Polonium compounds). However, its halides do resemble nonmetal halides in some ways, and many metal polonides (involving the Po2− anion) are known. However, only Na, the alkaline earth metals (except Ra), the lanthanides, and the group 12 elements (except Hg) are known to form ionic polonides; Hg, Pb, and Pt form intermetallic polonides. Double sharp (talk) 14:03, 27 June 2012 (UTC)
Consistent with the above observations, I struggle to find sufficient grounds for classifying polonium as a metalloid. It shows a few nonmetallic or intermediate properties but these are 'trumped' by its metallic properties, especially the combination of: metallic band structure; metallic conductivity; the presence of a polonium cation in mildly acidic aqueous solution; and the basicity of its oxide. Polonium would be better classified as a post-transition metal. As you note R8R, the nonmetallic properties of polonium are found in, for example, some of the transition metals yet these are not classified as metalloids.
Astatine suffers from having a relatively obscure chemistry. Per Double sharp, this means that it tends to inherit the default nonmetal status of its lighter halogen congeners. Apparently halogen membership trumps being next to the metal-nonmetal dividing line, artificial as the latter construct is. OTOH, reading selectively from the literature:
- Bresler (1952) comments that, 'the properties of the metal and the halogen are curiously combined in this element.'
- Edwards and Sienko (1983) speculate, on the basis of the Goldhammer-Herzfeld criterion for metallicity, that astatine is probably a 'semimetal' (i.e. a metalloid).
- Rössler (1985) highlights 'the chemical ambiguity of astatine between halogen and metal character.'
- Siekierski and Burgess (2002) contend or presume that astatine would be a metal if it could form a condensed phase—but do not give a specific basis for their claim.
Against this background, and noting...
- (1) its currently known and estimated properties
- (2) the transition from 'nonmetal-with-incipient-metallic-properties' (NWIMP) to metalloid, in the neighbouring elements selenium and tellurium; and
- (3) the NWIMP status of iodine,
...astatine is currently better classified as a metalloid. IMPs of iodine include: residual metallic luster; semi-conductivity (band gap = 1.35 ev); photoconductivity; electron delocalization within the layers of the solid iodine lattice; its metallic transformation under the application of relatively modest pressure; the metal-like electrical conductivity of the liquid form; the existence of the +1 iodine cation in pyridine solution and associated salts; and the polymeric structure of its most stable oxide, I2O5.
- Bresler SE 1952, Radioactive elements, 2nd ed., State Technical Theoretical Press, Moscow, in IM Korenman 1959, 'Regularities in properties of thallium', Journal of general chemistry of the USSR, English translation, Consultants Bureau, New York, vol. 29, no. 2, pp. 1366–1390 (1368)
- Edwards PP & Sienko MJ 1983, 'On the occurrence of metallic character in the periodic table of the elements,' Journal of Chemical Education, vol. 60, no. 9, pp. 691–696 (692)
- Rössler K 1985, ‘Handling of astatine’, in HK Kugler & C Keller (eds), Gmelin handbook of inorganic chemistry, At Astatine, 8th ed., Springer-Verlag, Berlin, pp. 140–156 (143)
- Siekierski S & Burgess J 2002, Concise chemistry of the elements, Horwood, Chichester, pp. 65, 122
Sandbh (talk) 07:31, 1 July 2012 (UTC)
- I would agree with colouring Po as a post-transition metal and At as a metalloid, but what do we do with the halogens category? For, after all, At is both a metalloid and a halogen! (The German compact periodic table template has a nice solution...) Double sharp (talk) 11:40, 2 July 2012 (UTC)
- Going through all the periodic table templates and changing Po to a post-transition metal everywhere. This is extremely patience-exhausting. (We then need to figure out what to do for At's metalloid status. The most drastic plan would be to get rid of the "halogens" category, but that would be too drastic for my tastes.) Double sharp (talk) 15:03, 2 July 2012 (UTC)
- Finally finished updating Po in all the templates. I might have missed a few, in which case you are free to fix it without telling me. Now we can talk about At. Double sharp (talk) 15:13, 2 July 2012 (UTC)
- Going through all the periodic table templates and changing Po to a post-transition metal everywhere. This is extremely patience-exhausting. (We then need to figure out what to do for At's metalloid status. The most drastic plan would be to get rid of the "halogens" category, but that would be too drastic for my tastes.) Double sharp (talk) 15:03, 2 July 2012 (UTC)
Astatine – metalloid or halogen?
I'm fairly convinced that At deserves to be called a metalloid, but that would clash with its (also relevant) halogen status. What do you propose? (Getting rid of the "halogens" category would solve the problem, but would be far too drastic for my tastes.) Double sharp (talk) 13:15, 3 July 2012 (UTC)
- If the current post-transition, metalloid and 'other nonmetal', categories can obliterate the pnictogen and chalcogen categories then I'm not sure if there is a need to treat the halogens differently (a preference is fine, but not the same as a need). For astatine, could it be shown as a metalloid but in the Element category box for the astatine article, change the name of the box to 'Element categories' and then enter two categories i.e. 'metalloid; halogen'? Same approach could work for e.g. gold: 'transition metal; noble metal; coinage metal;' or bismuth: 'post-transition metal; pnictogen'. — Preceding unsigned comment added by Sandbh (talk • contribs) 13:04, 4 July 2012 (UTC)
- Here's a halogen-less periodic table:
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
Double sharp (talk) 14:53, 4 July 2012 (UTC)
- The German Wikipedia solution has an image used as the background with both possible categorisations shown (alternating bands of the halogen and metalloid colour). This could be a useful idea for the compact table, but I'm not sure if it can work for any cell size. Double sharp (talk) 14:56, 4 July 2012 (UTC)
- Then again, the halogens are a very good example of group relationships (along with the alkali metals and noble gases). It would be a shame if they had to go. (The pnictogens and chalcogens do not show particularly strong group relationships.) Double sharp (talk) 11:27, 5 July 2012 (UTC)
- I was going to say that if this ends up going anywhere then the template should still include yellow shading somewhere. However, and I don't know why, this color scheme is beginning to grow on me. The argument about the halogens showing particularly strong group relationships is a good one. I think my counter argument is that the proposed categorization scheme is more consistent in terms of its logic. To add something else to the discussion I'll see if I can at least post a depiction of what the proposed template would look like. Sandbh (talk) 12:45, 6 July 2012 (UTC)
- How's the German one?
- I was going to say that if this ends up going anywhere then the template should still include yellow shading somewhere. However, and I don't know why, this color scheme is beginning to grow on me. The argument about the halogens showing particularly strong group relationships is a good one. I think my counter argument is that the proposed categorization scheme is more consistent in terms of its logic. To add something else to the discussion I'll see if I can at least post a depiction of what the proposed template would look like. Sandbh (talk) 12:45, 6 July 2012 (UTC)
- Then again, the halogens are a very good example of group relationships (along with the alkali metals and noble gases). It would be a shame if they had to go. (The pnictogens and chalcogens do not show particularly strong group relationships.) Double sharp (talk) 11:27, 5 July 2012 (UTC)
| Alkali metals | Alkaline earth metals | Lanthanides | Actinides | Transition metals | Post-transition metals | Metalloids | Other nonmetals | Halogens | Noble gases |
Double sharp (talk) 13:43, 6 July 2012 (UTC)
- I edited it slightly above; the original version is at de:Vorlage:Navigationsleiste Periodensystem. Double sharp (talk) 13:46, 6 July 2012 (UTC)
- I like/was pleasantly surprised by the fact that you can add images? to the cells to get the striped effect. I think the execution of this idea could be improved though. Just use divide the cell using a simple diagonal, for example. Sandbh (talk) 03:09, 7 July 2012 (UTC)
- I expect that a simple diagonal, whichever direction, will disturb the look of the table. It could suggest there is a sort of diagonal group running through it. Although most will known or learn what it really means, that is what the pattern structure as recognised suggests. In this table size, the 2+2 hatches and their direction look right to me. (Oh and this is somewhat preliminary: please take a look on what the dark German colors do. Text is less readable as you´ll notice, it gets more like a chaptke, especially with the hatching. But that is for when we would use these dark backgrounds). -DePiep (talk) 08:15, 12 July 2012 (UTC)
- I like/was pleasantly surprised by the fact that you can add images? to the cells to get the striped effect. I think the execution of this idea could be improved though. Just use divide the cell using a simple diagonal, for example. Sandbh (talk) 03:09, 7 July 2012 (UTC)
I think the halogens should be kept on the table. They are distinctly different from other nonmetals, and have their own chemical properties. However, in the case of astatine, I like the idea that was proposed in the Polonium--Metal or Metaloid section: See whether astatine has more metaloid or halogen properties. King jakob c (talk) 14:48, 17 August 2012 (UTC)
- At is more of a metalloid than a normal halogen, but it is still technically a halogen, so it doesn't really make sense to colour F, Cl, Br, and I as halogens and At as a metalloid, because the halogen group extends to At. The halogens don't show the strongest group relationships – there is still the nonmetal-through-metalloid-to-metal trend that you see when going down the p-block, whose effects are first seen at I and At. The other groups marked (alkali metals, alkaline earth metals, and noble gases) don't show such trends for their elements. Periodic trends for the halogens are typically shown for only F, Cl, Br, and I. As R8R Gtrs says in the next section, although the reason may vary from author to author, At is usually not included, and statements purported to be about the halogens may actually only apply to F, Cl, Br, and I. Double sharp (talk) 06:23, 18 August 2012 (UTC)
Eight category table
As flagged, here's a table that addresses the problems with the categorization of Al and At, and retains and expands the presence of group names:
Non-standard colour scheme. An old one I found in my files; haven't spent any time seeing if it could be improved.
Pre-transition metals. As used in the literature. Cox (2004, pp. 185–186) is a good example.
Rare earth metals. I've used this as a category name given it's more popular than 'lanthanides'. Google returned 1,170,000 hits for rare earth metals and 739,000 for lanthanides.
Poor metals. No accepted short-hand term exists for the well-documented notion of a bunch of 'second string' metals characterized by physically and chemically weaker metallic properties, and which generally occupy the region between the transition metals and the metalloids. I agree with Double sharp that 'poor metals' is a reasonable title for this category, given the problems with the alternatives e.g. 'B metals'; 'B subgroup metals'; 'chemically weak metals'; 'metametals'; 'other metals'; 'post-transition metals'; and 'semimetals'. [That being so, I like the old school feel of 'B subgroup metals' and could go with it as long other editors wouldn't see too much of a clash with the 'type a | borderline | type b' categorisation construct for the behaviour of metal ions.]
I've shown Be, Al, Cu, Ag, Au and Lr as partly belonging to this category. The non-metallic properties of Be (metallic-covalent bonding structure; predominately covalent chemistry; amphoteric oxides; anionic beryllate formation) are cited in the literature. Rayner-Canham and Overton (2006, pp. 29–30), for example, categorize Be as a chemically weak metal (ditto Al). For the latter, see also Metalloid#Aluminium. The elements Cu, Ag and Au are transition metals, and are included here in cognizance of the main-group chemistry of their univalent compounds; general tendency to form covalent compounds; and amphoteric oxides. Phillips and Williams (1966, pp. 4–5), for example, categorize Cu, Ag and Au as transition metals as well as B-metals. Lastly, I've counted Lr as being partly a poor metal on the basis of its predicted electronic structure of [Rn] 7s2 5f147p1 rather than [Rn]7s25f146d1.
We can discuss whether the Group 12 metals ought to instead be counted as transition metals. As I understand it, they aren't much chop physically in comparison to the transition metals proper, and chemically they're overwhelmingly not transition metals.
Core metals. No accepted short-hand term exists for the well-documented notion of a set of 'garden variety' nonmetals, between the metalloids and the noble gases. Accordingly, I suggest the descriptive title/phrase 'core metals' for this category, consistent with Wikipedia:Wikipedia is not a dictionary#Neologisms.
Group 3 membership. After thirty years, Jensen's argument (1982) for the placement of Lu and Lr in Group 3 still stands, and is better, in terms of its eloquence, than anything else on offer.
Metal-nonmetal line. I've positioned this above the centre of the metalloid category box rather than between the poor metals box and the metalloids box. I'm still in two minds as to which way would be better.
References
- Cox PA 2004, Inorganic chemistry, 2nd ed., Instant notes series, Bios Scientific, London
- Jensen WB 1982, 'The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table', Journal of Chemical Education, vol. 59, no. 8, pp. 634–636
- Phillips CSG & Williams RJP 1966, Inorganic chemistry, II: Metals, Clarendon Press, Oxford
- Rayner-Canham G & Overton T 2006, Descriptive inorganic chemistry, 4th ed., WH Freeman, New York
Sandbh (talk) 14:06, 20 July 2012 (UTC)
- First reation. I am new to the WP:ELEM community, but being a technical one I can understand the discussions.
- You talk about naming the categories: "poor metal" or "post transition metal"
- Colors: agree colors should follow their legend meaning, no problem.
- You talk about mix-grouping elements. e.g. Aluminium (Al) you mix-group (hatch) different compared to the Double sharp (German) table above.
- Colors and hatching and names, really, we can find later. Main problem: As long as there is no agreement on positioning (grouping) of mixed-category elements, there is no use talking about colors or hatching. I suggest the WP:ELEM community aim for a consensus on these mixed-category (and so mixed color) elements. I know it is old & tough, but isn't solving that what we are here for? -DePiep (talk) 21:03, 20 July 2012 (UTC)
- Here are my thoughts:
- Pre-transition metals: I still feel that the alkali and alkaline earth metals have such strong group relationships that they ought to be separated. Ditto for the halogens (which was why I was torn between putting At as a metalloid or halogen in the first place).
- Lanthanides/Rare earth metals: Technically, rare earth metals also includes Sc and Y, while lanthanides doesn't, so they're not exactly the same. (And I like the parallel between lanthanides and actinides.)
- Poor metals: No problems for me.
- Lr as a poor metal: But the Lr article states that it behaves chemically more like Lu and the actinides than Tl.
- Group 12: Here's an old discussion on the same thing. If we put Zn and Cd as post-transition (poor), then what do we do about Hg and Cn? Hg forms HgF4, and if one compound isn't enough (as Jensen states, and I also lean towards that position), Cn is predicted to show mainly the +4 oxidation state.
- Core nonmetals: I still feel somewhat uncomfortable with that term. "Other nonmetals" is far more obvious IMHO (it shows that the halogens and noble gases are also nonmetals, and that these are just the nonmetals that aren't under any other category used) and doesn't make people think that "core nonmetals" is a very standard term. I can see though that since these are the most ordinary nonmetals, "other nonmetals" might also mislead people into thinking that they're not ordinary nonmetals, so this isn't a very strong position. Don't know for sure what to do about it: maybe we should wait for the others to comment.
- Group 3: No problems for me there. R8R Gtrs converted me over from the La/Ac side to the Lu/Lr side last year. ;-)
- Metal-nonmetal line: I really like that idea. Double sharp (talk) 02:44, 21 July 2012 (UTC)
- Here are my thoughts:
- Quick feedback on all but 1,4 and 6 (pending more time).
- 2. Lanthanides/Rare earth metals: Not many more people other than chemists etc know what these are whereas 'Rare earths' are in the popular press regularly. The lanthanide title is still showing on the table right over the actinides title, albeit they do not have a colour box. Ten years ago I might've wondered about the basis for using 'rare earths' as category (I might've even said 'WTF are rare earths?'). But these days the rare earths category is almost pervasive (thanks to China). As well, the notion of parallels can be misleading. The light actinides behave more like transition metals. This is a weak argument given we say naught about the differences beween first row transition metals, and second/third row transition metals but I've thrown it into the mix for what it's worth.
- 3. Poor metals: Sweet.
- 5. Group 12: Jensen demolished the claims that Hg (in any meaningful sense) was a transition metal. I have however updated Cn to transition metal/poor metal status in light of your feedback.
- 7. Group 3: Beaut. I note Scerri supports this one too.
- 8. Metal-nonmetal line. Still not decided myself which means that, for now, I'll leave at as is/go with consensus. Sandbh (talk) 12:45, 22 July 2012 (UTC)
- First reation. I am new to the WP:ELEM community, but being a technical one I can understand the discussions.
Leave as is with halogens out. I don't want a big debate (already developed). Will try to say once and never revise the words.
- Don't group alkali and alkaline earth metals together. There is no widespread term for them all together. Pre-transition won't work. The mention on this page is the first time I've ever seen it. We'll hurt none by not combining them.
- Don't stripe the cells. Bad looks, unclarity. We're doing fine now (have been so long). Messes in heads. Please don't. There are ways out. They do exist.
- group 11 is by no means a poor-metal group (and again, destripe). If even there is basis, none (almost) recognizes it so.
- group 12...First of all, Jensen is one point of view. Not everyone agrees with him (synthesizers, reprinters, publishers). As I know, half the world finds the three transition, half not. I like the transition side, without claiming it is the right one. Pick your favorite. Think you add a d electron even with them no more reacting, it is the end of the transition and not a part of new series. And visual beauty. The only thing I ask of here: Please no stripes! BTW, copernicium hasn't been shown to be transition metal. Only a metal. (for your thoughts only, future-element chemistry is almost certian for it). By no means the stripes are suitable. (As you see, I'm really against the stripes).
- Astatine seems a metalloid to me (well-established cationic chemistry, no more words).
- halogens are not worth a color. Wait, alkaline earth are abut not halogens, you say? Yeah. Going down the table all the periods you can catch, they are still metals. Are halogens nonmetals? The what about At? Why you all think 117 will be one? There are chalcogens also, pnictogens. Halogens are just further right (no metal problem for the first four). Thus always thought as nonmetals, for some beginners even by definition. They're not. Of course many people show periodic trends on them. But because the four are nonmetals (astatine is typically out, you noticed? The reason may differ by the author, but the point is the fact is influenced by the four. Without astatine) And because you add 1 electron, easier it is. Not 2 for oxygen-sulfur-etc. You maybe noticed that the same works for alkali metals.
- Don't use yellow. More eye-catching than any other. More than our red. Or any. Any. Don't. Especially terrible between calm yet bright green and blue. Even the mild yellow makes the thing look like a peacock. In other places can bad better. Sure not good, though.
- Aluminum is pretty poor.
- Lawrencium is no poor. By the same logic is uranium a transiition element and not a actinoid. Chemistry shows the same. Eka-lutetium. Never seen literature than doesn't agree.
- Against thr rare earths. Even if the Chinese mine me as crazy (do they have their Stakhanovs?), the elements are still known as lanthanioids for most chemists (unless you are an old-school Soviet one). Rare earth is more a geologist term nowadays. If even it conquers the world, we may lose a good analogy. And we have to stripe to be correct. Why not then striping lutetium as both lanthanioid and a TM? No excitement. And no win.
Also, a funny thing that one of Berkeley reports I've seen uses our current scheme. We'll run them outdated if anything changes :-)--R8R Gtrs (talk) 14:17, 22 July 2012 (UTC)
- Some more comments based on R8R Gtrs':
- I'm on the Group-12-as-transition-metal side. If I considered group 12 as not being TMs, then I would agree with Jensen on Hg, but I prefer thinking of group 12 as a transition metal group, for reasons R8R Gtrs has already explained. Besides, not everyone agrees with Jensen, and the inclusion of group 12 into the transition metals is a matter of much debate.
- I really dislike the OR-ish "pre-transition metal" and "core nonmetal". Even if they've been used before, they are not terms even the average chemist (not to mention the average reader) will know of.
- I've never seen the group 11 elements not included into the transition metals. While they (like the group 12 elements) have filled d-subshells, the d-electrons do react (unlike the group 12 elements, which bury their d-electrons and treat them as inner electrons almost all the time, with for the exception of HgF4) and they behave like transition metals. Jensen includes them into the transition metals.
- I don't like the striping of the cells. I did mention the German one with striping, but that was only to get feedback. I don't like it myself – it's ugly. The diagonal line running through the cell just makes it worse IMHO (as someone else has said above, it could mislead people into thinking that there is a diagonal relationship running through the cell).
- I agree that astatine is certainly a metalloid. Looking at that, we probably have to get rid of the halogens category. I agree with R8R Gtrs that the line of bright yellow between the soft green and blue colours for the nonmetals and noble gases is jarring and breaks the colour trend. Even the bright red we use for the alkali metals conforms to the general colour trend; the bright yellow we use for the halogens doesn't. Anyway, At is not really a halogen if you call it a metalloid (the term halogen refers to a nonmetal in group 17), and since At is a metalloid the prospects for halogen-like chemistry for 117 look rather bleak. The Uus article itself says that the −1 oxidation state is predicted to be the least stable of the four (−1, +1, +3, +5).
- Even Jensen says that Lr isn't a poor metal, calling it just another of those Madelung rule exceptions that permeate the d- and f-blocks. Besides, it displays actinide-like and lutetium-like chemistry, and certainly not thallium-like chemistry (which would bolster the argument for calling it a poor metal).
- I agree with R8R Gtrs that Al is a rather poor metal. (Anything to avoid the stripes. Besides, once you eliminate "pre-transition metal" you are no longer able to call Al anything other than a poor metal.)
- Let's not use the "rare earths" category. Most chemists use "lanthanides" anyway. Certainly, the light actinides behave like transition metals. The lighter alkaline earth metals (Be and Mg) also behave like group 12 elements. Double sharp (talk) 10:36, 23 July 2012 (UTC)
Checkpoint summary
This is where we are up to, as I see it:
- Agree
- Poor metals (= p-block metals)
- Group 3 membership (Sc, Y, Lu, Lr)
- More evidence required as to category (no hurry; present if/when ready)
- Lr
- Cn
- Worth further arguments or consideration (IMO)
- Group 12
- Location of metal-nonmetal line in the category legend
- Quibbles: probably not worth revisiting right now
- Lanthanides v rare earths
- Group 11
- Pre-transition metals
- Colour scheme
- Agree, but how to implement?
- At is a metalloid but if we show this what do we call the remaining nonmetals between the metalloids and the noble gases?
- We could show hydrogen as its own category and the remaining nonmetals as 'typical nonmetals'. So there would be four colour-coded categories of nonmetals:
- Metalloids
- Hydrogen
- Typical nonmetals
- Noble gases
- Justification: 1. hydrogen is often given its own chapter (e.g. Cotton & Wilkinson; Greenwood & Earnshaw); 2. 'typical nonmetals' seems to be the most common term for the other nonmetals. Sandbh (talk) 13:58, 25 July 2012 (UTC)
- Disagree (ignore or continue the debate; you choose)
- Use of mixed category elements
- Elimination of halogen category
What do others say? Sandbh (talk) 13:12, 24 July 2012 (UTC)
- Since we're discussing the entire presentation of the whole periodic table(!!!) on Wikipedia, we really ought to get everyone in the project informed so you'll have more feedback than just me and R8R. Double sharp (talk) 14:39, 25 July 2012 (UTC)
- I did not follow this discussion in detail, but I am completely opposed to have one element stand out by its own color. Nergaal (talk) 15:58, 25 July 2012 (UTC)
- Nergaal, if that element has its own category in this, it should have its own legend color in this. Periodic table elements are edited from the wikipedia stone age. You were in this wp:elem project earlier that I was. What did I or you not get? -DePiep (talk) 21:21, 27 July 2012 (UTC)
- I think what Nergaal means is that there should not be legend colours used by only one element (in this case, H). Double sharp (talk) 06:21, 18 August 2012 (UTC)
- Nergaal, if that element has its own category in this, it should have its own legend color in this. Periodic table elements are edited from the wikipedia stone age. You were in this wp:elem project earlier that I was. What did I or you not get? -DePiep (talk) 21:21, 27 July 2012 (UTC)
- Yeah, it makes H stand out too much. R8R Gtrs already doesn't like the "peacock-y" even "mild yellow" colour for the halogens, and would probably strongly oppose making a single element (even more so than a single group) stand out with its own article. (BTW, what colour could we use for it? Apart from yellow, nearly everything has been used.) Also, we don't need too many colours. Already we have problems due to an overuse of reds.
- "Typical nonmetals" seems fine iff it's widely used.
- The most important things to talk about IMHO are probably group 12, the metalloid line, and the proposed elimination of the halogen category. Group 3 and poor/post-transition metals could be talked about with those who made the change several years ago. I really dislike the mixed-category diagonally-divided cells, and so does R8R Gtrs; what do others have to say on that matter? Double sharp (talk) 12:19, 26 July 2012 (UTC)
- I added an announcement on the noticeboard regarding this. Double sharp (talk) 05:04, 29 July 2012 (UTC)
- I did not follow this discussion in detail, but I am completely opposed to have one element stand out by its own color. Nergaal (talk) 15:58, 25 July 2012 (UTC)
A simple upgrade would be to:
(1) change 'post-transition metals' to 'poor metals';
(2) change the colour coding of astatine to that of a metalloid;
(3) change the 'halogen' category to a 'highly active nonmetals' category;
(4) change the colour coding of nitrogen and oxygen to that of highly active nonmetals (in light of their high Pauling electronegativity values); and
(5) change the 'other nonmetals' category to 'moderately active nonmetals'.
Such an upgrade would:
- fix Al and At;
- retain the spirit and colour of the halogen category;
- be consistent with the descriptive theme of the 'noble gases' category (i.e. noble = not very active);
- retain the 'metalloids' category like it is now, as a discrete top-tier category alongside metals and nonmetals; and
- not require any mixed category elements :)
I think an argument could be made that iodine is not quite in the same league as the other active nonmetals when it comes to EN and, for example, oxidizing power, but would be happy enough for now with the highly active nonmetals being shown as N, O and the remaining (nonmetal) halogens. Sandbh (talk) 08:30, 30 July 2012 (UTC)
- But nobody (well, except you) seems to call them "highly active nonmetals" and "moderately active nonmetals". Double sharp (talk) 08:58, 30 July 2012 (UTC)
- Hmm. I get 7,969 hits googling the web for "active nonmetals" and 604 on Google Books. The search results go along the lines of:
- "…fluorine, chlorine, bromine, and iodine are active nonmetals…";
- "The most active nonmetals are found in the upper right corner of the periodic table (excluding the noble gases)"
- "…across each period there is a gradual transition from an active (alkali) metal through less active metals and nonmetals to highly active nonmetals (halogen)…"
- "The elements change from active metals to less active metals, to metalloids, to moderately active nonmetals, to very active nonmetals…".
- Hmm. I get 7,969 hits googling the web for "active nonmetals" and 604 on Google Books. The search results go along the lines of:
- If I search instead for "reactive nonmetals" I get 16,700 web hits and another 352 via Google Books.
- I wouldn't mind if the two categories got called e.g. (a) less active, and active nonmetals, or (b) moderately actively and highly active nonmetals, or (c) moderately active and very active nonmetals, or some other combination. I like 'active' better than 'reactive', as active is a more encompassing term. Sandbh (talk) 12:57, 30 July 2012 (UTC)
- Interesting. It's always nice to learn something new. But I think we should use the most commonly accepted and understood terms in the legend. Otherwise, we'll have to explain exactly what "active" refers to here every time we include a periodic table on an article. We can use this terminology in the main articles, like mentioning in the halogen article that they are all (well, except At) very active nonmetals, and so are N and O. Double sharp (talk) 06:37, 4 August 2012 (UTC)
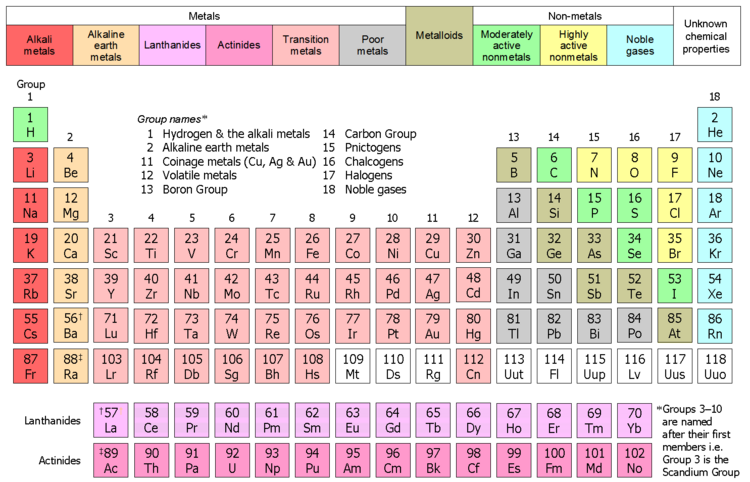
Time to deprecate 'other nonmetals'
Double sharp, thank you. I've been thinking about this for a while and was just about to post something when I saw your comments, just above. DePiep was right when he said, "I know it [element categorization] is old & tough, but isn't solving that what we are here for?." I'll post what I was going to say and then respond to your comments.
Here's a table showing the proposed element categories:
The colour scheme is the standard one. The halogens aren't shown with a separate colour, on account of the (very good) reason given by R8R Gtrs.
The distinction between highly active and moderately active nonmetals follows that of Wulfsberg (1987, pp. 159–161). He groups the nonmetals into two categories, based on their electronegativity value. Nonmetals having an electronegativity of > 2.8 he calls very electronegative nonmetals (= N, O, F, Cl, Br); the rest, including iodine, he calls electronegative nonmetals. I've used 'active' instead of 'electronegative' as the main adjective, in order to be consistent with the categorization of noble gases as 'noble' or 'inactive' nonmetals.
In chemistry terms, the two categories of highly active and moderately active nonmetals are congruent with HSAB theory: highly active nonmetals are hard or borderline bases; the remaining moderately active nonmetals (including H– and I–) are soft bases.
Response to your comments (Rant warning: not aimed at you, just a general one). The term 'Other nonmetals' is "malarky". It means nothing. It's a "garbage can" term, for the leftovers, when nothing better comes to mind. It has almost zero information content. The first time I saw 'Other nonmetals' was in Wikipedia. Having not seen the term before, I remember thinking WTF are other nonmetals? Metalloids: check! Halogens: check! Noble gases: check! But WTF are other nonmetals? Sure, I knew the individual elements but had no idea what the common thread was that caused them to be called other nonmetals. The 'other' in 'other metals' conveyed no sense of meaning. OTOH, the meaning of highly/moderately active nonmetals would be significantly more familiar to most readers, including chemistry, science, and related professionals (IMO). Whereas there is (almost) no chemistry behind 'other metals', no immediately obvious sense of meaning, and no value or information add. It's an awful term that we should seek to consign to the dustbin of history, given the existence of much better terminology, in terms of (1) precedent in the literature; (2) grounding in chemistry; (3) meaning; (4) value add; and (5) internal consistency (per R8R Gtrs' insightful observation). That concludes today's rant.
- Wulfsberg G 1987, Principles of descriptive inorganic chemistry, Brooks/Cole, Monterey CA, ISBN 0-534-07494-4
Sandbh (talk) 15:00, 4 August 2012 (UTC)
- The current one is not bad – I like it. And yes, anything to get rid of the "other nonmetals" grouping. (It's obvious what it means, but it doesn't make much chemical sense.) (I was thinking of "typical nonmetals" when I made that comment, BTW.)
- I have a feeling people will come and make noise with Lu/Lr being shaded as transition metals, BTW. They're also lanthanides. And they might even argue that shading them as lanthanides while not putting them together with the lanthanides is confusing(!!). We'll see what they think of this Lu/Lr shading; I'm on your side.
- Shouldn't we alert all the members of WP:ELEM, BTW? The announcement doesn't seem to be working. The only responses you've got so far are from me and R8R, and that's hardly a consensus (3 people only!). Double sharp (talk) 06:29, 5 August 2012 (UTC)
- And BTW, "volatile metals"? Could you please tell us where you found that term? It sounds like an interesting addition to the Group 12 element article. (And who used "coinage metals" for group 11?) Double sharp (talk) 06:34, 5 August 2012 (UTC)
- Five citations re Group 12 volatile metals:
- 1. '…the volatile metals, arsenic, antimony, zinc, cadmium, and mercury, are inverted into oxides when heated in air or oxygen' (Fownes 1869, p. 279).
- 2. 'Still further to the right the coinage metals (Cu, Ag, and Au) and volatile metals (Zn, Cd and Hg) show strong vertical relationships' (Simmons 1947, p. 589).
- 3. '…zinc, cadmium and mercury…are unusually low melting and volatile' (Massey 2000, p. 159).
- 4. 'Because zinc, cadmium, and mercury…have comparatively low boiling points, 907°, 768°, and 357°, respectively; they are referred to frequently as the volatile metals' (Beringer 2005, p. 368).
- 5. In New Scientist 21 April 2012, there is an article (p. 12) called 'A factory for elements that barely exist', by Kat Austen. In part, the article notes that, 'Copernicium...is more volatile than its homologue mercury, and is the only metal known to be a gas at room temperature'. Apparently this is a 'deduction' based on how far atoms of 112 travelled across a gold surface, so saying 'known' is hyperbole. An engaging conclusion, in any event.
- Beringer P 2005, Remington: The science and practice of pharmacy, 21st ed., Lippincott, Williams & Wilkins, Baltimore
- Fownes G 1869, A manual of elementary chemistry, Henry C Lea, Philadelphia
- Massey AG 2000, Main group chemistry, 2nd ed., John Wiley, Chichester
- Simmons PA 1947, 'A modification of the periodic table', vol. 24, no. 12, Journal of Chemical Education, Dec, pp. 588–591
So, after over a month of discussion, shouldn't we ask the members who haven't participated in this discussion what they think the outcome should be, similarly to what happened with our vote for a flagship article (which was decided to be hydrogen)? Double sharp (talk) 12:49, 28 August 2012 (UTC)
- Yeah, go 'head. Think of inviting Wp:CHEMS and Wp:SCIENCE members, also--R8R Gtrs (talk) 20:26, 4 September 2012 (UTC)
- P.S.I don't want to get in deep, but I'd not call nitrogen active...trying heating it with iron and then with "non-active" sulfur, and see which of the two reactions does occur
- I am neutral on the other proposals, but I don't agree with splitting into the current green and yellow (very OR). If we get rid of halogens, then just merge them with with the other nonmetals. Also, instead of reactive electronegativity could be used, but that would be still OR-ish. Nergaal (talk) 00:51, 5 September 2012 (UTC)
- Has anyone thought of giving hydrogen its own category? Just throwing this out there. StringTheory11 (t • c) 00:55, 5 September 2012 (UTC)
- That was discussed in the previous section, but Nergaal and I are against it, because it makes hydrogen stand out too much. (R8R already finds the yellow of the halogens too "peacock"-y, so I don't think he'd like that suggestion either.) And after, H is a nonmetal.
- We should change "other nonmetals" to a different name (non-OR, of course), though. "Typical nonmetals" was suggested, and seems fairly common. Double sharp (talk) 03:58, 5 September 2012 (UTC)
- Has anyone thought of giving hydrogen its own category? Just throwing this out there. StringTheory11 (t • c) 00:55, 5 September 2012 (UTC)
- I am neutral on the other proposals, but I don't agree with splitting into the current green and yellow (very OR). If we get rid of halogens, then just merge them with with the other nonmetals. Also, instead of reactive electronegativity could be used, but that would be still OR-ish. Nergaal (talk) 00:51, 5 September 2012 (UTC)
My proposal (close to R8R's):
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
(BTW, don't delete Template:Element color/Halogens after it becomes unused. That would break almost every old revision of a periodic table.)
Of course, H is not really a typical nonmetal. But then, what do we do with it?
(P.S. Cn is actually only known to be a metal, and not a transition metal, as R8R Gtrs states. However, we can't really use any other colouring than "transition metal".) Double sharp (talk) 05:43, 5 September 2012 (UTC)
- I reread this section Time to deprecate 'other nonmetals'. (Organisational note: I'll respond to that topic, so Group 12 is not involved; the discussion started earlier in to main thread section #Eight category table. Really, discussing multiple independent topics in one thread is not helpfull). This is what I found:
- - Sandbh and R8R propose: regroup & rename into "highly active nonmetals", "moderately active nonmetals" (or such names; based on electronegativity), no category named Halogen (as a group it stays though). Source: Wulfsberg G 1987.
- - Double sharp proposes "other nonmetals" (currently used).
- - Other suggestions ("core nonmetals", HSAB-based, ...) look less substantial to me.
- My points:
- 1. Although "other nonmetals" is not used widely outside of wp, if we need a description of that category we are perfectly free to do so. Maybe the capital should go as it is not a proper name, but a description we can provide, especially with a wikilink. I can also note that in the Venn-diagram of category names and descriptions it serves a completifying task (see row 3 & 4 in the basic legend: a complete, mutually exclusive set of category names and their colors).
- 2. That said, I cannot be convinced by some of Doublesharp's arguments: [1] (that's where legends & wikilinks are invented for); [2] ("I like"), and other argumentation. I admit all your intuition in this (most other editor's here) is far better than mine, but if one cannot argument or describe the base, it is more like OR or personal preference.
- 3. Editors seem to have unarticulated opposition (uncertainity? fear?) to removing Halogens from the list of categories. While really, that is a consequence of mixing up topics. Halogens is and will stay a group. As a category, if the logic, properties and sources require to split the set up: then it be so. Already now I count some six groups that are not homogeneous wrt category. No problem.
- 4. I support the proposal using "highly active nonmetals", "moderately active nonmetals" (Sandbh & R8R), because it is based on a source, has a straight scientific base in electronegativity, and has a small undecided grey area (an area that takes up so much space on this page). It implies we drop the Halogens category here.
- 5. Consequences would be: check and maybe split Halogen page, review legend colors &names. All this can follow a tough logic on category classification. -DePiep (talk) 10:11, 5 September 2012 (UTC)
- Hmm. In this thread, what I see is:
- Sandbh proposes the one in the image.
- I propose "typical nonmetals", with halogens merged into typical nonmetals.
- The way I see it, R8R was actually referring to my comment when he said "go 'head", and did raise a point that the "moderately active" and "highly active" is quite debatable (Fe will react with S, but not with N).
- Nergaal supports merging the halogens into the other nonmetals, and opposes the highly active/moderately active division because it is "very OR" (I agree; the source is talking about EN values, not reactivity) and is quite debatable (R8R raised the case of N not being that reactive, and yet having a high EN.)
- ST11 proposes giving H its own category (which would solve the problem with my and Nergaal's proposal where H is not a very typical nonmetal), but Nergaal and I had previously (see the previous subsection) opposed this because it would make H stand out too much. Double sharp (talk) 10:30, 5 September 2012 (UTC)
- Hmm. In this thread, what I see is:
how about reactive non-metals (i.e. not inert) instead of typical? Nergaal (talk) 14:28, 5 September 2012 (UTC)
==== random break ====
- Hey, don't make me look silly. I responded to a subsection (level 4) created by Nergaal. -DePiep (talk) 23:07, 9 September 2012 (UTC)
- Probably because nitrogen is nearly inert at room temperature; even hydrogen is "comparatively unreactive at room temperature…" (Greenwood & Earnshaw 1998, p. 43) Sandbh (talk) 14:18, 8 September 2012 (UTC)
- also, I would be fine with merging group 1 & 2 into s-block metals, and Ln and An into f-block metals, or just inner-trans metals. Nergaal (talk) 14:30, 5 September 2012 (UTC)
- Why two unrelated proposals in one subsection, why a new subsection (level 4) when you are adressing a running conversation in the previous (level 3), and why I would be fine with as an argument? How is the titleword random helpfull? -DePiep (talk) 14:41, 5 September 2012 (UTC)
end of ==== random break ====
Yes, I'm not really not in favor of cutting nonmetals in two (did offer invitations instead). I think the usage of the term shown is more like that of a useful phrase than a strict scientific term (doubt it that there are established definitions of "moderately active nonmetals"), this configuration's also tricky.--R8R Gtrs (talk) 14:56, 5 September 2012 (UTC)
- DePiep has hit the nail on the head with his point (3) above. I, too, shared the 'unarticulated opposition' — I might even say 'visceral reaction' — to eliminating Halogens, but it really is as he says, confusing Category with Group. Perhaps the solution, at least as far as the template goes, is to add emphasis for the groups. R8R Gtrs listed most of the groups in his picture [#Time to deprecate 'other nonmetals'|above]], but I think we could do even better. Maybe a complete list of group numbers and names at the bottom, including the groups named after their first element, and each one is hyperlinked to the wp article about the group. And, if someone can figure out the wikimarkup magic, have the group column and its number/name highlighted when the cursor hovers over it. Something like this would serve to accentuate the groups including halogens and so reduce opposition to eliminating halogens as a category. YBG (talk) 05:49, 8 September 2012 (UTC)
- That was Sandbh's picture, actually. Double sharp (talk) 07:00, 8 September 2012 (UTC)
This came to my mind: can we call that category "unspecified nonmetals" (halogens and noble gases being the specified ones of course)? In standalone use, outside of the categories, the wording "other nonmetals" is awkward indeed. The reader thinks he or she is missing something (correctly). -DePiep (talk) 18:59, 25 November 2012 (UTC)
Hydrogen (and categories v groups)
Sorting out hydrogen is the key, as I see it. StringTheory11 was on the mark with his suggestion along the lines of showing hydrogen in its own non-metal sub-category. As per DePiep's approach, there is strong support for such a treatment in the literature. For example: "The chemistry of hydrogen is so unique that this element is in reality in a class by itself" (McCoy & Terry 1920, p. 562); "The chemistry of hydrogen bears little resemblance to that of any other element…" (Emsley 1971, p. 20); "Hence the chemistry of hydrogen is the only chemistry of its kind, as it were, the chemistry of an elementary particle, the proton." (Trifonov & Vlasov 1987, p. 24). Nergaal: unless I've missed something your opposition to this proposal, on the basis that hydrogen would then stand out too much, appears to lack a strong scientific basis.
If hydrogen is recognized as a nonmetal in its own subcategory, the rest of the categorization puzzle more or less falls into a better place. Major categories would be Metals, Nonmetals, and Unknown chemical properties. Subcategories would be (current) alkali metals through transition metals, plus poor metals; then metalloids, hydrogen, typical nonmetals, and noble gases. Having thought about this some more I'd also recommend that the shared borderline between the metals category box and the nonmetals category box be positioned over the middle of the metalloid subcategory box (in the same manner as is shown in the eight category table), rather than the current practice of showing metalloids as a major category.
YBG, re your vision of a periodic table showing both categories and groups: sign me up.
- Emsley J 1971, The inorganic chemistry of the non-metals, Meuthen Educational, London
- McCoy HN & Terry EM 1920, Introduction to general chemistry, McGraw-Hill, New York
- Trifonov DN & Vlasov LG 1987, Silhouettes of chemistry, Mir Publishers, Moscow
Sandbh (talk) 14:18, 8 September 2012 (UTC)
- Here's a compact periodic table using Sandbh's latest proposal (just above):
| H | He | |||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | |||||||||||
| ||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||
I've deliberately used a soft colour here for H, so that it doesn't stand out too much, but this colour is in no way finalized and is open to suggestions. Double sharp (talk) 14:56, 8 September 2012 (UTC)
- Thank you Double sharp. I've edited the table by moving the hydrogen sub-category box from being in front of the alkali metal sub-category box to being in-between the metalloid and typical nonmetal sub-category boxes. On the question of color, and as a personal preference, I believe there ought to be room for some yellow somewhere, not necessarily for H. Sandbh (talk) 23:32, 8 September 2012 (UTC)
- Maybe replace the poor metals color with yellow? Gray is never a good color to use for something known. StringTheory11 (t • c) 00:00, 9 September 2012 (UTC)
- Again and again I say: use a straight scientific criterium, and then stick with that. Afterwards, we can do colors & borders & descriptions. -DePiep (talk) 00:10, 9 September 2012 (UTC)
- Maybe replace the poor metals color with yellow? Gray is never a good color to use for something known. StringTheory11 (t • c) 00:00, 9 September 2012 (UTC)
- How is hydrogen more special then helium? Helium has the highest electronegativity, is the least reactive, is the only element that is fine with 2 electrons in the electron shell (H is typicall happy with 0) and so on? Why aren't we emphasizing carbon also? Is the only element that can form long chans of single bonds that are stable under noral conditions? why not lead since it is the heaviest stable element? I can give many other examples why a lot of elements are unique. the point of the periodic table is to be informative, not to give a color to each element. Nergaal (talk) 00:47, 9 September 2012 (UTC)
- I'm a little bit confused about "typical nonmetals." I perfectly remember that my teacher back in school told me fluorine was the only typical nonmetal, since she named the ability to form positive OS a metal-like property and fluorine is the only one not to. The opponents say "other" is a bad category. I'd rather say so not. It's a very easily understood term. It is very general. You may not understand what "transition" about the pink cells is, but "other" is clearly gettable. Plus we could have minus a color by getting this (in my opinion, a very good) category. It is not a term for scientists... But we are specializing mainly on the casual people, right? I'm referring to the fact that "typical" may puzzle a non-chemist.--R8R Gtrs (talk) 17:50, 10 September 2012 (UTC)
- I suppose "typical" can mean different things to different people. But then, so does "transition" (group 12, and to a much lesser extent 3 and 11 some time ago.)
- I understand your rationale, R8R. But "other metals" doesn't provide that much information for a reader. At least with a name like "transition metal", even if what the transition is is not obvious, at least the reader knows that there is some sort of transition. With generic nondescript names like "other metals", though, the reader doesn't get this information, and won't understand how exactly these elements are similar to each other. Double sharp (talk) 10:55, 11 September 2012 (UTC)
- I'm a little bit confused about "typical nonmetals." I perfectly remember that my teacher back in school told me fluorine was the only typical nonmetal, since she named the ability to form positive OS a metal-like property and fluorine is the only one not to. The opponents say "other" is a bad category. I'd rather say so not. It's a very easily understood term. It is very general. You may not understand what "transition" about the pink cells is, but "other" is clearly gettable. Plus we could have minus a color by getting this (in my opinion, a very good) category. It is not a term for scientists... But we are specializing mainly on the casual people, right? I'm referring to the fact that "typical" may puzzle a non-chemist.--R8R Gtrs (talk) 17:50, 10 September 2012 (UTC)
- By analogy to "poor metals", why not "ignoble non-metals" or "peasant non-metals"? :) YBG (talk) 06:58, 11 September 2012 (UTC)
- LOL. But I don't think any researcher has used such terms, except perhaps in jest! :-) Double sharp (talk) 10:55, 11 September 2012 (UTC)
- "Ignoble" would be a good antonym of "noble", though. For example, "Caesium is the most ignoble metal..." Double sharp (talk) 12:27, 14 September 2012 (UTC)
- LOL. But I don't think any researcher has used such terms, except perhaps in jest! :-) Double sharp (talk) 10:55, 11 September 2012 (UTC)
- By analogy to "poor metals", why not "ignoble non-metals" or "peasant non-metals"? :) YBG (talk) 06:58, 11 September 2012 (UTC)
Hydrogen is more special than other elements because its chemical behavior is distinctly odd—much more so than any other element. Two results then come to mind. The first is that hydrogen doesn't have any nonmetal or metal peers (unlike helium -> noble gases; carbon -> typical nonmetals; and lead -> poor metals). The second is that hydrogen doesn't map well to any periodic table group—witness over 100 years of arguments as to which group hydrogen should belong to (including e.g. 1, 14, 17, no group, its own group or multiple groups), and Mellor's description of hydrogen as a 'rogue' element.
In terms of its chemical behavior, hydrogen:
- is a good reducing agent
- is liberated at the cathode during electrolysis
- is displaced by more active metals from dilute acids or water
- forms its most stable compounds with non-metals
- forms a solvated cation in aqueous solution
- is capable of forming alloys (solid solutions) with metals.
This is very strange behavior for a nonmetal. Even when behaving more conventionally as a non-metal, in the form of a hydride ion, the resulting compounds are intrinsically unstable since the hydrogen atom, with its single proton, is unable to sufficiently control two valence electrons.
Hydrogen doesn't satisfy any descriptive criterion or set of criteria attempting to define a metal that I'm aware of. It's clearly a non-metal in that sense. But neither is its chemistry—which is the chemistry of the proton rather than the chemistry of an element—typical (so to speak) of typical non-metals. Sandbh (talk) 11:58, 11 September 2012 (UTC)
- OK, let's go on thinking hydrogen is quite different than the other nonmetals. I think the current "other nonmetals" punchline works; why would separate them? Let's color Alaska or Hawaii differently than the rest of U.S. There are differences in climat, population, lots of stuff. But these are still U.S.! (I know it's kinda poor an analogy, it's to make an impression, not to serve as a point)
- When did I say I like "other metals"? No, I love poor more. It's an established term with more-or-less fixed borders.
- (citation: "But "other metals" doesn't provide that much information for a reader. At least with a name like "transition metal", even if what the transition is is not obvious, at least the reader knows that there is some sort of transition. With generic nondescript names like "other metals", though, the reader doesn't get this information, and won't understand how exactly these elements are similar to each other.") Huh! Like your point about the transition. Good. But think it should be used slightly differently. I see your point about not getting the info. Yet I think that if it doesn't work, "typical" won't work either, cause, well, it's not that common. If you understand it, than you would also understand the point of "other," as well. I even heard concerns that phosphorus and selenium are not typical nonmetals in Wiki quite lately (don't recall the context and exact phrasing). What I'm getting at is, it's not that necessary to separate other nonmetals and the lonely hydrogen. Because there is a common criterion to include them all together (nonmetallicity or whatever the word is). Want the thing to be as basic as it keeps sence. Also, why is H chemistry called chemistry of a particle? There are also deuterium, tritium, that are almost identical on chemistry (a couple of degrees diff in water mp and the Iike.) If it is related to abundance, then it's not strictly scientific, more a figure of speech.
- What do we gain by separating hydrogen? I'm pointing at the non-chem audience again. There are more people who would not clearly get the point than those who would. Don't fix it if it ain't broke.
- Sorry, feel sick, so not sure if I'm understandable, hope you get my points--R8R Gtrs (talk) 19:39, 12 September 2012 (UTC)
- R8R Gtrs, I don't understand your points. The term 'other nonmetals' is bad, unhelpful, lazy science. It says (almost) nothing about the nature of these elements. On the other hand, calling them 'typical nonmetals' says that that is just what they are–'typical' for their kind. So, if you want to learn about nonmetals, start with these as the most typical examples of nonmetals. Even non-chemists would be able to work that out.
- If you think non-chemists won't be able to work out what 'typical nonmetals' means, fair enough–we will have to disagree on that point. Your assertion that if someone understood 'typical nonmetals' then they would also understand 'other nonmetals' makes no sense to me.
- The subcategory of typical nonmetals still shows some variability in degrees of nonmetallic character. As you note, fluorine is probably the most non-metallic of the typical nonmetals. Whereas phosphorous and selenium are neighbors of the metalloids so they show some metalloid-like attributes. But they are not quite metalloids and can still (mostly) be accommodated within the typical nonmetals subcategory.
- H chemistry is called the chemistry of the proton since, once H loses its single valence electron it doesn't have an inner electron screening its nucleus, unlike all other elements. Sandbh (talk) 11:27, 14 September 2012 (UTC)
- I dunno. I feel I got nothing left, so every point I can think of has been said already. You're good, I like your points, it's starting to get me... Yet I don't feel like it. Really dunno. I may explain what I meant when writing those. I think that-- well, no agree, but at least you do understand why I am in favor of that one, right?-- the "other" is easy to get. And typical needs more knowlegde to get it. No description, okay. I just realized why you protect your points. I still think the less categories as it keeps sense, the better (colors go before names, IMO, even though that's the point you disagree with, right?). You go 'head and disagree. That's awesome, since you back up yourself with citations and keep head cool. And I, in contrast, have mentioned no source really, and now got nothing left. I'll be now more okay with your scheme. Thanks for explaining it over to make me understand your points (will try to go unblinded more now)
- (About particle chem) Still, H doesn't completely lose the electron (always covalent bonding), so some screening still remains. Naked proton is still unknown in chemistry, huh?--R8R Gtrs (talk) 19:21, 14 September 2012 (UTC)
- Yep, no sight of a naked proton, as far as I know Sandbh (talk) 12:31, 30 September 2012 (UTC)
- @R8R: Whether you colour Alaska and Hawaii differently from the rest of the U.S. or not depends on what you are trying to emphasize, doesn't it? You would do that if you were emphasizing climate or population, as you say, but not if you were emphasizing current political boundaries. We've decided to classify the elements chemically. We could just as well have classified them by blocks. Chemically, H stands out quite noticeably from the other nonmetals. Double sharp (talk) 12:27, 14 September 2012 (UTC)
- Stands out... I may be fine with separating on the grounds of getting a more meaningful name, but that one is still not for me (worked years fine, and without that main reason it seems no good. I think that nonmetals are a label, like countries to some extent are (like that triangle in the north of Sudan, or jungle borders in Central Africa, sometimes controled by no state, just worldwide recognized claims exist, but no real control. There are still uncontacted people no state can control. Hydrogen is recognized as a nonmetal (no opposition), can go under the same title and color (so like the map analogy (was no argument, just analogy to make easier to understand), do you get it now?). Like those Central African jungleforests. You maybe remember I like fever colors as it keep having sense. Do as you like, though. Got nothing more to say--R8R Gtrs (talk) 19:15, 14 September 2012 (UTC)
- It seems to me that the problem here is that "Other nonmetal" and "Typical nonmetal" makes is sound like there is a specific group named "Other" or "Typical", which there is not. So that led me to think that maybe the thing to do was to set up the grid so that it was intuitive that "Other" was NOT the name of a group, but simply a label that meant "none-of-the-above". It would be a whole lot easier if it could come after (i.e., to the right) of the groups with specific names, but that rather spoils the left-to-right correlation with the order of appearance in the Periodic Table. Anyway, the wikimarkup is lousy, but maybe this would get the creative juices flowing and help us find a way forward. YBG (talk) 03:41, 15 September 2012
- Stands out... I may be fine with separating on the grounds of getting a more meaningful name, but that one is still not for me (worked years fine, and without that main reason it seems no good. I think that nonmetals are a label, like countries to some extent are (like that triangle in the north of Sudan, or jungle borders in Central Africa, sometimes controled by no state, just worldwide recognized claims exist, but no real control. There are still uncontacted people no state can control. Hydrogen is recognized as a nonmetal (no opposition), can go under the same title and color (so like the map analogy (was no argument, just analogy to make easier to understand), do you get it now?). Like those Central African jungleforests. You maybe remember I like fever colors as it keep having sense. Do as you like, though. Got nothing more to say--R8R Gtrs (talk) 19:15, 14 September 2012 (UTC)
| Metal groups | Metaloid |
Nonmetal | Unknown chemical properties | |||||||
| Alkali metal |
Alkali earth metal |
Lanthanide |
Actinide |
Transition metal |
Post-transition metal |
Other nonmetals |
Halogen |
Noble gas | ||
- "Other nonmetals" is probably not used as a group name, but "Typical nonmetals" seems to be used that way. So I don't think there is a problem if we changed to using "typical nonmetals". (Of course, we'll need to sort out hydrogen, as its properties are definitely atypical for a nonmetal.) Double sharp (talk) 05:50, 15 September 2012 (UTC)
Reactive nonmetals and noble gases
Nergaal was right when he said, 'how about reactive non-metals (i.e. not inert) instead of typical? Nergaal (talk) 14:28, 5 September 2012 (UTC)'. Sure, hydrogen is comparatively unreactive at room temperature and nitrogen is nearly inert but both are way more reactive elements—in general—than the noble gases, as are the rest of the non-noble nonmetals. Using reactive nonmetals instead of typical nonmetals makes the hydrogen problem go away. And whereas I can't find much specific usage in the literature of other nonmetals there are many more mentions I can find of reactive nonmetals or variations thereof (e.g. less reactive, reactive, more reactive, highly reactive, most reactive etc). So, it's a big metalloid vote from me to replace other nonmetals with reactive nonmetals.
The only question then remaining would be whether to distinguish between highly reactive nonmetals (O?, F, Cl, Br?) and the rest of the (merely) reactive nonmetals. The halogens may not have their own color anymore but there may still be some value in highlighting the highly reactive nonmetals, given this terminology is often associated with the elements in the vicinity of the top right hand corner of the periodic kingdom. Sandbh (talk) 23:00, 19 September 2012 (UTC)
- Here are some quotes from the literature showing usage of the expression 'reactive nonmetals':
- 1. 'Fluorine and chlorine are the most active of the reactive non-metals, while nitrogen is the least active of this class.' (Biddle & Bush 1949, p. 181)
- 2. 'If the position of the Group 0 elements is excluded, the most reactive non-metals are found in the upper right-hand portion of the table.' (Murphy & Rousseau 1969, p. 81)
- 3. 'In each case, there is a left-to-right trend from very reactive metals to less reactive metals followed by less reactive nonmetals and on to quite reactive nonmetals, ending with an essentially unreactive, or inert, element. This pattern is referred to as periodic behaviour.' (James, Schreck & BeMiller 1980, p. 26)
- 4. 'Adjacent to the inert gases in the outlined portion of the diagram are sixteen elements that are reactive non-metals. The group includes solids such as carbon (C), phosphorus (P), and sulfur (S), as well as gaseous elements (H, N, O, F, CI).' (Leinfelder, Taylor & Morr 1982, p. 13)
- 5. '…nonmetals can be split into two groups, the 'noble' or inert gases and the reactive nonmetals.' (Long & Hentz 1986, p. 84)
- 6. 'By the end of 8th grade, students should know that…There are groups of elements that have similar properties, including highly reactive metals, less reactive metals, highly reactive nonmetals (such as chlorine, fluorine and oxygen) and some almost completely unreactive gases (such as helium and neon).' (AAAS 1994, p. 78)
- 7. 'From left to right across the periodic table, elements change from being reactive metals, through less reactive metals, metalloids, less reactive non-metals to reactive non-metals. (On the extreme right are the noble gases.)' (Hill & Holman 2000, p. 49)
- 8. 'Describe how groups of elements can be classified based on similar properties, including highly reactive metals, less reactive metals, highly reactive nonmetals, less reactive nonmetals, and some almost completely nonreactive gases.' (Padilla, Cyr & Miaoulis 2005, p. 27)
- 9. 'Nonmetallic elements span the range from extremely reactive fluorine to relatively unreactive elements such as carbon and nitrogen.' (House 2012, p. 393)
- 1. 'Fluorine and chlorine are the most active of the reactive non-metals, while nitrogen is the least active of this class.' (Biddle & Bush 1949, p. 181)
Sandbh (talk) 04:08, 22 September 2012 (UTC) Sandbh (talk) 08:50, 25 February 2013 (UTC)
- American Association for the Advancement of Science 1994, Benchmarks for science literacy, Oxford University Press, New York
- Biddle C & Bush GL 1949, Chemistry today, Rand McNally
- Hill G & Holman J 2000, Chemistry in Context, 5th ed., Thomas Nelson & Sons, Cheltenham
- House JE 2012, Inorganic chemistry, 2nd ed., Academic Press, Burlington MA
- James ML, Schreck JO & BeMiller JN 1980, General, organic, and biological chemistry: chemistry for the living system, D. C. Heath, Lexington, Mass.
- Leinfelder KF, Taylor DF & Morr DV 1982, Laboratory and clinical dental materials, 3rd ed., University of North Carolina Press, Chapel Hill
- Long GG & Hentz FC 1986, Problem exercises for general chemistry, 3rd ed., John Wiley & Sons, New York
- Murphy DB & Rousseau V 1969, Foundations of college chemistry, The Ronald Press Company, New York
- Padilla MJ, Cyr M & Miaoulis I 2005, Prentice Hall Science Explorer (Indiana, Grade 6): Teacher's Edition, Prentice Hall, Upper Saddle River, NJ
- Sure, I can see that the expression "reactive nonmetals" is used. But then, how do we decide what to put in it? The precise elements in that category seem to be somewhat debatable (Br? N? H?). Double sharp (talk) 04:18, 22 September 2012 (UTC)
- Are you asking if there would be a basis to distinguish between highly reactive nonmetals v (merely) reactive nonmetals? If so, my first response would be to suggest that any nonmetal capable of reacting with a noble gas to form a simple compound ought to considered a highly reactive nonmetal. Without having looked into noble gas chemistry too much I suspect that such an initial criterion would apply only to oxygen and fluorine. Sandbh (talk) 03:22, 23 September 2012 (UTC)
- Possibly chlorine would also count, because of XeCl2, but it's still uncertain whether it is really a true compound. So that criterion would give the list O, F, and possibly Cl. (But aren't there many possible criteria for distinguishing them? Then how do we decide which to use? You did use a different one in the previous table where N and Br were also added to this list, and R8R Gtrs pointed out that Fe reacts with S, but not with N.) Double sharp (talk) 05:15, 23 September 2012 (UTC)
- Are you asking if there would be a basis to distinguish between highly reactive nonmetals v (merely) reactive nonmetals? If so, my first response would be to suggest that any nonmetal capable of reacting with a noble gas to form a simple compound ought to considered a highly reactive nonmetal. Without having looked into noble gas chemistry too much I suspect that such an initial criterion would apply only to oxygen and fluorine. Sandbh (talk) 03:22, 23 September 2012 (UTC)
(outdent) What about dividing nonmetals into (1) Hydrogen (2) Solid nonmetals and (3) noble gases? YBG (talk) 05:53, 23 September 2012 (UTC)
- Except, bromine is a liquid.--Jasper Deng (talk) 06:02, 23 September 2012 (UTC)
- Details, details, details. (Headslap) And non-gasseous non-metals is a bit too cumbersome, to say nothing about the clumsiness of defining the group by what it isn't. YBG (talk) 07:35, 23 September 2012 (UTC)
- Besides, N, O, F and Cl are gases, but are certainly not noble gases. If we include them (and Br, the liquid nonmetal) into the "Solid nonmetals" category, that category has then become "Typical nonmetals". We shouldn't colour the entire cell based on state at STP – that's what the colouring of the atomic number is for. Double sharp (talk) 08:46, 24 September 2012 (UTC)
- How could I have been so rediculous! I was trying to think outside the box, but apparently I slipped into some sort of an alternate reality. Many appologies for wasting your time. You guys deserve a barnstar for your polite responses to me!!!! Many thanks!!! YBG (talk) 14:31, 24 September 2012 (UTC)
- Besides, N, O, F and Cl are gases, but are certainly not noble gases. If we include them (and Br, the liquid nonmetal) into the "Solid nonmetals" category, that category has then become "Typical nonmetals". We shouldn't colour the entire cell based on state at STP – that's what the colouring of the atomic number is for. Double sharp (talk) 08:46, 24 September 2012 (UTC)
- Details, details, details. (Headslap) And non-gasseous non-metals is a bit too cumbersome, to say nothing about the clumsiness of defining the group by what it isn't. YBG (talk) 07:35, 23 September 2012 (UTC)
Looking through the literature I find that O, F, Cl, Br are the only nonmetals that are more or less consistently referred to as being 'highly reactive', or the like. In contrast, I can't get a clear highly reactive bead on any of the rest of the nonmetals:
- H is sometimes described as being very reactive in the presence of a catalyst or at elevated temperatures, and at other times is described as being relatively unreactive at room temperature.
- 'In spite of a high bond strength hydrogen is moderately reactive, combining with most non-metals and the more electropositive metals directly to give hydrides.' (Shaw 1968, p. 8)
- 'Dihydrogen is relatively inert, due its high bond energy. Its reactions usually require activation by heat or radiation energy, or the presence of a suitable catalyst.' (Eagleson 1994, p. 509)
- 'Hydrogen...is comparatively unreactive at room temperature...At higher temperatures hydrogen reacts vigorously, even explosively, with many metals and non-metals to give the corresponding hydrides.' (Greenwood & Earnshaw 1998, p. 43)
- C (as graphite) is 'chemically an inert element and reacts with other elements only at elevated temperatures or with some other from of energy input.' (Wiberg 2001, p. 792).
- N: 'In general, nitrogen is very unreactive at normal temperatures…reactivity is significantly increased by raising the temperature.' (Wiberg 2001, p. 600).
- Oxygen:
- 'Breathing air has its drawbacks. Oxygen is a highly reactive molecule, producing molecules of peroxide (O22–) and superoxide (O2–) after it reacts during respiration to produce water. These highly reactive forms of oxygen would damage the delicate biological structures within a cell. Consequently, most organisms that breathe air have evolved enzymes to deal with these dangers.' (Arber, Garner & Hasnain 1988, p. 50)
- 'Considering the high reactivity of oxygen, its existence as free O2-gas in the atmosphere is possibly the result of constant new O2 production and addition. Withou replenishment by photosynthesis, our atmospheric oxygen may be consumed in about 3,000 years due to oxidation processes in the Earth's crust...' (Scharpenseel & Becker-Heineman 1990, p. 5)
- Oxygen:
- P is sometimes described as being highly reactive (in the form of white P) until it is pointed out that black P, which is the most thermodynamically stable form of P, is almost inert.
- S is occasionally described as being very reactive, and at other times as being, for example, almost inert in comparison to oxygen.
- '...sulfur is displaced slowly from a hydrogen sulfide solution by iodine.' (Hogg & Bickel 1941, p. 426)
- 'Sulfur should be a typical nonmetallic substance, but since the nonmetallic properties decrease as the atomic weight increases we should expect sulfur to be less active than oxygen. This decrease in chemical activity is so evident that we shall do well to contrast the activity of oxygen with the chemical inertia of sulfur.' (Hopkins & Bailar 1956, p. 258)
- 'Elementary sulfur is quite reactive, even at ordinary temperatures, although generally less so than oxygen.' (Nebergall, Holtzclaw & Schmidt 1963, p. 347)
- 'Sulfur is a very reactive element especially at slightly elevated temperatures (which presumably facilitates cleavage of S–S bonds). It unites directly with all elements except the noble gases, nitrogen, tellurium, iodine, iridium, platinum and gold, though even here compounds containing S bonded directly to N, Te, I, Ir, Pt and Au are known.' (Greenwood & Earnshaw 1998, p. 662)
- 'Sulfur is not regarded as a particularly good oxidizing agent.' (Rogers 2011, p. 504)
- Se: The Simple English Wikipedia describes Se as being 'not very reactive'.
- I: Being a halogen, I is mostly described as being chemically very active. However the reactivity of iodine is sometimes qualified—explicitly or implicitly—when compared to that of the other halogens:
- 'In its chemical properties iodine shows a general resemblance to the other halogens, although it is much less [italics added] active.' (Lowry & Cavell (1944, p. 297).
- 'By analogy one would predict that iodine would react with water as do bromine and chlorine...This reaction does take place but the extent of the reaction is negligible since iodine is too insoluble and too unreactive...Iodine is too unreactive to displace most of those elements which are displaced by bromine and chlorine.' (Hogg & Bickel 1941, p. 426)
- Sidgwick (1950) describes F as 'quite exceptionally reactive' (p. 1101); Cl as 'extremely reactive, although less so than F' (p. 1139); and Br as 'very reactive' (p. 1141). Tellingly (in my view) he has nothing to say about the reactivity of I.
- 'Bromine is a fairly strong oxidizing agent' (Brown & Sallee 1963, p. 311); 'Iodine is a mild oxidizing agent…' (Munn 2000, p. 379); 'Fluorine, chlorine and bromine will all oxidize Fe2+ (aq) to Fe3+ (aq)…Iodine, however, is such a weak oxidizing agent [italics added] that it cannot remove electrons from iron(II) ions to form iron(III) ions.' (Hill & Holman 2000, p. 230). These observations speak to the relatively modest reactivity of iodine since, for a nonmetal, reactivity is usually gauged by its ability to serve as an oxidizing agent (Brady & Senese 2009).
- In discussing halogenation, Eagleson (1994, p. 477) says that fluorination is often explosive whereas iodination is the most difficult form of halogenation due to 'the low reactivity of iodine' (Cl and Br effectively being in-between, and comparable in reactivity).
- 'Chlorine is also very reactive, but iodine is only moderately reactive.' (Hill 1999, p. 220).
- The relatively low reactivity of iodine means that HI, for example, dissociates at moderate temperatures into hydrogen and iodine (Wiberg 2000, p. 271). At higher temperatures even the more stable iodides show appreciable dissociation (Mills 1968, p. 296).
- 'In its chemical properties iodine shows a general resemblance to the other halogens, although it is much less [italics added] active.' (Lowry & Cavell (1944, p. 297).
On the above basis, it seems to me that a reasonable argument can be made for distinguishing between three categories of nonmetals:
Reactive nonmetals (7): H, C, N, P, S, Se, I
Highly reactive nonmetals (4): O, F, Cl, Br
Noble gases (6): He, Ne, Ar. Kr, Xe, Rn
The benefits of such a taxonomy, as I see it, are: It gets rid of the execrable term 'Other nonmetals'. It eliminates any difficulties with hydrogen. It preserves the current three-fold categorization of the nonmetals: less reactive | more reactive | nonreactive. It is grounded in the literature. It doesn't disturb the current colour scheme. Sandbh (talk) 07:29, 29 September 2012 (UTC)
- Arber J, Garner D & Hasnain S 1988, 'X-rays highlight metals in biology', New Scientist, 21 January, pp. 48–51
- Brady JE & Senese F 1999, Chemistry: The study of matter and its changes, John Wiley & Sons, New York
- Brown GH & Sallee EM 1963, Quantitative chemistry, Prentice-Hall, New York
- Eagleson M 1994, Concise encyclopedia of chemistry, Walter de Gruyter, Berlin
- Greenwood NN & Earnshaw A 2002, Chemistry of the elements, 2nd ed., Butterworth-Heinemann, Oxford
- Hill G 1999, GCSE science, 2nd ed., Letts Educational, London
- Hill G & Holman J 2000, Chemistry in context, 5th ed., Nelson Thornes, Cheltenham
- Hogg JC & Bickel CL 1941, Elementary general chemistry, D Van Nostrand Company, New York
- Hopkins BS & Bailar JC 1956, General chemistry for colleges, 5th ed., DC Heath and Co., Boston
- Lowry TM & Cavell AC 1944, Intermediate chemistry, 3rd ed., Macmillian and Co., London
- Mills JF 1968, 'Iodine', in CA Hampel (ed.), The encyclopedia of the chemical elements, Reinhold Book Corporation, New York, pp. 290–299
- Munn B 2000, 'Molecular electronics', in N Hall (ed.), The new chemistry, Cambridge University Press, Cambridge, pp.375–406
- Nebergall WH, Holtzclaw HF & Schmidt FC 1963, College chemistry with qualitative analysis, 2nd ed., DC Heath, Boston, MA
- Parsons C & Dover C 1966, The elements and their order: Foundations of inorganic chemistry, Sampson Low, Marston & Co., London
- Rogers GE 2011, Descriptive, inorganic, coordination, & solid-state chemistry, 3rd ed., Brooks/Cole Cengage Learning, Belmont CA
- Scharpenseel HW & Becker-Heineman P 1990, 'Overview of the greenhouse effect' in HW Scharpenseel, M Schomaker & A Ayoub (eds), Soils on a warmer Earth, Elsevier, Amsterdam, pp. 1–14
- Schlesinger HI 1937, General chemistry, 3rd ed., Longmans, Green and Co., New York
- Shaw BL 1968, Inorganic hydrides, Pergamon, Oxford
- Sidgwick NV 1950, The chemical elements and their compounds, vol. II, Oxford University Press, London
- Wiberg N 2001, Inorganic chemistry, Academic Press, San Diego
- Interesting analysis. But I still feel that the categories should correspond to the most top-level categories of elements. The topmost level would be "metals", "metalloids" and "nonmetals". "Metals" and "nonmetals" are too large and are broken down further, but still we restrict this to the top two levels to prevent things from being too messy (and also removing problems with division methods not being commonly used or not completely agreed on). The way I see it, "less reactive nonmetals" and "more reactive nonmetals" are third-level categories, parts of the second-level category "typical nonmetals", which is part of the first-level category "nonmetals". Besides, looking at what you have written, there seems to be some disagreement on whether H qualifies as being a "more reactive nonmetal". Double sharp (talk) 07:52, 30 September 2012 (UTC)
Here's what I mean:
| H | He | |||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | |||||||||||
| ||||||||||||||||||||||||||||||||||||||||||
Sandbh (talk) 09:13, 30 September 2012 (UTC)
- Double sharp: H is relatively inert due to its high bond energy. While H can be very reactive if the conditions are right (heat or a suitable catalyst), such conditional reactivity, as I understand the descriptive chemistry literature, doesn't gel with 'high reactivity' status. Categorizing H as a reactive nonmetal—for sure. Categorizing H as a highly reactive nonmetal—akin to O, F, Cl, Br—no. Sandbh (talk) 13:38, 30 September 2012 (UTC)
- You're starting to convince me. :-) What does everyone else think? Double sharp (talk) 09:49, 1 October 2012 (UTC)
- Looks nice to me, if the community is willing to listen to me after my gaffe about solid nonmetals :) I'd still like the templates to mention the group names (halogens, etc.) somehow or other — as groups, not as categories. YBG (talk) 03:42, 2 October 2012 (UTC)
- I've added a few more (indented) quotes about H, O, S and I. Sandbh (talk) 01:36, 7 October 2012 (UTC)
- Looks nice to me, if the community is willing to listen to me after my gaffe about solid nonmetals :) I'd still like the templates to mention the group names (halogens, etc.) somehow or other — as groups, not as categories. YBG (talk) 03:42, 2 October 2012 (UTC)
- You're starting to convince me. :-) What does everyone else think? Double sharp (talk) 09:49, 1 October 2012 (UTC)
Predictions
Since the "halogens" category is going to disappear no matter what proposal we adopt, what category (predicted, of course) should we put elements 117, 167, and 217 into in {{Compact extended periodic table}}? Would 118, 168, and 218 be reasonably able to be predicted as being noble gases? Double sharp (talk) 15:20, 2 October 2012 (UTC)
- Is anyone looking at this sub-subsection? Double sharp (talk) 07:20, 26 October 2012 (UTC)
- A further problem is that predictions don't help, because those would follow the relativistic Pyykkö model, rather than the non-relativistic Aufbau principle–organized extended periodic table. (Should we change the compact extended table to a Pyykkö one?) Double sharp (talk) 12:01, 16 November 2012 (UTC)
- Yeah, good one (and a good two is there too). First of all, I agree, we should switch to Pyykkö. It's, we'll, actual science. It's the most common model nowadays among those who really try to get it. I've seen that in multiple source studying the problem. More often than any other thing about that (in fact, I've never seen anything different).
- I also have had the idea we could replace the symbols like Utb with numbers like 132 in the extended table. It's just not comfortable to go through with that many symbols which mean nothing. Numbers make it a much easier understanding. If you think there are rules... Well, yes, there are, but it's more a call for common sense and good navigation than rushing that system.
- And yeah, guys, let's try it once again. How about removing the current Halogens section with simple annexing by Other nonmetals before there is an agreement if it could be split?
- If the three have their support, we could think about coloring more closely.--R8R Gtrs (talk) 13:02, 16 November 2012 (UTC)
- (Well, At should probably be labelled as a metalloid instead of a nonmetal.) Double sharp (talk) 14:07, 16 November 2012 (UTC)
- A further problem is that predictions don't help, because those would follow the relativistic Pyykkö model, rather than the non-relativistic Aufbau principle–organized extended periodic table. (Should we change the compact extended table to a Pyykkö one?) Double sharp (talk) 12:01, 16 November 2012 (UTC)
| g p |
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||||||||||||||||||||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||||||||||||||||||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||||||||||||||||||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||||||||||||||||||||||||
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | 113 | Fl | 115 | Lv | 117 | 118 | |||||||||||||||||||||||||||||||||
| 8 | 119 | 120 | * | 141 | 142 | 143 | 144 | 145 | 146 | 147 | 148 | 149 | 150 | 151 | 152 | 153 | 154 | 155 | 156 | 157 | 158 | 159 | 160 | 161 | 162 | 163 | 164 | 139 | 140 | 169 | 170 | 171 | 172 | ||||||||||||||||||||||||||||||||
| 9 | 165 | 166 | 167 | 168 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | 121 | 122 | 123 | 124 | 125 | 126 | 127 | 128 | 129 | 130 | 131 | 132 | 133 | 134 | 135 | 136 | 137 | 138 | |||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
- I've coloured At as a metalloid for reasons given above. For my colouring of 117 and 171 as metalloids, I've added quotes from Haire below:
- 117:
- "In element 117, the 1− oxidation state becomes less important than that of the lighter group 17 halide ions due to the destabilization of the 7p3/2 orbital....the 3+ state should be at least as important as the 1− state...." (Haire, p. 1728)
- "Analogous to its lighter homologs, Element 117 should form dimers X2." (Haire, p. 1728)
- 171:
- "The energies of these orbitals [9p1/2 and 8p3/2] are so close to each other that this situation is analogous to the nonrelativistic p-shell in the 3d period. Thus, the common oxidation states of elements 167 to 179 will be 3+ to 6+. Element 171 is expected to have many states from 1− to 7+, as do halogens. H(171) would form due to its high electron affinity of 3.0 eV. Compounds with F and O are also expected." (Haire, p. 1733)
- 117:
- Double sharp (talk) 14:07, 16 November 2012 (UTC)
- For 118, Haire always compares 118 with its "lighter homologs within the group" (Haire, p. 1728), Xe and Rn, implying that it is a "noble" gas (although it should be more reactive than Xe or Rn). For 172, Haire writes "Element 172 might be a noble gas similar to Xe due to the similar values of their IPs [ionization potentials]. The major difference is that element 172 is predicted to be a liquid or a solid at normal temperatures because of its large atomic weight." (Haire, p. 1733).
- Haire, Richard G. (2006). "Transactinides and the future elements". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. ISBN 1-4020-3555-1.
{{cite book}}: CS1 maint: ref duplicates default (link) Double sharp (talk) 14:12, 16 November 2012 (UTC)
- This presentation by J. V. Kratz contains another PT extended to the eighth period. He puts 139 and 140 with the other superactinides and moves 169–172 down to the ninth period to join 165–168. This conforms more to Haire and Fricke's predicted electron configurations for the elements until 172. Double sharp (talk) 14:30, 16 November 2012 (UTC)
- (off topic) This presentation also states that Fl should be more reactive than Cn(!), and that they should form a new category of elements due to the electron shell closure (gaseous metals). It gives P. Armbruster's prediction that the next closed proton shell is at Z = 122. It also states that Cn to 118 are stabilized against fission because their nuclei are oblate, implying that 119 and 120 should be (according to the presentation you left on my talk page) the last elements we can synthesize for now without new methods. The cross-sections of reactions synthesizing 119 and 120 should also give important information about the end of the periodic table. Double sharp (talk) 14:35, 16 November 2012 (UTC)
- Haire, Richard G. (2006). "Transactinides and the future elements". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. ISBN 1-4020-3555-1.
- I've coloured At as a metalloid for reasons given above. For my colouring of 117 and 171 as metalloids, I've added quotes from Haire below:
Hey, I just realized that a switch to Pyykkö means we need to go alter heavier homologs from like Upo to Uhn in articles like hassium (see infobox). Can someone give it a try?--R8R Gtrs (talk) 17:06, 18 November 2012 (UTC)
 Done Double sharp (talk) 04:06, 19 November 2012 (UTC)
Done Double sharp (talk) 04:06, 19 November 2012 (UTC)
- Gaseous metals, work of relativistic effects, are not worth a separate group we (we) could use a new color for. Wouldn't probably be if they were available in macroscopic amounts. There is a group of liquid metals (of one liquid metal, okay), a child of relativity as well-- and none gives a damn for real classification. About Kratz, I don't think he really meant period nine for those six (9p are energetically less stable than 8p, it's gotta be so). I rather think it has a little different meaning (see also the normal, 118 elements table with some super heavies also torn apart for a little).
- I like your long table.--R8R Gtrs (talk) 19:26, 16 November 2012 (UTC)
- Of course, but relativistic effects make them closer to each other. Besides, we are comparing 9p1/2 and 8p3/2: 9p1/2 would be stabilized while 8p3/2 is destabilized, moving their energy levels closer to each other.
- Yes, gaseous metals don't deserve a new colour. But I did change Cn from solid to gas due to this. (I didn't change Fl, since its state and chemical properties seem to be more disputed. More discussion would be nice.) Double sharp (talk) 14:33, 20 November 2012 (UTC)
![]() Done {{Compact extended periodic table}} now stops at 172 and uses the Pyykkö model. I changed the systematic names to simple atomic numbers: although using the systematic symbols is more consistent with the non-extended table, it's easier for the reader if the number is used, since the reader might not know what the systematic symbols mean. Double sharp (talk) 06:19, 17 November 2012 (UTC)
Done {{Compact extended periodic table}} now stops at 172 and uses the Pyykkö model. I changed the systematic names to simple atomic numbers: although using the systematic symbols is more consistent with the non-extended table, it's easier for the reader if the number is used, since the reader might not know what the systematic symbols mean. Double sharp (talk) 06:19, 17 November 2012 (UTC)
Categorisation of 117, 118, 171, 172
- @Sandbh: Do you agree with my classification of 117 and 171 as metalloids based on Haire's predictions about their properties (quoted above)? Double sharp (talk) 11:12, 19 November 2012 (UTC)
- Your question will take me a little longer to respond to; have started to think about it; need to look closer at the figures---off the cuff I'd be surprised if 171 was anything but a metal Sandbh (talk) 11:00, 20 November 2012 (UTC)
- Yes, I also find it surprising. I chose a metalloid status for 171 due to Haire's statement that it would form a −1 state and compares it with halogens, but he doesn't mention how stable or common the −1 state would be. Double sharp (talk) 14:29, 20 November 2012 (UTC)
- Based on an initial consideration of the limited data on the elements in question, and with a big, big caveat as to uncertainty surrounding relativistic effects, it seems to me that 117 could plausibly (and conservatively) be predicted to be a metalloid (see also Topich 2000 p. 13; Emsely 2003, p. 468); 171 a poor metal; and 118 and 172 = reactive nonmetals. I've made these suggestions largely based on simple (non-relativistic) Goldhammer-Herzfeld criterion ratio extrapolations, together with the impact of the predicted semi-closed valence shell configurations of 117 and 118 as described in their articles. On the basis of current to near-future technology I'm also hugely sceptical that 171 and 172 could ever even be synthesized or stabilized for long enough to observe their properties.
- Grateful for the thoughts of others as to the reasonableness of these predictions. Sandbh (talk) 03:32, 24 November 2012 (UTC)
- Topich J, Solutions manual: Chemistry (McMurray Fay) 2000, 3rd ed., Prentice Hall, Upper Saddle River, NJ: 'Element 117 might have the properties of a semimetal.'
- Emsley J 2003, Nature's building blocks: An A-Z guide to the elements, Oxford University Press, Oxford: 'From its position in the periodic table, in group 17 below astatine, this element [117] should have the physical properties of a halogen or a metalloid…'
- When you are predicting the properties of elements with such high atomic number, you have to consider relativistic effects. Here's B. Fricke's and W. Greiner's paper. Fricke gives this table:
- Yes, I also find it surprising. I chose a metalloid status for 171 due to Haire's statement that it would form a −1 state and compares it with halogens, but he doesn't mention how stable or common the −1 state would be. Double sharp (talk) 14:29, 20 November 2012 (UTC)
| g p |
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||||||||||||||||||||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||||||||||||||||||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||||||||||||||||||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||||||||||||||||||||||||
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | 113 | Fl | 115 | Lv | 117 | 118 | |||||||||||||||||||||||||||||||||
| 8 | 119 | 120 | * | 141 | 142 | 143 | 144 | 145 | 146 | 147 | 148 | 149 | 150 | 151 | 152 | 153 | 154 | 155 | 156 | 157 | 158 | 159 | 160 | 161 | 162 | 163 | 164 | ||||||||||||||||||||||||||||||||||||||
| 9 | 165 | 166 | 167 | 168 | 169 | 170 | 171 | 172 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | 121 | 122 | 123 | 124 | 125 | 126 | 127 | 128 | 129 | 130 | 131 | 132 | 133 | 134 | 135 | 136 | 137 | 138 | 139 | 140 | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
- (Well, he puts Y over La, but we have agreed to put Y over Lu.) Most presentations seem to use this table.
- Period 8: "...the elements Z = 115, 116, and 117 will have 1, 2, and 3 as their normal oxidation states. The higher oxidation states will be possible only for strong oxidizers. [Its ionization energy should be around 8.2 eV and it should show −1, +1, and +3 oxidation states.] The "noble gas" at Z = 118 will be a very weak noble gas in the sense of He and Ar but as well in comparison to Xe and Rn. The ionization energy is so small [around 9.0 eV] that normal covalent bondings are expected with oxidation states of 4 and 6. The extrapolation of Cunningham expects a boiling point of −15 °C so that it will be nearly a "noble fluid"."
- Period 9: Fricke and Greiner state that period 9 will be very much like period 3. "Between Z = 167 and Z = 172 the 9p1/2 and 8p3/2 electrons will be filled in and it is quite an accident that the energy eigenvalues are so close together that a p shell will result very analogous to the 2nd and 3rd period...Z = 171 is expected to have many possible oxidation states between −1 and 7. Element Z = 172 will be a very good noble gas with a closed p shell outside. The ionization energy of this element shown in Fig. 8, is very near to the value of Xe, so that it might be very similar to this element. The great difference Xe and Z = 172 will only be that element Z = 172 is expected to be fluid or even solid because of its large atomic weight." They predict 171 to show mainly the +3 and +7 states, have a first ionization energy of 10.2 eV, a metallic or covalent(?) radius of 165 pm, and a density of 16 g/cm3. 172 is predicted to show the 0, +4, +6, and +8 states, have a first ionization energy of 11.3 eV, a metallic or covalent(?) radius of 220 pm, and a density of 9 g/cm3. They caution that the values for the radii and densities are only first approximations.
- So what do you think, based on this and Haire's info (above)? (Pyykkö does not comment on these elements.) Looking at the ionization energies, 117's ionization energy of 8.2 eV is close to that of boron, and 118's value of 9.0 eV is close to those of Cd, Te, and Pt. 171's ionization energy of 10.2 eV is close to that of sulfur, and 172's value of 11.3 eV is close to that of carbon. Double sharp (talk) 06:46, 24 November 2012 (UTC)
- Also, the atomic masses of elements 117, 118, 171, and 172 should be 316, 319, 500, and 504 respectively. Double sharp (talk) 06:52, 24 November 2012 (UTC)
- If the above link doesn't work, this one should. See also this. Double sharp (talk) 11:38, 24 November 2012 (UTC)
- E117 we agree on metalloid. For E171 extrapolated GH criterion ratio is so high that metallic status is near certainty. Only impact of relativity would be to increase likelihood that E171 was a metal (like what happens with Po). Even if it still showed –1 oxidation state that would not be unusual behaviour for a poor metal. Even transition metals can do this (Pt, Au). Nothing in Fricke contradicts. For E118 Fricke etc also say it 'will be a very bad noble gas with possibly very high oxidation states and normal chemical compounds' which I think could reasonably be interpreted as = reactive nonmetal. For E172 they predict outer shell config of 9s29p28p4 with 8p forming a closed subshell. E172 therefore expected to be very good noble fluid or solid, which is a reasonable conclusion. Sandbh (talk) 12:20, 24 November 2012 (UTC)
- If the above link doesn't work, this one should. See also this. Double sharp (talk) 11:38, 24 November 2012 (UTC)
- Also, the atomic masses of elements 117, 118, 171, and 172 should be 316, 319, 500, and 504 respectively. Double sharp (talk) 06:52, 24 November 2012 (UTC)
Thanks for the paper! Here's what I can conclude after reading this:
118 will be a very bad noble gas. Some scientific papers require reading between the lines to correctly interpret them when it jumps from calculations to conclusions. In our case, they still do not refuse to call it a noble gas, but if they called it so no more, it would sound like a sensation back then probably. At least, this would be more interesting to write about. They do mention the reduction of this nobility character, but this shows us they do not think it to be a regular metal. At least not sure in that. The "normal compounds" is a vague term -- is radon difluoride a normal compound? If the answer is yes (which I would pick), then it's not a point-- we still call radon noble. Anyway, such a move (recoloring 118) would be quite bold and thus require more backup proof. Note also it's a part of the FT "Noble gases."
- The abstract of the Fricke paper says that 172 is a noble gas but does not say anything about the nature of 118—why not? My guess is because they doubted its bona fides as a noble gas but, in agreement with R8R Gtrs, it would have been too big a call in 1971 to call it something other than a noble gas, without further research.
- At two points in the article they refer to 118 as 'the "noble gas" at Z = 118.' The quotes around "noble gas" imply, as I read it, that they regarded 118 as a noble gas only in name and not in substance. As noted, they described 118 as a very bad noble gas and a very weak noble gas even in comparison to Xe and Rn. Not just 'bad' or 'weak' but 'very' as in 'really or truly entitled to the name or designation' (Oxford English Dictionary). And we know Xe forms hundreds of compounds. And cationic behaviour of Rn has been reported.
- Our own article on 118 notes that (emphasis added):
- 'Following the periodic trend, ununoctium would be expected to be slightly more reactive than radon. However, theoretical calculations have shown that it could be quite reactive, so that it probably cannot be considered a noble gas. In addition to being far more reactive than radon, ununoctium may be even more reactive than elements flerovium and copernicium. The reason for the apparent enhancement of the chemical activity of ununoctium relative to radon is an energetic destabilization and a radial expansion of the last occupied 7p-subshell.More precisely, considerable spin–orbit interactions between the 7p electrons with the inert 7s2 electrons, effectively lead to a second valence shell closing at flerovium, and a significant decrease in stabilization of the closed shell of element 118. It has also been calculated that ununoctium, unlike other noble gases, binds an electron with release of energy—or in other words, it exhibits positive electron affinity.
- Because of its tremendous polarizability, ununoctium is expected to have an anomalously low ionization energy (similar to that of lead which is 70% of that of radon and significantly smaller than that of flerovium) and a standard state condensed phase.'
- On the basis of 118's currently understood properties, a colour change is reasonable. The arguments for so doing are stronger than the arguments for not so doing. Further comments on 171 to follow. Sandbh (talk) 00:02, 25 November 2012 (UTC)
- Looking at the ionization potentials of the noble gases (from ionization energies of the elements (data page):
- In comparison, Fl's ionization potential is around 8.54 eV, and Cn's is around 11.97 eV. Pb's value is a lot lower at 7.417 eV. The 118 value looks like it continues the trend down from Kr reasonably well, and is very close to that of Os. Double sharp (talk) 07:42, 25 November 2012 (UTC)
171 can be an actual metalloid. My first thought after reading the article was even "It's a nonmetal!", but it's probably not the case. I'll try to explain how this went on, and you try to correct me, ok? So, the p electrons split into two parts, the faster the higher Z is. See graph. (These effects alter chem of the elements by 10% and 18-33%, respectively, while the figure for iodine is 1%, and for bromine, chlorine, and fluorine, this is too small to consider, much like we don't consider quantum stuff for macroscopic objects, which is a very very very close approximation, or mass loss during a chemical reaction). The 9p(1/2) electrons are too stabilized and the 8p(3/2) are too destabilized, so they actually very close in energy, and form a p shell of six electrons, which behaves like a single one (despite its contents!). Simply extrapolating that data, we could say that the p energy level is not going to be too close to zero, so it would take a strong oxidizer to get high oxidation states. And also the -1 state seems to be more likely than for 117, both by imagination and according to calculations. 172 is reasonable to stay in its color as well.--R8R Gtrs (talk) 13:01, 24 November 2012 (UTC)
- Hmm. I don't understand how 171 could be regarded as a metalloid. Around a quarter of metals exhibit negative oxidation states yet we don't classify any of these as metalloids. On what basis are you suggesting 171 is metalloid? Sandbh (talk) 04:39, 25 November 2012 (UTC)
- They don't exhibit negative oxidation states as common states (except Sn and Po) – see List of oxidation states of the elements. We need to consider how important the −1 state is for its chemistry. Haire states that "In element 117, the −1 oxidation state becomes less important than that of the lighter group 17 halide ions due to the destabilization of the 7p3/2 orbital. The EA of element 117 is the smallest in the group (2.6 eV as predicted by Cunningham, 1969 and 1.8 eV as given by Waber et al., 1969). Therefore, the +3 state should be at least as important as the −1 state, so that element 117 might resemble Au3+ in its ion-exchange behavior in halide media." OTOH, he writes for 171: "Element 171 is expected to have many states from −1 to +7, as do halogens. H(171) would form due to its high electron affinity of 3.0 eV (Fricke et al., 1971). Compounds with F and O are also expected." Looking at the electron affinity values of the halogens from electron affinity (data page):
- it seems clear that due to its high electron affinity (close to that of I and At), the −1 state of 171 is going to be much more important than that of 117. We must also note that relativistic effects are causing 9p1/2 and 8p3/2 to behave together as one p-shell, and so should not really be compared with how it affects p-shells made of np1/2 and np3/2 subshells with the same n. This p-shell is predicted to behave like the 2p and 3p ones, so that the full shell of 172 is stabilized (making it a very good noble fluid/solid) and making the −1 state of 171 more stable. Double sharp (talk) 07:17, 25 November 2012 (UTC)
- Your line of reasoning is plausible. However I am concerned about the EA figure for 171 of 3.0 understood to be given by Fricke et al 1971. Fricke etc give no such figure, as I recall. This is important given the very high EA of gold, due to the potential stability of the filled 6s subshell (look at how 'inert' mercury, with a filled 6s subshell, is), yet even so gold shows no great predilection for the –1 oxidation state. In other words I am querying the validity of the relatively high EA figure given for 171. Sandbh (talk) 12:08, 30 November 2012 (UTC)
- Oddly, Haire gives this figure, attributing it to Fricke et al., when it isn't given there... Double sharp (talk) 02:12, 1 December 2012 (UTC)
- In Haire's bibliography, he gives two papers by Fricke et al. dating from 1971. One of these is the one I linked to. The other is from Actinides Rev., 1, 433–85. Could this be what he's citing? Double sharp (talk) 02:37, 1 December 2012 (UTC)
- If he is citing the other paper it looks like this will be quite hard to get a copy of (try Googling "Actinides Reviews"---very discouraging results) Sandbh (talk) 12:21, 1 December 2012 (UTC)
- I found it! :-D Double sharp (talk) 12:26, 1 December 2012 (UTC)
- Stunned amazement. Dude, you rock! Sandbh (talk) 12:38, 1 December 2012 (UTC)
- It gives the figure of 3.0 eV for 171's EA, and even predicts the properties of the early period 10 elements – the "eka-superactinides" – until 184 (although not placing them into a PT). Double sharp (talk) 12:30, 1 December 2012 (UTC)
- OMG and he invokes HSAB principles! Haven't finished reading it yet. May not be able to give feedback until later. Sandbh (talk) 12:46, 1 December 2012 (UTC)
- It also states that 171− would be a hard base (comparable to Cl−, although it gives Cl's EA as around 3.0 eV(!)), and that H171 would really be a hydrogen halide (unlike AtH and 117H, which are "halogen" hydrides). 172 should be a strong Lewis acid. In this paper, Fricke and Waber dare to call 118 an "in-noble gas", although they then call it a "very weakly noble gas" later in the same sentence. Double sharp (talk) 12:52, 1 December 2012 (UTC)
- Hmm. Given 171's similarities to Cl and Br here, shouldn't it really be coloured as a nonmetal?
- It also states that 171− would be a hard base (comparable to Cl−, although it gives Cl's EA as around 3.0 eV(!)), and that H171 would really be a hydrogen halide (unlike AtH and 117H, which are "halogen" hydrides). 172 should be a strong Lewis acid. In this paper, Fricke and Waber dare to call 118 an "in-noble gas", although they then call it a "very weakly noble gas" later in the same sentence. Double sharp (talk) 12:52, 1 December 2012 (UTC)
- OMG and he invokes HSAB principles! Haven't finished reading it yet. May not be able to give feedback until later. Sandbh (talk) 12:46, 1 December 2012 (UTC)
- It gives the figure of 3.0 eV for 171's EA, and even predicts the properties of the early period 10 elements – the "eka-superactinides" – until 184 (although not placing them into a PT). Double sharp (talk) 12:30, 1 December 2012 (UTC)
- Stunned amazement. Dude, you rock! Sandbh (talk) 12:38, 1 December 2012 (UTC)
- I found it! :-D Double sharp (talk) 12:26, 1 December 2012 (UTC)
- If he is citing the other paper it looks like this will be quite hard to get a copy of (try Googling "Actinides Reviews"---very discouraging results) Sandbh (talk) 12:21, 1 December 2012 (UTC)
- In Haire's bibliography, he gives two papers by Fricke et al. dating from 1971. One of these is the one I linked to. The other is from Actinides Rev., 1, 433–85. Could this be what he's citing? Double sharp (talk) 02:37, 1 December 2012 (UTC)
- Oddly, Haire gives this figure, attributing it to Fricke et al., when it isn't given there... Double sharp (talk) 02:12, 1 December 2012 (UTC)
| g p |
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||||||||||||||||||||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||||||||||||||||||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||||||||||||||||||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||||||||||||||||||||||||
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | 113 | Fl | 115 | Lv | 117 | 118 | |||||||||||||||||||||||||||||||||
| 8 | 119 | 120 | * | 141 | 142 | 143 | 144 | 145 | 146 | 147 | 148 | 149 | 150 | 151 | 152 | 153 | 154 | 155 | 156 | 157 | 158 | 159 | 160 | 161 | 162 | 163 | 164 | ||||||||||||||||||||||||||||||||||||||
| 9 | 165 | 166 | 167 | 168 | 169 | 170 | 171 | 172 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | 121 | 122 | 123 | 124 | 125 | 126 | 127 | 128 | 129 | 130 | 131 | 132 | 133 | 134 | 135 | 136 | 137 | 138 | 139 | 140 | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Which version of extended periodic table?
Also, after reading the text, I realized that the two schemes in this section is exactly the same, interpreted differently. I like the first one more, and if anyone thinks so not, I can write my arguments.--R8R Gtrs (talk) 13:01, 24 November 2012 (UTC)
- I want to hear your arguments. I personally prefer Fricke's (the second one), though. The 5g, 6f, 7d, and 8p1/2 electrons are all being filled in at once in the superactinide series. The 8p1/2 electrons are filled already at 121 to 127, and I don't see why 139 and 140 should be split apart simply to account for the 8p1/2 electrons if they are already there earlier and those two elements are filling the 5g, 6f, and 7d subshells. Pyykkö's argument that the 1385+ and 1386+ ions do not contain 8p1/2 electrons doesn't strike me as particularly convincing – after all, the Eu2+ ion doesn't contain 6s electrons. It seems to just indicate that the 8p1/2 electrons have lower ionization energies than the 5g and 8s ones. For 169–172, the 8p3/2 electrons actually have slightly higher energies than the 9p1/2 electrons (Pyykkö says so, citing Fricke et al. as his reference), and they behave like a single p-subshell analogous to the 2p and 3p subshells. So I think it is better to show how they behave like a single p-subshell due to the fact that their energies are very similar by putting 169–172 in period 9 rather than following the orbitals by putting them in period 8, which also obscures the result of the calculation that the 8p3/2 subshell has higher energy than the 9p1/2 one. Double sharp (talk) 14:48, 24 November 2012 (UTC)
Interestingly, the Fricke paper seems to state that 157 will be in group 3, 158 will be in group 4, etc., and that 164 will really be a group 10 element, despite putting them in groups 5–12 in the periodic table figure given. Double sharp (talk) 14:35, 24 November 2012 (UTC)
However, Haire states that the 9s and 9p1/2 states will be readily available for hybridization, so the fact that 8s are no longer valence electrons might not actually change much. Double sharp (talk) 05:08, 25 November 2012 (UTC)
- You know, I did some more thinking. I think it depends on how we define a period. I always thought that the main feature of a period was a constant n+l value, with the exception of the s-block. If a period is a alkali metal--noble gas row, then it's clearly 119--172 without 165-168. I'm ready to accept the need of another two superactinides if I have to. But it still seems weird to me that when we fill up the ninth period when our eight is incomplete, and the pieces being filled (169-172) certainly belong to it (filling the 8p(3/2) orbital), even if the chem is identical. Also, we're finishing a period with a metal for little of reason. But we get thereby a hole of two between 164 and 169, someone needs into there.
- You interpret the term "period" slightly differently. I don't know which is right: I'm only using intuition and the known data. That's a more a chem philosophy question anyway.
- Sorry for a temporary inability to add some electron configuration arguments (I wanna have my sleep), your arguments are entirely valid and correct. The position is reasonable as well: those Germans chose it for a reason as well. I'm just trying to take a different approach. But I think that debate should ultimately lead to the question "what is a period?" rather than anything. And I understand your position, won't argue if we choose the philosophy to get your variant.
- (Also, is what we're discussing here properly reflected on the period 8 element page?)
- And at last, I'd love to have a look at Pyykko's paper again (maybe tomorrow), can you bump me a link? (I'm sure I had it somewhere, but don't remember where, or was it another paper?)--R8R Gtrs (talk) 20:03, 24 November 2012 (UTC)
- It's not properly reflected on the period 8 element article (it still uses the non-relativistic 119–168 period 8), but I'll try to fix it to talk about the relativistic period 8 instead sometime next week.
- Here's a link to Pyykkö's paper.
- Yes, finishing a period with a metal is unprecedented in the periodic table. But Fricke et al. predict 164 to be a very good noble metal and to have close similarities (e.g. ionization energy and electron configuration) to 118. Fricke et al. also note that He has a filled s-subshell, Ne–118 have filled p-subshells and 164 has a filled d-subshell. (Haire predicts something different to happen – see Group 12 element#Extensions. Here's another paper solely predicting the properties of 164.) Double sharp (talk) 05:08, 25 November 2012 (UTC)
- Helium is not an analogy (much like heavy Pyykko's ions are also weak an argument), different reasons. Whatever. I think it's a philosophy question and not a data one (the data is quite consistent). Thanks for the paper, also! And... Well, given it's a question of arbitrary judgment, I'd use Pyykko's idea as more modern. Have no more principally new arguments; I try not to repeat the same thing (hope you won't either). Well, whichever is fine.--R8R Gtrs (talk) 18:38, 25 November 2012 (UTC)
- I don't really think there's any strong reason for ending period 8 at 164, except that 164 is a good noble metal and the next element is an alkali metal. But I do find it better than the alternative (Pyykkö) for reasons I've mentioned previously (and so will not repeat). (Also, the presentation on 114 and 112 being gaseous metals that I linked to earlier uses Fricke's table.) Double sharp (talk) 03:51, 26 November 2012 (UTC)
- I changed the template to Fricke as being more consistent with the predicted chemical properties of the elements. (I should rewrite many of the extended PT articles to make them relativistic soon; otherwise, the different forms of the extended PT used on WP will probably confuse readers.) Double sharp (talk) 08:56, 27 November 2012 (UTC)
- P.S. Fricke et al. also imply that 154 will be a very good noble metal because its 6f shell is filled(!) (contradicts the electron configuration table I posted below, but then the 5g and 6f shells are buried deep inside) and has no electrons with wave functions outside the (relativistically very stabilized and thus chemically inactive) 8s and 8p1/2 shells, even stating that "[t]his would mean that this element would be chemically very inactive with a behaviour of a noble gas."(!) Double sharp (talk) 14:12, 28 November 2012 (UTC)
- P.P.S. Fricke et al. also, contrary to the electron configuration table I posted below, state that the valence electron configurations of 156–164 should be simply 7d2, 7d3, 7d4, ..., 7d10, and that 9s will not be filled in any orbitals. I think using Fricke et al.'s table but Haire's electron configurations is a reasonable choice – Haire's electron configurations are more recent, but there are no very substantial differences, and I think Fricke et al.'s interpretation makes more sense than Pyykkö's for reasons I've already stated above. Double sharp (talk) 14:16, 28 November 2012 (UTC)
- P.S. Fricke et al. also imply that 154 will be a very good noble metal because its 6f shell is filled(!) (contradicts the electron configuration table I posted below, but then the 5g and 6f shells are buried deep inside) and has no electrons with wave functions outside the (relativistically very stabilized and thus chemically inactive) 8s and 8p1/2 shells, even stating that "[t]his would mean that this element would be chemically very inactive with a behaviour of a noble gas."(!) Double sharp (talk) 14:12, 28 November 2012 (UTC)
- I changed the template to Fricke as being more consistent with the predicted chemical properties of the elements. (I should rewrite many of the extended PT articles to make them relativistic soon; otherwise, the different forms of the extended PT used on WP will probably confuse readers.) Double sharp (talk) 08:56, 27 November 2012 (UTC)
- I don't really think there's any strong reason for ending period 8 at 164, except that 164 is a good noble metal and the next element is an alkali metal. But I do find it better than the alternative (Pyykkö) for reasons I've mentioned previously (and so will not repeat). (Also, the presentation on 114 and 112 being gaseous metals that I linked to earlier uses Fricke's table.) Double sharp (talk) 03:51, 26 November 2012 (UTC)
- Helium is not an analogy (much like heavy Pyykko's ions are also weak an argument), different reasons. Whatever. I think it's a philosophy question and not a data one (the data is quite consistent). Thanks for the paper, also! And... Well, given it's a question of arbitrary judgment, I'd use Pyykko's idea as more modern. Have no more principally new arguments; I try not to repeat the same thing (hope you won't either). Well, whichever is fine.--R8R Gtrs (talk) 18:38, 25 November 2012 (UTC)
Nonmetal subcategories
Do I understand correctly that what is being proposed is three major categories of elements, looking like this(?):
- Metals: comprising Alkali metals | Alkaline earth metals | Inner transition metals (Ln, An, Super-An etc) | Transition metals | Post-transition metals
- Metalloids: comprising B, Si, Ge etc
- Nonmetals: comprising Other nonmetals | Noble gases
Sandbh (talk) 00:50, 17 November 2012 (UTC)
- Yes. We can do this as an initial switch, and then discuss more about how to deal with "Other nonmetals". (It's less confusing for readers, as there is then a period where the Halogens colour is unused before it takes a different meaning.) Double sharp (talk) 06:19, 17 November 2012 (UTC)
- I have a suggestion for dealing with "Other nonmetals".
- he nonmetals can simply be categorised as:
- Reactive nonmetals: H, C, N, O, P, S, Se
- Corrosive nonmetals: F, Cl, Br, I
- Noble gases: He, Ne, Ar, Kr, Xe, Rn
- he nonmetals can simply be categorised as:
- The noble gases are a no brainer.
- As per previous citations the rest of the nonmetals (including N) are plainly 'reactive' elements in comparison to the noble gases. That's fine except that the literature also commonly distinguishes between less reactive nonmetals and more reactive nonmetals. Rather than revisit old ground, the less reactive metals can simply be categorised as plain old 'reactive nonmetals' and the more reactive nonmetals as 'corrosive nonmetals' (which they certainly are, unlike the rest of the nonmetals).
- It's important to appreciate that each of the proposed categories encompasses a reactivity spectrum rather than being homogenous or necessarily discrete. For the noble gases the reactivity order is (as I recall the research): Ne, He, Ar, Kr, Xe, and Rn (the latter also reportedly showing cationic behaviour). The corrosive nonmetals are easy (hi-lo): F, Cl, Br, I. For the reactive nonmetals my hi-lo guess would be O (by a sizeable margin), H, S, C, Se, P (as black P) and N; with the reactivity of O overlapping some way into the corrosive nonmetals, but without O being recorded in the literature as a 'corrosive' nonmetal.
- I know that I've previously argued that oxygen should be categorised as a 'highly reactive' nonmetal, in the same category as F, Cl and Br, however I believe this proposal, as explained so far, is better.
- I've also previously argued that iodine is not in the same oxidising power league as its lighter congeners. However, that doesn't necessarily detract from its corrosiveness nor its toxicity. For example, I was surprised to recently learn that tincture of iodine will smoothly dissolve gold. (Nakao Y 1992, 'Dissolution of noble metals in halogen-halide-polar organic solvent systems', Journal of the Chemical Society, Chemical Communications, pp. 426–427).
- The 5th edition of Proctor and Hughes' chemical hazards of the workplace (2004) further notes that, 'Iodine in crystalline form or in strong solution is a severe skin irritant; it is not easily removed from the skin, and the lesions resemble thermal burns with brown staining. Cutaneous absorption may be significant and result in systemic symptoms and death.' (pp. 402–403)
- Aside from F, Cl, and Br none of the rest of the nonmetals are included in Proctor and Hughes, except for O, as ozone, and P, as 'yellow' P, however neither of the latter allotropes represent the most thermodynamically stable forms of these elements.
- Of all of my categorisation suggestions for superseding the 'other nonmetals' category this one is the most consistent with, and well anchored in, the literature, as well as being simple, plausible, non-controversial and (IMO) intuitive. Sandbh (talk) 12:21, 17 November 2012 (UTC)
- It certainly is. But I still think am not into it. It is, first of all, counter-intuitive (which is definitely bad). O is not corrosive? The what is it wrong with the old garages standing near my house? Why are their corners brown while they are green? Corrosion is a well-known thing only for oxygen. It's weird that you exclude that given it's not sourced. Also, radon reacts with fluorine at r.t., nitrogen doesn't. How is the word "reactive" relevant? There is no word perfectly describing the situation. At least, we haven't heard it. I can only offer you "other." It's style-neutral. It doesn't say anything, true; but it is therefore not wrong. I think the other should remain for now; I disagree with that this category should be split by any means. Still, I'm open to new suggestions. What would you now say?--R8R Gtrs (talk) 14:20, 17 November 2012 (UTC)
- Hello R8R Gtrs, good to hear from you.
- O. In suggesting the term 'corrosive nonmetal' I was thinking in terms of hazardous substances (i.e. flesh burning or eating). O, as a strong oxidant, is capable of causing corrosion. But O is not a corrosive substance in the manner of F, Cl, Br and I. Hence O is not listed in Proctor and Hughes. As Wulfsberg (1987, p. 184) notes: '…the halogens (not H2) are very active oxidizing agents that are quite corrosive and must be handled with some care.'
- Reactivity. It doesn't matter that Rn reacts with F. It is still called a noble gas. So is Xe even though it forms hundreds of compounds. It doesn't matter that N is relatively inactive. It has a rich chemistry. It is far more reactive than the noble gases. It is clearly a reactive nonmetal in comparison to the noble gases.
- Other nonmetals. There is no precedent in the literature for referring to all nonmetals other than noble gases as 'other nonmetals'. If there are only two categories of nonmetals, and one of those is called 'noble gases' why would the rest of the nonmetals be called 'other nonmetals' rather than 'reactive nonmetals', given reactivity was used as a basis to categorize the noble gases? I do not see the logic. As Long & Hentz wrote: '…nonmetals can be split into two groups, the 'noble' or inert gases and the reactive nonmetals.' (1986, p. 84). As per Hill & Holman: 'From left to right across the periodic table, elements change from being reactive metals, through less reactive metals, metalloids, less reactive non-metals to reactive non-metals. (On the extreme right are the noble gases.)' (2000, p. 49).
- I'll think some more about all of this. Sandbh (talk) 06:23, 18 November 2012 (UTC)
- I'm still against corrosion. No, I mean, it's okay, you can explain it to me and we're good, but millions of people see that table. And most of them will scratch the head about that corrosion doesn't extend to O. Or report an error. I'm simply against giving such a hard time to many people. If even it's referenced, it's counter-intuitive. Not that it's wrong (even though I still think it is). Again, the point is surprising too many readers with no possibility to explain. Not correctness or in correctness. (Not to say corrosive is a subset of reactive)
- But you know, I really get your reactivity point. Agreed in the sense we could use the word to replace "Other." (say, if we simply changed the label "other nonmetals" to "reactive nonmetals," would you be satisfied as some kind of compromise?)--R8R Gtrs (talk) 15:24, 18 November 2012 (UTC)
- You're right about corrosive nonmetals. Replacing 'other' with 'reactive' seems to be the only logical way to go. The nonmetals can be subcategorized as (a) the noble gases; and (b) the reactive nonmetals. Sandbh (talk) 11:07, 19 November 2012 (UTC)
- So, can we now implement this change (i.e. rename "other nonmetals" to "reactive nonmetals", and then annex the "halogens" category into the "reactive nonmetals" category, but putting At as a metalloid)?
- I'd rather say yes, but let's see if anyone else has any objections.--R8R Gtrs (talk) 11:23, 19 November 2012 (UTC)
- So, can we now implement this change (i.e. rename "other nonmetals" to "reactive nonmetals", and then annex the "halogens" category into the "reactive nonmetals" category, but putting At as a metalloid)?
- You're right about corrosive nonmetals. Replacing 'other' with 'reactive' seems to be the only logical way to go. The nonmetals can be subcategorized as (a) the noble gases; and (b) the reactive nonmetals. Sandbh (talk) 11:07, 19 November 2012 (UTC)
In our multi-category periodic table do we categorise elements on the basis of their most thermodynamically stable states at ambient conditions or their most common form? My question relates to P. Black P is the most stable allotrope and is relatively inert; red P is far more common and relatively stable; white P is the commonest allotrope of all but is unstable. I gather the 'standard state' of P is regarded as being white P on the basis of its ease of preparation, industrial importance and commonality, despite its extreme instability. This approach has never seemed to me to represent good science—provides no basis for valid comparison. Sandbh (talk)
- I would rather vote for the white one, becaus e it is commonlĞy considered so, so more people know about this point of view, but you know I am pro-popular in such discussions. We can always describe the nuances in the main P article.-R8R Gtrs (talk) 12:28, 22 November 2012 (UTC)
- I was looking at the word 'caustic' as in 'Burning, corrosive, destructive of organic tissue.' (Oxford English Dictionary). Sulfuric acid is caustic, as are phenol and silver nitrate ('lunar caustic'); so is sodium hydroxide. Wondering if 'caustic' could be used as a sub-category of nonmetals. Motivation is concerns that the millions of average readers will expect to see halogens standing out among the nonmetals. If they are annexed as part of the reactive nonmetals they won't stand out. Only a few nonmetals are caustic: F, Cl, Br, I. Of the other reactive nonmetals O2 is corrosive, but that's it; Se is toxic if taken if taken to excess, but that's all: O3 is toxic, corrosive and caustic but is an unstable, uncommon form of O. How to treat white P? Average reader would expect to see white P counted as a caustic nonmetal. Highly unstable structure results in white P being self-igniting near room temperature---and burning white P is obviously caustic. So that would mean caustic nonmetals are F, P, Cl, Br, I. Reactive nonmetals would be H, C, N, O, S, Se. Noble gases are usual culprits. So lighter halogens would still stand out in accordance with common teaching that most reactive nonmetals are on right side of the periodic table (just before noble gases). But they would be accompanied by caustic white P. Seems slightly odd but is byproduct of treating unstable white P as standard state of phosphorus. Comments welcome. Am still thinking about this difficult topic. Sandbh (talk) 12:09, 23 November 2012 (UTC)
- I don't know – the halogens are just a group (used in the sense of "vertical column"). So they are not that hard to find. (Although we probably should have a separate key giving the names of the labelled vertical columns, something like "1 – alkali metals; 2 – alkaline earth metals; 11 – etc.", to make sure that it is still quite visible.)
- A further note: At is a halogen, but is not marked here. Your table might give the impression to readers that At is not a halogen, especially if they don't read the legend and see that the same colours are being used and assume that nothing has changed. (A few with even less knowledge of chemistry might even think P is a halogen!) There are some cases where people don't seem to read important information – all but one of the comments (abuse is not counted) on the Astatine article is asking for a picture of solid astatine, while the article already states "Elemental astatine has never been viewed, because a mass large enough to be seen (by the naked human eye) would be immediately vaporized by the heat generated by its own radioactivity."
- I would personally prefer using the most stable allotrope of each element (i.e. black P instead of white P), but can see reasons for both sides. Double sharp (talk) 13:25, 23 November 2012 (UTC)
- I was looking at the word 'caustic' as in 'Burning, corrosive, destructive of organic tissue.' (Oxford English Dictionary). Sulfuric acid is caustic, as are phenol and silver nitrate ('lunar caustic'); so is sodium hydroxide. Wondering if 'caustic' could be used as a sub-category of nonmetals. Motivation is concerns that the millions of average readers will expect to see halogens standing out among the nonmetals. If they are annexed as part of the reactive nonmetals they won't stand out. Only a few nonmetals are caustic: F, Cl, Br, I. Of the other reactive nonmetals O2 is corrosive, but that's it; Se is toxic if taken if taken to excess, but that's all: O3 is toxic, corrosive and caustic but is an unstable, uncommon form of O. How to treat white P? Average reader would expect to see white P counted as a caustic nonmetal. Highly unstable structure results in white P being self-igniting near room temperature---and burning white P is obviously caustic. So that would mean caustic nonmetals are F, P, Cl, Br, I. Reactive nonmetals would be H, C, N, O, S, Se. Noble gases are usual culprits. So lighter halogens would still stand out in accordance with common teaching that most reactive nonmetals are on right side of the periodic table (just before noble gases). But they would be accompanied by caustic white P. Seems slightly odd but is byproduct of treating unstable white P as standard state of phosphorus. Comments welcome. Am still thinking about this difficult topic. Sandbh (talk) 12:09, 23 November 2012 (UTC)
Highly reactive nonmetals
Based on all of the lengthy discussion so far, including about O and P, how does the following scheme look(?):
- Reactive nonmetals: H, C, N, S, Se
- Highly reactive nonmetals: O, F, P (white), Cl, Br, I
- Noble gases: usual culprits
Highly reactive nonmetals are those that are either caustic or corrosive, or both. Sandbh (talk) 10:56, 25 November 2012 (UTC)
- I think we're (in general) walking in circles. We already had the idea. In principle, I simply don't like the idea of cutting the non-noble nonmetals. There is no name well known by everyone to comply with being a hard scientific term. Like alkali metal, I mean. More reactive, less reactive are just degrees of the reactivity. This is no reason to cut down (what about more reactive transition metals, noble trnasition metals, more chem-rich actinides?). There are halogens, but halogen is not a subset of nonmetals. I personally wouldn't expect them to stand out. There's plenty of everything we've got in books (more reactive, corrosive, etc.), but they are not hard terms. Either a "(said property) nonmetal" or a degree of that. They don't build too strong a picture. To say more, this one would puzzle me also, because, well, I just wouldn't expect that. I honestly think that we should not make our low-educated readers wonder why we chose this. Which is why, in principle, I don't like anything looking unusual for our table. Even "nonmetallic halogens" would be better if I wanted to make the four stand out (but I don't). And even that leaves no idea how to name the others since everyone doesn't like the word "other."
- What we're probably going to use (I'm referring to the big green "Reactive nonmetals" group) is quite a propert--R8R Gtrs (talk) 20:27, 25 November 2012 (UTC)y as well, but it's at least one such category instead of two.
- Hi R8R Gtrs. I don't feel like we've been walking in circles. Rather, it seems to me that we've looked at, considered and discussed a series of categorization suggestions. Most of these were found to be unsatisfactory for scientifically grounded reasons. This is good science in the search for truth. As DePiep said around five months ago, "I know it [element categorization] is old & tough, but isn't solving that what we are here for?"
- In descriptive chemistry, O, F, P (white), Cl, Br and I are commonly described as being highly reactive. This is an old and well established concept. Just like nitro-glycerine or hydrogen peroxide are described as being highly reactive. There is no soft science in that.
- This proposal would retain the current number of nonmetal categories, which I think is conceptually a good thing and consistent with the literature.
- Personally, I don't see a particular need for further cuts e.g. reactive transition metals and noble transition metals but feel free to put up an argument for so doing.
- I didn't think Wikipedia was written for low-educated readers. As I understand it, Wikipedia is written for readers of average education. Even so, I'm confident that even low-educated readers could easily understand and appreciate the difference between reactive and highly reactive nonmetals.
- Noble gases aside, it is good, well established chemistry to distinguish between two subcategories of nonmetal based on reactivity. I've put up plenty of citations along those lines. It's just that it's taken a little while to hone in on an accurate way of distinguishing and describing the two subcategories. This is what we've been trying to do for the past five months, through a process of elimination and refinement, and have now succeeded at IMO, with help from lots of contributors.
- It doesn't matter if it will look a little odd, to have O and P coloured the same as F, Cl, Br and I. Nature is what it is. Chemists will get it---they write about it using these terms. Science professionals will get it. Less educated readers may get a little surprise---F, Cl, Br and I will look familiar in terms of their characteristics but what's the story with O and P? Oh, right, O causes rust, oxidation, corrosion etc, and P burns of its own volition---now I get it :)
- I can't think of anything else to say. I feel like I've exhausted the plentiful descriptive chemistry literature on this topic. I've run out of intellectual puff (at least for now). Sandbh (talk) 11:58, 28 November 2012 (UTC)
-
- Hi Sandbh. What I think is that for all these schemes where we split our non-noble (ignoble? :-P) nonmetals category, we need a lot of explanation. Otherwise you will have readers wondering and asking why element X is not listed as reactive and element Y is when element Z reacts with element X but not element Y, or other similar arguments that have been brought up here before. You can't expect to explain the rationale behind the categorization to every reader, especially when our compact periodic table is on all the elements articles, and do we really want to insert this explanation on every element, even when it is not very relevant (e.g. for the lanthanides)?
- Oh, sorry to read that. I don't know if the proposed scheme would need a lot of explanation? Chemists would get it. Science professionals would soon work it out. Other people either wouldn't know any better or would have their curiosity piqued sufficiently by the different colours to want to read further. Which is the beauty of Wikipedia---to facilitate further reading. Same thing happened to me when I first wondered what was the significance of the elements coloured to denote they were 'other nonmetals', as well as what was the basis for colouring some elements as metalloids but not others. I soon worked it out by clicking on the periodic table link included at the top of each element's page. Sandbh (talk) 12:02, 29 November 2012 (UTC)
- Also, does any authority classify the categories of the periodic table this way? I know you've mentioned a lot of quotes, but does anyone actually split the non-noble nonmetals when colouring a periodic table? There is a difference between an in-depth description of the properties of an element, such as whether it is more or less reactive (which we can cover in the main element articles), and a description that is satisfactory for colour-coding the periodic table. After all, H behaves very strangely in some ways for a nonmetal, which you've mentioned above, and I would expect many authors to cover these oddities in H's behaviour when concentrating on H alone (and not every element). But nobody classifies it as anything other than a nonmetal at STP. Well, I did find one periodic table online (here) which colour-codes H as an alkali metal, but this may be because of its atomic structure rather than its chemical properties. (Sometimes – most of the time, I think – authors simply bail out and simply give it its own category, but later describe it as a nonmetal when covering it separately: when an element is given its own colour on the PT, we obviously have to look at the author's treatment of it when it is covered on its own. Some are even strangely inconsistent, using an "Other nonmetals" category but at the same time colouring H separately(!).) Double sharp (talk) 14:07, 28 November 2012 (UTC)
- There are no authorities that I have found that colour a periodic table (as opposed to describing the applicable nonmetals in this way). It's funny. When I think about it most colour their tables into metals, metalloids or nonmetals, or into groups + transition metals, lanthanides etc. Not that many do a good a job as a Wikipedia and quite a few are straight copies of Wikipedia. OTOH, as you note, the distinction between categories such as reactive metals, less reactive metals, metalloids, less reactive nonmetals, more reactive nonmetals, and noble nonmetals is a reasonably common one, at least in writing. In that context, I see no particular issues with colour schemes that draw on or are based around these distinctions. I recall seeing a few periodic tables that classify H as a nonmetal as well as giving it its own colour.
- Overall it seems to me that I have the science right, as it is described in chemistry texts, but that I am not 'winning' on the emotional response/feelings front. Sandbh (talk) 12:02, 29 November 2012 (UTC)
- I think I've been caught with my foot in my mouth on this page enough to count as one of "our low-educated readers", so I'll add my 2¢ worth here. I initially reacted strongly (pun intended) to the elimination of the halogen category, but then I was enlightened by the cogent discussion that clarified the difference between "categories" (shown in WP with background colors) and "groups" (shown in periodic tables as columns). Now I'm convinced that the halogens don't need a special color. I tend to react slightly to the idea of just two non-metal categories; probably partly a holdover wanting something to replace the halogen category, and partly because if there are only two subcategories of non-metals, it seems like the obvious choices would be "X" and "non-X", but then there isn't a nice way to prefix "non-" onto "noble gas" and make any kind of sense. I tend to agree that it would be nice to get rid of the term "Other nonmetals", but for purely stylistic reasons and not scientific reasons. If the "Other nonmetals" were to the right of the noble gases in the periodic table so that the categories were "X nonmetals", "Y nonmetals" and "Other nonmetals", it might work stylistically. But since the catch-all category is listed before the definite category(ies), stylistically it seems extremely clumsy. If the impressions of this "low educated reader" are helpful, great; otherwise, feel free to ignore them.
- But let me ask one nagging question that I have. Given that it seems all have acquiesced to the idea that "halogen" is a group but not a category, does this same argument also apply to the metalic end of the table? Specifically, why should "Alkali metals" and "Alkaline earth metals" exist as categories and not be merely considered groups (columns)? Other than the ever-present thorn-in-the-flesh Hydrogen, is there any reason why these two groups should not be treated the same way this discussion is treating the halogens? YBG (talk) 23:18, 25 November 2012 (UTC)
Becausee At is a halogen but not a nonmetal (it's a metalloid), and the problem gets worse for 117. On the other end of the periodic table (group 1 and 2), no matter how far down you go on the periodic table, the elements are still metals. Double sharp (talk) 03:39, 26 November 2012 (UTC)
- Thanks for the clarification. YBG (talk) 03:48, 26 November 2012 (UTC)
- Hmm. Reading YGB's comments and nagging question again, and your response Double sharp, raises doubts for me. If they (the s-block metals) are all metals, why do we split them into two groups rather than one category such as s-block metals or reactive metals or some such, an idea which Nergaal and I have both mentioned (and YGB is alluding to? Keeping the two groups presumably on the basis of their different levels of overall reactivity (?) would strengthen the argument for splitting the reactive nonmetals category into two, as previously canvassed. Haven't thought about this too much so feel free to point out any shortcomings in my line of thought. Sandbh (talk) 23:16, 23 February 2013 (UTC)
Proposal #6
Here's a summary of the proposals for renaming and/or subdividing the non-noble nonmetals:
| # | 1st subcategory | 2nd subcategory | Categorization basis |
| 1 | Core: H, C, N, O, F, P, S, Cl, Se, Br, I | The essential part of a thing | |
| 2 | Hydrogen: H | Typical: C, N, O, F, P, S, Cl, Se, Br, I | H is not a typical nonmetal |
| 3 | Moderately active: H, C, P, S, Se, I | Active: N, O, F, Cl, Br | Electronegativity; HSAB principle |
| 4 | Unspecified: H, C, N, O, P, S, Se | Halogens: F, Cl, Br, I | Awkwardness of "Other nonmetals" category |
| 5 | Reactive: H, C, N, O, F, P, S, Cl, Se, Br, I | Reactive nometals are reactive compared to noble gases | |
| 6 | Reactive: H, C, N, P, S, Se, I | Highly reactive: O, F, Cl, Br | Consistent literature descriptions of "highly reactive" nonmetals as being highly reactive |
| 7 | Reactive H, C, N, O, P, S, Se | Corrosive: F, Cl, Br, I | Corrosive characteristics, as described in literature (overlooked O) |
| 8 | Reactive: H, C, N, S, Se | Highly reactive: O, F, P, Cl, Br, I | Corrosive or caustic characteristics, as described in literature |
Of these proposals, #6 was comparably well-received. I like it because there are no issues with allotropes (e.g. P); it preserves the top right hand corner feel for the most reactive nonmetals; it maintains two categories of non-noble nonmetals; it results in a nice progression of subcategories down the halogens; and it looks OK visually (bright yellow colour aside).
Is there any further support for this proposal? Sandbh (talk) 12:21, 1 December 2012 (UTC)
Where is H in proposal #3?Double sharp (talk) 12:22, 1 December 2012 (UTC)- Floating around in the back of my brain :) err, should be on board now Sandbh (talk) 12:31, 1 December 2012 (UTC)
- I'm a number 5 man. No breaks for the nonmetal category (which seem a little made up to me, not as obvious as the other breaks we use, e.g. TMs--PTMs, etc.), and you know my other arguments. Also, to get a fairer vote, would anyone bother to note Wp:CHEMISTRY or a few ones like that (have no idea what the public opinion would be, not trying to cheat somehow)?--R8R Gtrs (talk) 20:08, 11 December 2012 (UTC)
Comments sought from WP:CHEMISTRY
Here. Sandbh (talk) 21:42, 1 January 2013 (UTC)
Quantitative properties and nonmetal reactivity
The purpose of this contribution is to provide a quantitative basis for distinguishing between reactive and highly reactive nonmetals. To do this, the following table gives the properties of all the non-noble nonmetals, across nine properties associated with reactivity:
| Nonmetal | Electron affinity kJ/mol |
Electro- negativity |
Enthalpy of dis- sociation kJ/mol |
Standard reduction potential V |
Caustic? | Corrosive? | Pyro- phoric? |
HSAB | Forms noble gas com- pounds? |
Reactivity boxes |
|---|---|---|---|---|---|---|---|---|---|---|
| F | 334 | 3.98 | 159 | 2.87 | 1 | 1 | 0 | H | Y | 8 |
| Cl | 355 | 3.16 | 242 | 1.36 | 1 | 1 | 0 | H/B | P | 7.25 |
| Br | 331 | 2.96 | 193 | 1.07 | 1 | 1 | 0 | B/S | N | 6.25 |
| O | 147 | 3.44 | 498 | 1.23 | 0 | 1 | 0 | H | Y | 5 |
| I | 301 | 2.66 | 151 | 0.54 | 1 | 1 | 0 | S | N | 4 |
| S | 207 | 2.58 | 266 | 0.14 | 0 | 0 | 0 | S | N | 2 |
| Se | 201 | 2.55 | 332 | –0.40 | 0 | 0 | 0 | S | N | 2 |
| P | 78 | 2.19 | 198 | 0.01 | 0 | 0 | 1 | S | N | 2 |
| N | 0 | 3.04 | 945 | 0.27 | 0 | 0 | 0 | B | N | 1.5 |
| H | 79 | 2.20 | 436 | 0.00 | 0 | 0 | 0 | H | N | 1 |
| C | 128 | 2.55 | 346 | 0.13 | 0 | 0 | 0 | S | N | 0 |
| Average | 196 | 2.85 | 342 | 0.66 |
Note: Caustic = destructive of organic tissue.
If a particular property is quantitative, the last row gives the average value of the listed nonmetals.
I've then used yellow shading to indicate which nonmetals have an above average value for that property, and light grey shading for those that have a below average value (the other way 'round in the case of enthalpy of dissociation). Aqua shading denotes an intermediate value. If a property is binary (e.g. Caustic?) then the distinction between above average and below average is self-explanatory. In the case of HSAB rating I've assigned a value of 1 to 'hard' (H); a value of 0.5 to borderline (B); and a value of 0 to 'soft' (S). If a nonmetal is sometimes listed as more than one HSAB category, I've assigned it the average of the applicable values. In the Forms noble gas compounds? column, Cl has a value of P for possibly since that is the way I read the literature on this question.
The last column shows how many 'above average' property boxes a particular nonmetal has ticked. Since 9 is the greatest number of property boxes that can be ticked it follows that > 4.5 boxes is above average and < 4.5 is below average.
In case anybody is wondering, and as an example, enthalpy of dissociation (or element bond strength) is associated with reactivity: 'The high dissociation enthalpy of the O2 molecule, 498 kJ/mol, is the reason that molecular O. is relatively unreactive and its reactions usually require thermal or photochemical activation.' (Eagleson 1994, p. 768)
On the basis of the above table, nonmetals of above average or high reactivity are F, Cl, Br and O, an outcome that is consistent with the literature.
Overall, there isn't much involved with this approach. It would be expected that any particular nonmetal with an above average number of properties that have above average values for properties associated with reactivty, would have above average or high overall reactivity. I didn't know what the outcome of this approach would be when I started and I didn't really care, because the values would fall where they fell, but I did think that F and Cl would be up there.
Yes, I'm still proposing we distinguish between reactive and highly reactive nonmetals since this is a well established periodic trend in descriptive chemistry, as consistently cited in this thread. The above table provides a non-subjective way of distinguishing between the two categories, by drawing on known values of relevant properties and sorting these into simple above or below average boxes. Sandbh (talk) 11:57, 27 February 2013 (UTC)
- Eagleson M 1994, Concise encyclopedia chemistry, Walter de Gruyter, Berlin
I'm not well-versed in the WP policies, but I'm wondering how WP:OR applies in this instance. Clearly, the individual entries in the cells are not OR, but I'm not so sure about the conclusion as to where the line should be drawn. I don't feel strongly about this, but I'm just trying to think things through. And just for interest, I made the above table sortable. YBG (talk) 05:38, 28 February 2013 (UTC)
- Rawlins, Struble & Gatewood (1961, p. 58) give the nonmetal activity series as Fl, Cl, Br, O, I and S, as do Krauskopf and Beiser (2006, p. 318). So the table reactivity box order is consistent with the literature. Also, in discussing the chemistry of the nonmetals, Cox (2004, p. 147) distinguishes F, Cl, Br and O as good oxidizing agents, and Clugston and Flemming (2000, p. 221) similarly note that F, Cl, Br and O often act as oxidants. So that's consistent with where the above average/below average line happens to fall. Iodine's position in the below average cohort is consistent with the repeated comments it gets about its relatively modest reactivity—I provided eleven citations to this end early on in the Reactive nonmetals and noble gases section. Thank you for making the table sortable. I was a bit tired when I posted it. Sandbh (talk) 11:43, 28 February 2013 (UTC)
- Clugston MY & Flemming R 2000, Advanced chemistry, Oxford University Press, Oxford
- Cox PA 2004, Inorganic chemistry, 2nd ed., Instant notes series, Bios Scientific, London
- Krauskopf KB & Beiser A 2006, The physical universe, 11th ed., McGraw-Hill, New York
- Rawlins GM, Struble AH & Gatewood CW 1961, Chemistry in action, Heath, Boston
- Some more contributions and thoughts about this: first a few more citations, and then about the OR question.
- Citations
- 'The alkali metals, such as Cs, K, Na, which tend to lose electrons readily, are powerful reducing agents. Others, like F, O, Cl, Br, are powerful oxidizing agents because they take up electrons readily.' (West 1956, p. 54)
- 'Oxygen is not the only agent which supports combustion. Fluorine is even more active, and we must also classify as oxidising agents a number of fluoride compounds such as… Chlorine and bromine are also oxidising agents, especially in rich mixtures of hydrogen. Iodine, which usually separates out in non-combined form in explosions, is not.' (Medar 1989, p. 10)
- 'Carbon is neither a strong metal nor a strong nonmetal and has less electron-attracting power than the strong nonmetals bromine, chlorine, fluorine, and oxygen, in their covalent bond formation.' (Reel 2006, p. 263)
- 'Iodine is a moderately strong oxidizing agent…' (Young 2006, p. 1285)
- 'Bromine is a very strong oxidizing agent. Of all the elements, only oxygen, chlorine, and fluorine are stronger.' (Young 2009, p. 1173)
- Citations
- OR?
- Showing in picture form what is written in the literature is not OR. The literature:
- notes a left-right trend across the standard periodic table from very reactive to less reactive metals, followed by metalloids, less reactive nonmetals, and more reactive nonmetals; and then largely by unreactive nonmetals;
- says that the most reactive nonmetals are located in the upper right-hand corner, excluding the noble gases;
- records a reduction in reactivity going down the halogens and an increase in metallic character;
- describes oxygen, fluorine, chlorine and bromine more or less consistently as highly reactive nonmetals, or the like; and
- does not consistently describe any other nonmetals in comparable overall terms, apart from noting a few isolated features such as, for example, the relatively high electronegativity of nitrogen or the corrosiveness of iodine.
- I don't think I've overlooked anything in the literature about this and am happy to stand corrected. Sandbh (talk) 03:48, 3 March 2013 (UTC)
- Medard LA 1989, Accidental explosions, vol. 1 (Physical and chemical properties), Ellis Horwood, Chichester
- Reel KR 2006, The best test preparation for the AP Chemistry exam, 9th ed., Research & Education Assocation, Piscataway, New Jersey
- West ES 1956, Textbook of biophysical chemistry, 2nd ed., Macmillan, New York
- Young JA 2006, 'Iodine', Journal of Chemical Education, vol. 83, no. 9, p. 1285
- Young JA 2009, 'Bromine', Journal of Chemical Education, vol. 86, no. 10, p. 1173
- An interesting citation which I haven't been able to quite attribute with 100% certainty. It appears to be from an updated version of an article by Young & Sessine (2000, p. 189), which now sits behind an institution-only pay wall, and none of the libraries near me have access:
- 'The Periodic Table can be used to predict ability of species to act as oxidizing agents. The reactivity of nonmetallic elements often parallels their electronegativity: the more electronegative the element, the more powerful an oxidant. Fluorine is the strongest oxidant of all the elements. Oxygen, the next most electronegative element, is also a powerful oxidant, as are chlorine and bromine. Elemental nitrogen, however, is only capable of oxidizing a few metals such as lithium and magnesium.' (Young 2006) Sandbh (talk) 11:32, 4 March 2013 (UTC)
- Young RV & Sessine S (eds) 2000, 'Oxidation', in World of chemistry, Gale Group, Detroit
- Young RV (ed.) 2006, 'Oxidation' in World of Chemistry, Science Resource Center (now Science in Context), Thomson Gale, Detroit
- An interesting citation which I haven't been able to quite attribute with 100% certainty. It appears to be from an updated version of an article by Young & Sessine (2000, p. 189), which now sits behind an institution-only pay wall, and none of the libraries near me have access:
- The following citation is a no-brainer but it illustrates the contribution, in this case, of the two properties of dissociation enthalpy and reduction potential:
- 'Fluorine is the most reactive of the elements, in part because of the weakness of the F—F bond, (B.E. F—F = 153 kj/mol), but mostly because it is such a powerful oxidizing agent (E° = +2.889 V).' (Masterton & Hurley 2008, p. 556) Sandbh (talk) 11:55, 4 March 2013 (UTC)
- Masterton WL & Hurley CN 2008, Chemistry: Principles and Reactions, 6th ed., Brooks/Cole Cengage Learning, Belmont, California
A streamlined option
Is there any appetite for (1) replacing the alkali metal, and alkaline earth metal categories, with the single category of s-block metals; and (2) having just two categories of nonmetals: the reactive nonmetals, and the noble gases? I still advocate showing the group names on our periodic table, so the 'alkali metal' and the 'alkaline earth metal' groups would still be identifiable, just as all the other group names would be included. I also still think dividing the reactive metals into reactive and highly reactive categories is markedly more informative chemistry, but I could live with an eight category table as a better construct than what we have now. Sandbh (talk) 11:29, 19 March 2013 (UTC)
- re (1): Don't they have distinguishable chemical and physical properties & behaviour? In other words, could you write the article s-block metal without constantly having to separate into two descriptions? And why drop the two well-established names (which include the word "metal", what this categorisation is about) into a mere descriptive, not proper named, one? -DePiep (talk) 15:30, 19 March 2013 (UTC)
- Not really. Main differences are reactivity. Article easy to write. I recall reading articles on chemistry of s-block metals. See, frex Advanced Chemistry by Clugston and Rosalind (2000, Oxford University Press), who spend 18 pages on an intregated treatment of the the s-block elements (pp. 280-97). Reactive nonmetals, in contrast, are much, much more diverse. Of course, that does not stop authors of books writing about the chemistry of nonmetals but result is always rambly reading. Drop names for same reason drop halogens: internal logic. Proposal is to keep all three names anyway but as group names not category names. And we can link groups to their articles. See fuller explanation of logic, below. Sandbh (talk) 11:38, 20 March 2013 (UTC)
- I tend to agree with DePiep on (1). On (2), you have been persuading me about the reactive/highly reactive categorization, and I am now somewhat convinced (although unsure how best to do this still). But what do others think? Double sharp (talk) 15:51, 19 March 2013 (UTC)
- (1): strongly no. DePiep perfectly explains why.
- (2): strongly yes. At very least because the 8-categories scheme has undisputed (metalloids maybe aside) category borders. See everything I wrote about it in 2012.--R8R Gtrs (talk) 18:54, 19 March 2013 (UTC)
- R8R, just a question, please point to that 8-categories scheme (expected, somewhere on this page, right?). -DePiep (talk) 00:20, 20 March 2013 (UTC)
- I should have found it myself, sorry. -DePiep (talk) 09:01, 21 March 2013 (UTC)
- Fuller explanation of logic. Our periodic table is based on categories not groups. If we divide the nonmetals into just reactive nonmetals and noble gases then there is no logical reason for dividing the s-block metals, which essentially only differ in their reactivity, into alkali and alkaline earth metal categories. Put another way, the differences in reactivity amongst the reactive nonmetals, not to mention their chemistries, differ much more than is the case with the s-block metals. Yet we propose to lump the non-noble nonmetals all into just the one category of reactive nonmetals and retain the categories of alkali metal and alkaline earth metal. Illogical. OTOH if we divide the reactive nonmetals into (a) reactive nonmetals and (b) highly reactive nonmetals then it makes sense to divide the s-block metals into (a) alkali metal and (b) alkaline earth nonmetals. Either option works, but a mixed logic one doesn't. There is no perfect categorisation scheme but I reckon either of these options are better than the chimera we have now. I mean no disrespect to anybody by calling our current categorisation scheme a chimera however I don't believe it has ever been subject to some thorough analysis. PS R8R: Minor quibble. Borders of transition metals, (arguably lanthanides, actinides), poor metals and what we currently call 'other nonmetals' are not undisputed. Sandbh (talk) 11:40, 20 March 2013 (UTC)
- As an open question: The high level categories are metal–metaloid–nonmetal. Using reactiveness as a criteria for further subdivision, is that related to the metalishness of an element? (Or is it like: grouping vehicles in trucks/cars, and then subdivide by color: i.e., using unrelated criteria) -DePiep (talk) 09:01, 21 March 2013 (UTC)
- Kind of, in that the presence or absence of metallishness in elements with incomplete valence shells corresponds to the left-right trend across the standard periodic table from very reactive to less reactive metals, followed by metalloids, less reactive nonmetals, and more reactive nonmetals. Sandbh (talk) 13:12, 22 March 2013 (UTC)
- You make me (the layman) doubting. This is a good, eh, usefull criterium. Of course our current colors or names do not matter now. -DePiep (talk) 23:43, 2 April 2013 (UTC)
- This question: why is this not OR? DePiep (talk) 21:21, 5 April 2013 (UTC)
- Hmm. It doesn't advance a new position. It is consistent with literature observations of a more or less steady left-right transition from reactive metals, through less reactive metals, metalloids, less reactive nonmetals, more reactive nonmetals, and inactive nonmetals. It is consistent with an established and associated literature-based division of nonmetals into three sub-categories. It is consistent with statements made in the literature that the most reactive nonmetals (not counting the noble gases) are found in the top right-hand corner of the standard form of periodic table. It is consistent with the literature-stated relationship between the oxidising power and reactivity of non-metals, and the identification of O, F, Cl and Br (and not I) as strong oxidants. It is consistent with statements made in the literature as to the increasing metallicity, and decreasing reactivity, of the heavier nonmetal congeners. It is consistent with literature-based qualitative descriptions of the reactiveness of the nonmetals. Sandbh (talk) 01:38, 6 April 2013 (UTC)
- You make me (the layman) doubting. This is a good, eh, usefull criterium. Of course our current colors or names do not matter now. -DePiep (talk) 23:43, 2 April 2013 (UTC)
- Kind of, in that the presence or absence of metallishness in elements with incomplete valence shells corresponds to the left-right trend across the standard periodic table from very reactive to less reactive metals, followed by metalloids, less reactive nonmetals, and more reactive nonmetals. Sandbh (talk) 13:12, 22 March 2013 (UTC)
- As an open question: The high level categories are metal–metaloid–nonmetal. Using reactiveness as a criteria for further subdivision, is that related to the metalishness of an element? (Or is it like: grouping vehicles in trucks/cars, and then subdivide by color: i.e., using unrelated criteria) -DePiep (talk) 09:01, 21 March 2013 (UTC)
- R8R, just a question, please point to that 8-categories scheme (expected, somewhere on this page, right?). -DePiep (talk) 00:20, 20 March 2013 (UTC)
- I tend to agree with DePiep on (1). On (2), you have been persuading me about the reactive/highly reactive categorization, and I am now somewhat convinced (although unsure how best to do this still). But what do others think? Double sharp (talk) 15:51, 19 March 2013 (UTC)
- Not really. Main differences are reactivity. Article easy to write. I recall reading articles on chemistry of s-block metals. See, frex Advanced Chemistry by Clugston and Rosalind (2000, Oxford University Press), who spend 18 pages on an intregated treatment of the the s-block elements (pp. 280-97). Reactive nonmetals, in contrast, are much, much more diverse. Of course, that does not stop authors of books writing about the chemistry of nonmetals but result is always rambly reading. Drop names for same reason drop halogens: internal logic. Proposal is to keep all three names anyway but as group names not category names. And we can link groups to their articles. See fuller explanation of logic, below. Sandbh (talk) 11:38, 20 March 2013 (UTC)
- I oppose merging alkali and alkaline earth metals. Isn't the point of color-coding the periodic table to group elements by chemical properties? And alkali and alkaline earth metals have quite different chemical properties.King Jakob C2 12:20, 20 March 2013 (UTC)
- Yes, I understand the color codings mostly correspond to broad, higher level chemical patterns, rather than the more distinct chemical properties of individual groups. At this level of categorisation their is no essential difference between the alkali and alkaline earth metals. They are 'typical' very electropositive and reactive elements, forming strongly alkaline oxides, and are almost invariably found in oxidation states expected for ions in noble-gas configs (e.g. Na+, Mg2+). Occur widely in nature in silicate minerals although weathering gives rise to concentrated deposist of halides (e.g. NaCl, CaF2), carbonates (CaCO3) and hydroxides. See, for example Doc Brown ('chemically very similar') or Ramsden 'The s block metals have much in common'. Sandbh (talk) 08:27, 21 March 2013 (UTC)
- Your logic is correct. If there were no established organization, I'd use yours (with s-block metals and reactive nonmetals forming two and not four groups). But can't we just say "none knows what 's-block metals' are"? How about making an exception? (The second form the the verb "see" is "saw" after all and not "seeed," as I always wanted it to be, someone's
makedmade a exception everyonebeginedbegan to follow, and I'm pretty sure there are a lot of people who will be staring at "s-block metals" (or whatever) as amused as if theysawseeed that "seeed" :)--R8R Gtrs (talk) 18:56, 23 March 2013 (UTC)- I like your explanation. Presumably this means we should keep two subcategories of non-noble nonmetals. Sandbh (talk) 09:58, 7 April 2013 (UTC)
- Your logic is correct. If there were no established organization, I'd use yours (with s-block metals and reactive nonmetals forming two and not four groups). But can't we just say "none knows what 's-block metals' are"? How about making an exception? (The second form the the verb "see" is "saw" after all and not "seeed," as I always wanted it to be, someone's
- Yes, I understand the color codings mostly correspond to broad, higher level chemical patterns, rather than the more distinct chemical properties of individual groups. At this level of categorisation their is no essential difference between the alkali and alkaline earth metals. They are 'typical' very electropositive and reactive elements, forming strongly alkaline oxides, and are almost invariably found in oxidation states expected for ions in noble-gas configs (e.g. Na+, Mg2+). Occur widely in nature in silicate minerals although weathering gives rise to concentrated deposist of halides (e.g. NaCl, CaF2), carbonates (CaCO3) and hydroxides. See, for example Doc Brown ('chemically very similar') or Ramsden 'The s block metals have much in common'. Sandbh (talk) 08:27, 21 March 2013 (UTC)
The precedent and basis for two nonmetal categories
I want to briefly re-address concerns raised in this thread about splitting the nonmetals in two. Our periodic table has always split the nonmetals into two categories: other nonmetals, and halogens. This split is broadly consistent with the well-established distinction made in the literature between less reactive nonmetals and more reactive nonmetals. Now, if we get rid of the halogens category, concerns have been raised that there is no other established basis to split the nonmetals in two. To alleviate these concerns I've cited several sources referring to:
- the broad categories of less reactive nonmetals, and more reactive nonmetals (i.e., the well known L-R periodic trend in reactivity);
- stand-out oxidants among the nonmetals being O, F, Cl and Br, noting that for a nonmetal, reactivity is usually gauged by its ability to serve as an oxidising agent (Brady & Senese 2009); and
- the patchy reactivity or oxidising power of iodine.
In conclusion, splitting the nonmetals into the categories of reactive nonmetals, and highly reactive metals (O, F, Cl, Br) would be consistent with, and supported by the literature. Sandbh (talk) 09:27, 23 March 2013 (UTC)
Option 10
Subcategorise the nonmetals according to their molecular structures:
- Noble gases: He, Ne, Ar, Kr, Xe, Rn
- Diatomic nonmetals: H, N, O, F, Cl, Br, I
- Polyatomic nonmetals: C, P, S, Se
The lines of demarcation are apparent. Structure generally corresponds to metallishness: Metals generally have high coordination numbers (CNs); metalloids have intermediate CNs; and nonmetals have low CNs, culminating in the noble gases with CN = 0. There are no issues with white P v black P; both are polyatomic.
The terms 'diatomic' and 'polyatomic' are both found in the nonmetal literature: 'Figure 3.2 shows seven nonmetal elements that can exist as diatomic molecules. Atoms of a smaller number of elements can form polyatomic molecules. Two examples shown in Figure 3.2 are phosphorus, which can form P4 (read as "P-four"), and sulfur, which can form S8 (read as "S-eight")." (Miller T 1987, Chemistry: a basic introduction, 4th ed., Wadsworth, Belmont, CA, p. 62)
We have probably all seen periodic tables that highlight the molecular structures of the non-metals (S8, O2 etc).
A similar categorisation pattern can be seen going rightwards across the standard form of the periodic table. Whereas most metals crystallise in close-packed structures with high coordination numbers (8+ to 12, or higher), the poor metals have more complex structures, with lower coordination numbers (4+ to 6+). This can be attributed to the influence of partially covalent bonding in their crystal structures, which dictates fewer nearest neighbours.
It is not a perfect categorisation. But it is simple, and interesting (in my view). Sandbh (talk) 01:58, 7 April 2013 (UTC)
- I like it also. From a purely stylistic point of view, categories like "more xxx" and "less xxx" beg the question "Where do you draw the dividing line?" which has the potential to lead to endless discussion, for example on this talk page, to say nothing of the literature which has been cited here. As long as the division between diatomic and polyatomic is clear and unambiguous, that is. I could easily see Diatomic nonmetals and Polyatomic nonmetals articles which would be interesting in themselves. Even if this particular categorization is not selected, I think it points in a good direction: find some characteristic that clearly and unambiguously divides the nonmetals into subcategories. YBG (talk) 15:54, 7 April 2013 (UTC)
- When it comes to personal preferences (not regarding the fact it may/may not go), it is my close second, sounds like a good idea, out of box indeed. Like it in general (I really do).
- But as always, I'm gonna ruin the fun :(
- The problem is: why divide into di- and polyatomic? Even leaving the allotropes problem aside, is the most stable state's structure a property characterizing enough to break a category in two? I mean, why not "reactive nonmetals whose Hungarian names start with F" and "reactive nonmetals whose Hungarian names don't start with F"?
- I mean, there are actually relatively sophisticated reasons behind this, but they're not obvious.
- It looks like it's a break to break.
- In that sense, even our current scheme is better.
- I'm in general against mirroring the s-block metals/p-block nonmetals division simply because they're similar.
- There's one thing standing behind it: s-block metals are easy to break. They make two equal linear categories, despite being similar in some respects, they differ in hardness, mp/bps, valence, etc. This break is well-established. All overview chem books I know (like Earnshaw&Greenwood) treat them separately. Even, to some extent, the fluorine article: "The alkali metals react with fluorine with a bang (small explosion), while the alkaline earth metals react at room temperature as well but not as aggressively." Those statements were actually around 30 pages apart in the real book. It's worth discussing if they should be drawn together, but (almost) the whole world doesn't, and thus we shouldn't either.
- What's the most similar element to sulfur? Right, selenium. To arsenic? Antimony. In almost all cases (except for, sometimes, periods 2 and 6) it's vertical relationship that matters the most. The halogens. The chalcogens/oxygen group. Nitrogen group. Carbon group. Boron group. It's also why most overview chem books divide p-block into periods as well. There's only one more trend of general interest-- metal/nonmetal. But it's hard to describe the nonmetals as a whole. (My school chem book tried to, was very poor at it. My amazing uni book, definitely a few leagues higher than other uni chem books I've seen, talks about groups.) Since we don't do the per group grouping, the metal/nonmetal is the best thing on offer. My whole point there are only two categorizations possible for groups 13--17. Per group (and then we can draw the Uganda flag with p-block (or, okay, the gay flag)). Or per metallicity.
- While the terms "polyatomic" and "caustic" and whatever else can be traced to science books, I bet they're not used for ultimate categorization other than caustic/noncaustic. In an actual PT colored AM/AEM/TM/lanthanides/etc./etc./caustic nonmetals/noble gases. I, for example, have never seen such one.
- Other categorizations either eyebrow-raising or have nonreasonable (break just to break) breaks (or both). I don't claim they're all such, but all we've seen are, to me at least.
- That's my point.--R8R Gtrs (talk) 18:52, 7 April 2013 (UTC)
- Points very well taken. Let me add to my list of criteria for division:
- Clear. The criterion for division should be easily explained
- Unambiguous. It should be (relatively) obvious which category each element fits into
- Meaningful. The categories should have significance more than just dividing for the sake of dividing. There should be enough within-group similarity and enough between-group dissimilarity so that each group could be the subject of a separate encyclopedia article.
- I think what @R8R is saying is that Option 10 is lacking in the Meaningful category. I'll readily admit that I was quick off the mark because not only because the proposal meets the first two criteria, but also deals so elegantly no only with phosphorus but also with the problem child of every nonmetal categorization: hydrogen. YBG (talk) 05:10, 8 April 2013 (UTC)
- Yes, exactly. Agree on your division criteria scheme.--R8R Gtrs (talk) 14:58, 8 April 2013 (UTC)
- Hmm. I only have a few comments. R8R Gtrs: I was very pleased to get a close second. That means I only have to give it a nudge to get it into first :)
- The proposed division is not concerned with matching the split of the s-block metals. Rather, it's concerned with maintaining, like R8R Gtrs said, the 'established organization' of splitting the non-noble nonmetals into two categories.
- As noted, the general meaning of this division is metallicity, which has a well established connection to bonding structure, which in turn has a fundamental connection to atomic structure. I say 'general' since, as with all periodic table categorisations, including our current scheme, there are rub points at the boundaries, as well as overlaps.
- In accordance with YBG's elegant criteria, there is sufficient within-group similarity and enough between-group dissimilarity to make this division clearly more than 'a break just to break'. There would be a couple of quite interesting encyclopaedia articles in it. Here's an outline of each sub-category:
- Diatomic nonmetals. H, N, O, F, Cl, Br, I. Consistent with their low coordination numbers, which are all 1 in this case, the diatomic nonmetals show less metallic character than their polyatomic neighbours. The diatomic nonmetals are mostly highly insulating non-reflective gases that are more electronegative than the polyatomic nonmetals. They all have: lower melting points than the polyatomic nonmetals (in their thermodynamically most stable forms); and lower boiling points than those of the polyatomic nonmetals (bothersome white P included). Boundary phenomena occur with hydrogen (comparatively low electronegativity; seems to act like a metal in some of its chemistry on account of it having a single valence electron); and iodine (lustre, two-dimensional semiconductivity etc).
- Postscript: I overlooked the similarity that all the diatomic nonmetals, bromine and iodine included, are volatile at room temperature. I also overlooked the dissimilarity to the polyatomic nonmetals in that, among the latter elements, only white phosphorus (the thermodynamically least stable form of phosphorus) shows any appreciable room temperature volatility. Sandbh (talk) 12:33, 15 April 2013 (UTC)
- Polyatomic nonmetals. C, P, S, Se. Consistent with their higher coordination numbers (2 to 3) the polyatomic nonmetals show more metallic character. They are all solid, mostly semi-lustrous semiconductors or photoconductors with lower electronegativities than the diatomic metals. S is the poor cousin although even here it is a photoconductor (sometimes described as a semiconductor); it is malleable in its plastic form; it has a well-established cationic chemistry; and its trioxide is a glass-former (as are oxides of P and Se; ditto CO2 but only at 40 GPa). All the polyatomic nonmetals, unlike the diatomic nonmetals, show a marked tendency to catenation and allotropy.
- Yes, there are boundary issues. Just as there are with, for example, beryllium (borderline poor); the transition metals (do they/don't they include group 3); gold (king of the metals with multiple nonmetallic properties); the poor metals (do they/don't they include the coinage metals); and radon (borderline metalloid). I don't think these are showstoppers, as long as the categorisation scheme is based on simple, understandable criteria that provide useful insights. Now that I've looked into it some more, this scheme does that very well, IMO. Sandbh (talk) 14:17, 10 April 2013 (UTC)
- (re below)
- Yes, exactly. Agree on your division criteria scheme.--R8R Gtrs (talk) 14:58, 8 April 2013 (UTC)
- Points very well taken. Let me add to my list of criteria for division:
When I don't reply for a few days, it means, I'm taking a broader perspective. It took me a few days to realize that I'm still very doubtful about this.
Like, I started top listen to a lot of indie music much (don't care about trendiness, just like it), but that doesn't mean I would use an indie song to illustrate the music article. That kind of a close second.
Not trying to match s-block? good then (I just like it, nothing more)
"the 'established organization' of splitting the non-noble nonmetals into two categories." I really really feel this doesn't count as a good argument, but I'm not sure if I can prove it. I do understand your point, not just reject it. But I think that if a border isn't clear (since there are a few disputable ways to break 'em in two), it's not a reason to draw one. Moreover, relatively arbitrarily. Ah, I had said I would've been poor at it.
"Here's an outline of each sub-category": here's another one coming. Okay, you can tell me, but you surely won't put it into the template, and how will then the less acknowledged readers get the point apart from main allotrope structure itself?
And this, I think, is why we don't have a common opinion. I want to make it reader-friendly, as reader-friendly as possible, and you want to organize it at your best, and you're good at that. As a guy who could explain you the theory behind this di-/polyatomism, I find it very good (that's how you got the close second, yeah, a good explanation!). As an average reader who hadn't taken in this discussion, I would say, wtf? so num of atoms matters now? why not Hungarian names?
My belief is: If we were writing a book, that, given an appropriate explanation is also there, would be fine and good. A book is not designed for looking through, so this wtfs wouldn't be a problem, as a reader would stay on anyway and we could explain him the point. (The inner me is not opposed to breaking them in two in general, when a reader has a few mins to get the point) Wikipedia is, though, designed for looking though, and even if not, is used for it. This means we have to be as laconic as possible. That's what I don't like about it. A reader won't be thinking. Won't be able to get the point w/o chem education.
Summarizing: He'll be surprised about this previously unseen categorization. Or wtfing. Or a combination. Don't want any of three.
(P.S. If we already have boundary problems, it doesn't mean we should add new. Especially when this can be avoided.)--R8R Gtrs (talk) 20:32, 13 April 2013 (UTC)
- I do not think that a concern—based on one's feelings or beliefs or the Hungarian names of elements—as to whether Joe Average will get the point about diatomic and polyatomic nonmetals, should outweigh multiple literature-based arguments and related advantages that support such a scheme. It is a manifestly better scheme than the exemplary wtf 'other nonmetals' category or, what would make matters even worse, a scheme in which the remaining halogens would be annexed into a jumbled 'all sorts' category. Sandbh (talk) 14:06, 14 April 2013 (UTC)
- I've prepared a draft makeover of the existing nonmetal article, in my sandbox, using the three proposed sub-categories of noble gases, diatomic nonmetals, and polyatomic nonmetals. Haven't included any new citations yet. The draft reads well and is interesting, in my view. Please have a look and tell me what you think. Sandbh (talk) 06:07, 21 April 2013 (UTC)
Selenium – nonmetal or metalloid?
This is the only other plausible candidate for being called a metalloid, and I would be interested in hearing what you (Sandbh) think about Se. Double sharp (talk) 13:44, 6 July 2012 (UTC)
- I think its electronegativity (2.55) is a little too high for a metalloid. The electrical conductivity of highly purified pure selenium doesn't cut much chop either, being less than that of bromine. OTOH it does have some metal-like properties the most marked of which is probably its photoconductive capacity. It also has a lustrous appearance, and its structure has been characterized as featuring weakly metallic interchain bonding. So it depends on which criteria are being used to establish metalloid status. If selenium was to be regarded as a metalloid then carbon and phosphorous would probably also have to be admitted into the club, not to mention iodine. From the perspective of the wisdom of the masses, I see that selenium only appears around half as often as polonium and astatine do in the list of metalloid lists and they, in turn, appear around half as often as the recognized metalloids. I can understand what is going on with polonium and astatine but the low appearance frequency of selenium, in light of its established properties, is quite telling. Also relevant is the observation of Rochow (1957), the guy who later wrote one of the few monographs on metalloids, when he said that, 'In some [italics added] respects selenium acts like a metalloid and tellurium certainly does.' Having regard to the literature, I think selenium would probably be better shown as straddling the boundary between metalloids and core nonmetals, along with carbon, phosphorous and iodine. Sandbh (talk) 02:47, 7 July 2012 (UTC)
- Rochow EG 1957, The chemistry of organometallic compounds, John Wiley & Sons, New York, p. 224
- Rochow EG 1966, The metalloids, DC Heath and Company, Boston
- OK, no selenium then. Double sharp (talk) 05:55, 7 July 2012 (UTC)
- It seems that selenium is however regularly categorized as a metalloid in aquatic (as well as soil and/or environmental) chemistry, e.g. '...Selenium traditionally is considered a metalloid in aquatic chemistry...' (Meyer et al. 2005, p. 284) This is curious since it isn't immediately obvious to me what it is about Se that would warrant such a categorization in just these fields of chemistry. Perhaps by association with 'heavy metal' poisoning? Sandbh (talk) 11:55, 9 October 2012 (UTC)
- Meyer JS, Adams WJ, Brix KV, Luoma SM, Mount DR, Stubblefield WA & Wood CM (eds) 2005, Toxicity of dietborne metals to aquatic organisms, Proceedings from the Pellston Workshop on Toxicity of Dietborne Metals to Aquatic Organisms, 27 July–1 August 2002, Fairmont Hot Springs, British Columbia, Canada, Society of Environmental Toxicology and Chemistry, Pensacola, FL
- It seems that selenium is however regularly categorized as a metalloid in aquatic (as well as soil and/or environmental) chemistry, e.g. '...Selenium traditionally is considered a metalloid in aquatic chemistry...' (Meyer et al. 2005, p. 284) This is curious since it isn't immediately obvious to me what it is about Se that would warrant such a categorization in just these fields of chemistry. Perhaps by association with 'heavy metal' poisoning? Sandbh (talk) 11:55, 9 October 2012 (UTC)
Flerovium
What should we colour it as? (See Talk:Flerovium.) Double sharp (talk) 14:35, 20 November 2012 (UTC)
- Since chemical characterization has been performed, Fl's chemical properties are certainly not unknown, as stated in the current table. However, different studies seem to draw different conclusions regarding its chemical behaviour. Double sharp (talk) 09:08, 27 November 2012 (UTC)
- FWIW, zh. colours Fl as a poor metal. Double sharp (talk) 14:16, 18 February 2013 (UTC)
Is anyone looking here, BTW? (This post has two objectives in mind: firstly to "bump" the whole section so it won't get archived for a while longer, and secondly because saying this seemed to make discussion come in the "Predictions" subsection, so I think I should try it again to tie up this particular loose end.) Double sharp (talk) 14:20, 23 March 2013 (UTC)
Making this go live
When we get our new scheme to go live, we need to:
- Replace "post-transition metals" with "poor metals";
- Get rid of the halogens category, absorbing F, Cl, Br, and I into "other nonmetals" and At into "metalloids";
- Replace "other nonmetals" with "reactive nonmetals";
- Other minor things about the extended periodic table (colouring of some 7p and 9/8p elements, layout, and making articles use relativistic models)
Double sharp (talk) 09:08, 27 November 2012 (UTC)
Names in basic PT
- See {{Periodic table}} and {{Periodic table/sandbox}}
A feedback remark (can't find it right now, but trust me) said something like "the periodic table should show the element name". That is a useful thing. I have made a sandbox version for this, trying to show the name without making the template too wide for a wikipage. See {{Periodic table/sandbox}}. Any comments? (todo: positioning of name in the cell to top; check smaller user screen, I am on a wider one). -DePiep (talk) 12:46, 7 January 2013 (UTC)
- This was the feedback: [4]. -DePiep (talk) 16:33, 7 January 2013 (UTC)
- It looks quite good, noting your to-do's. Educators would appreciate. Sandbh (talk) 23:53, 8 January 2013 (UTC)
- Why not just use mouseover text? It can be readily explained ("Hovering over a cell in the periodic table will cause [insert desired information] to be displayed, etc.") and doesn't require people to click on the element names, and yet is not as cluttered. Double sharp (talk) 12:46, 9 January 2013 (UTC)
- Mouseover text is only a supporting means, not a primary information thing. (see WP:NOSYMBOLS: Do not use techniques that require interaction to provide information, such as tooltips or any other "hover" text.). And this is not just a WP-link to block off your argument, but because all screen-user interaction these days has subltle differences and meanings. The whole topic is under WP:ACCESS. That includes taking care of variant situations such as printing, mobiles, or disabled readers. As for priorities, I thing a reader might expect the full name to be there. -DePiep (talk) 13:12, 9 January 2013 (UTC)
- I'd build in a switch that switches the normal PT and the extended one (i.e., with names), but I don't know if this is possible for the wiki code. Is it?--R8R Gtrs (talk) 07:58, 10 January 2013 (UTC)
- AFAIK, not possible in the way you imagine. First, it would require a readers action to show main information (bad. see wp:access prev reaction, above). So the high-end WP interface designers here do not like that, with reason, and so they have not build such a thing. That is why we (editors) do not have that option. Even the show/hide button in a table is advised to be in (secondary!) navboxes only. (I tried it somewhere else, about Unicode strange characters, to no effect). Second, there could be a javascript extension or so for this, but that is not generally (for all readers) available. Maybe I'll ask around once more. -DePiep (talk) 09:03, 10 January 2013 (UTC)
- I'd build in a switch that switches the normal PT and the extended one (i.e., with names), but I don't know if this is possible for the wiki code. Is it?--R8R Gtrs (talk) 07:58, 10 January 2013 (UTC)
- Mouseover text is only a supporting means, not a primary information thing. (see WP:NOSYMBOLS: Do not use techniques that require interaction to provide information, such as tooltips or any other "hover" text.). And this is not just a WP-link to block off your argument, but because all screen-user interaction these days has subltle differences and meanings. The whole topic is under WP:ACCESS. That includes taking care of variant situations such as printing, mobiles, or disabled readers. As for priorities, I thing a reader might expect the full name to be there. -DePiep (talk) 13:12, 9 January 2013 (UTC)
- Why not just use mouseover text? It can be readily explained ("Hovering over a cell in the periodic table will cause [insert desired information] to be displayed, etc.") and doesn't require people to click on the element names, and yet is not as cluttered. Double sharp (talk) 12:46, 9 January 2013 (UTC)
- It looks quite good, noting your to-do's. Educators would appreciate. Sandbh (talk) 23:53, 8 January 2013 (UTC)
Todo's: I set up a smaller screen size (regular flat: 1280-1024; old tube 1024-768). Had to squeeze squeeze out more pixels in the width (the height is no problem), even dropped the occurrence cellframes (4px per cell), but still could not get the 18 cols within the page. Not solved yet. -DePiep (talk) 09:03, 10 January 2013 (UTC)
- I have worked into this version: {{Periodic table/sandbox}}. These are the compromises (costs): 1. remove frame (border; occurrence) to save px width. 2. Reduce name font to 80%, about smallest aceptable legible. 3. Introduce hyphenation in the names. (I think the names are more important than the frames). Still, it is too wide for small screens: at (old tube) 1024x768 group 18 is off the page (requires horizontal scrolling). Will ask at WP:VPT what to do. -DePiep (talk) 18:40, 28 January 2013 (UTC)
- I do not really like the way you reduced the frames to single lines, as now it is unclear to the reader whether a frame is applying to Te or Po (or any two elements directly above each other), for example. Double sharp (talk) 15:25, 29 January 2013 (UTC)
- As it is now, it is experimental (just trying). It can be left out, and it can be tweaked (away from the bottom?). One fact is certain: if we add the name, there is no width enough for the border-code (in 1024x768 screen). Now the occurrence is a descriptive property, the name is a identifying property, so I made the name(-argument) prevailing. Remember, very very few of our readers are that familiar with the PT that they know where to find "gold". Also I ask to consider that WP:ELEM editors are very familiar with the PT as it is, but that is not a good guidance ('liking' or 'why change at all'?). -DePiep (talk) 15:42, 29 January 2013 (UTC)
- Meanwhile, that vertical separation has been changed. -DePiep (talk) 16:37, 29 January 2013 (UTC)
- At the moment, it looks do-able. That is: not even a compromise needed (such as: "allow table being too wide for small screen"). I repeat: not. Yes there are changes to our habitual view, but I hope ELEM-editors here can see and approve the improvement. See {{Periodic table/sandbox}}. -DePiep (talk) 17:01, 29 January 2013 (UTC)
- The lack of a complete border makes it a bit difficult to tell which element the border code belongs to, even with the extra space added between rows. Maybe have top and bottom borders? Or even partially restore side borders in the lower half of the cell. What I have in mind is a big cell with a top border only and two lines of text, then an interior single-celled table with left, right and bottom borders, something like this:
- At the moment, it looks do-able. That is: not even a compromise needed (such as: "allow table being too wide for small screen"). I repeat: not. Yes there are changes to our habitual view, but I hope ELEM-editors here can see and approve the improvement. See {{Periodic table/sandbox}}. -DePiep (talk) 17:01, 29 January 2013 (UTC)
- Meanwhile, that vertical separation has been changed. -DePiep (talk) 16:37, 29 January 2013 (UTC)
- As it is now, it is experimental (just trying). It can be left out, and it can be tweaked (away from the bottom?). One fact is certain: if we add the name, there is no width enough for the border-code (in 1024x768 screen). Now the occurrence is a descriptive property, the name is a identifying property, so I made the name(-argument) prevailing. Remember, very very few of our readers are that familiar with the PT that they know where to find "gold". Also I ask to consider that WP:ELEM editors are very familiar with the PT as it is, but that is not a good guidance ('liking' or 'why change at all'?). -DePiep (talk) 15:42, 29 January 2013 (UTC)
- I do not really like the way you reduced the frames to single lines, as now it is unclear to the reader whether a frame is applying to Te or Po (or any two elements directly above each other), for example. Double sharp (talk) 15:25, 29 January 2013 (UTC)
| Element name |
| A # |
- I believe the way I have set this up, the vertical space for the margin is still taken up and so there are no extra pixels for the element name. However, I believe that you could use a nested table for the lower half of the cell so that the pixels for the left and right border would not take space away from the element name. YBG (talk) 05:46, 30 January 2013 (UTC)
- First I think that there is no confusion left on which cell the border belongs to, and so that is no reason to change. Then about your proposal. Indeed it could be constructed within the cell, horizontally. But horizontal inter-cell space is only 1px, which is unpleasant small between two such borders. Visually too less separation. Also, the U creates visually a break within the cell. As if adding a horizontal cut halfway (strengthened by there being a row of such cells). The basic cell structure is very powerful for the eye, because one recognises the structure easily without having to look twice. That visible structure would be distracted from. So I do not think that is an improvement over the bottom-bar. -DePiep (talk) 11:21, 30 January 2013 (UTC)
- I agree with most of your comments. However, for me there is still a bit of confusion – I find that I have to look carefully at the top row and the bottom row to figure out for sure whether the colored line is on the bottom or on the top of each cell. It doesn't jump right out at me. The following I think is a bit of an improvement over the previous one; admittedly, this only partly rectifies the problem of the U creating a visual break.
- First I think that there is no confusion left on which cell the border belongs to, and so that is no reason to change. Then about your proposal. Indeed it could be constructed within the cell, horizontally. But horizontal inter-cell space is only 1px, which is unpleasant small between two such borders. Visually too less separation. Also, the U creates visually a break within the cell. As if adding a horizontal cut halfway (strengthened by there being a row of such cells). The basic cell structure is very powerful for the eye, because one recognises the structure easily without having to look twice. That visible structure would be distracted from. So I do not think that is an improvement over the bottom-bar. -DePiep (talk) 11:21, 30 January 2013 (UTC)
- I believe the way I have set this up, the vertical space for the margin is still taken up and so there are no extra pixels for the element name. However, I believe that you could use a nested table for the lower half of the cell so that the pixels for the left and right border would not take space away from the element name. YBG (talk) 05:46, 30 January 2013 (UTC)
| Mo |
| Molyb- denum |
| 42 |
- Notice that there is background color in the margin outside the two U-shaped borders; I couldn't figure out how to get rid of this. The two U's visually pull the cell together, but the horizontal band of a borderless region will still tend to break things apart. If you still think this is visually distracting, then I would suggest that you at least have both top and bottom borders and increase the whitespace between the rows. Thanks for your hard work trying to include the element names. It is much appreciated. YBG (talk) 05:56, 31 January 2013 (UTC)
- re, details: Thanks for the written appreciation, well received. The pink outer line you mention we can get rid of, but then you get two 2px wide borders (dark colored) with just a 1px white vertical between. With me (in my eyes), these borders then tend to flow over (or mix up, or flicker), a visual effect. See the current {{periodic table}}. Another detail: this layout changes the vertical sequence into number-name-symbol, while most other templates have sequence: (name, if present)-number-symbol. So putting the name in between would break that similarity over our templates. Such similarity is great to get first-glance recognition. As a third, I think the (interrupted) border adds too many details in a tight space. Not cluttered maybe, but 'bizzy' I'd say. And indeed the optical line over cells that is not part of the PT structure. The bottom whitespace can be 3px from 2px now, I found it a bit too much. I will give it a try. Doing both a top and bottom bar as you suggest: see my questions below. One more point: in the end the legend should show the same border pattern, of course (todo).
- re, in general. Now my question to ELEM editors is, how important is the border-information (natural occurrence), and how important is it that it should look like the known border. The importance, of course, is no problem since we can add the info to the cell. Question asnswered: it can be there for almost free. Second question: how then? My proposal is the single bottom bar. Trying to create a sort of old style border, as all around as possible, is visually cluttering or distracting (into details, into optical non-structures). So my question is: why is the square border form an aboslute requirement, or can I ask you to step over a habit, and see the elegance in the proposed solution. Of course, if you editors are all going to protest in front of me here, we should get as much squares as possible. But please do only after consideration. -DePiep (talk) 17:35, 31 January 2013 (UTC)
- I agree that there is benefit in maintaining a consistent order of presentation between the different PT presentations. I'm not sure whether I'd prefer bottom-only border or a top & bottom border ... it may be that with the wider white space between rows, the single bottom row would work OK. An interesting side note ... having the border only on the bottom makes the border lines visually run together, which at first sounds like it would be a negative. But on the other hand, it does accentuate the fact, for example, that there is a long string of naturally occurring elements broken by the single exception of Tc. So over all, I think I'll give up on the idea of trying to use a U-shaped partial border.
- In addition to comparing the bottom-only with top+bottom, I'd like to consider yet another way of representing this information. What about putting the border just around the atomic number or atomic symbol? Like the partial border, it represents a departure from the normal PT representation, but I think it is one worth at least considering.
- Thanks once again. The element name is a rally important piece of information and it is worth the effort you are putting in to make it more easily accessible. YBG (talk) 04:32, 1 February 2013 (UTC)
- I do not favor the double line (top+bottom) nor the boxing the atomic number ideas. Too many visual elements that do not really add informatiion or clarification. And I too noticed the bar series that pop up clearly, which is a nice extra. I can shorten the bar horizontally (away from cell edges). Whitespace below now is 3px (I think 2px was OK, but alas).
- Now I am not unwilling to try your suggestions in the sandbox. It's just that I try to react here first. Most of the ideas have gone through my mind (and keyboard) too, earlier in the process. That is why I can formulate my reasoning here, mostly about visual effects. The only reasons for using some kind of box so far is that we, project editors, are used to it. And of course it is our standard. Only in this one it is too crowded. -DePiep (talk) 15:07, 1 February 2013 (UTC)
- OK, sounds good. Thanks for being patient with my ideas. It is patience like this that helps develop consensus at WP. YBG (talk) 03:57, 2 February 2013 (UTC)
- Observing patience is an excercise for me too. -DePiep (talk) 12:33, 2 February 2013 (UTC)
- OK, sounds good. Thanks for being patient with my ideas. It is patience like this that helps develop consensus at WP. YBG (talk) 03:57, 2 February 2013 (UTC)
- Notice that there is background color in the margin outside the two U-shaped borders; I couldn't figure out how to get rid of this. The two U's visually pull the cell together, but the horizontal band of a borderless region will still tend to break things apart. If you still think this is visually distracting, then I would suggest that you at least have both top and bottom borders and increase the whitespace between the rows. Thanks for your hard work trying to include the element names. It is much appreciated. YBG (talk) 05:56, 31 January 2013 (UTC)
May I conclude that we have a consensus to add the names as is done in {{Periodic table/sandbox}}? -DePiep (talk) 12:33, 2 February 2013 (UTC)
- I still don't like it.
- Not every PT has names. The version of the PT we show our readers on the first page, IMHO, should have only the things every PT has: atomic numbers and symbols. The PT article itself states that some PTs include more info, while some don't. IMHO there should be links from the template (transcluded to the article) to PTs containing more info, but such PTs should not IMHO be in the main article, as they may have too much info, may scroll off smaller screens, and may distract from the text in the article. Double sharp (talk) 13:06, 2 February 2013 (UTC)
- I have to agree. The standard table gains nothing as we already have three other tables with the names. Instead, the proposed change adds to much height (especially with narrow screens), and it doesn't make it any easier to spot an element by name; it's quite hard to spot one out of 118 names with this small font and wrapped words. It also breaks consistency with the other tables in terms of presentation. This is the basic table; it should retain it's basic layout. — Edokter (talk) — 13:58, 2 February 2013 (UTC)
- (edit conflict) First of all: this periodic table does not exceed page width. -DePiep (talk) 14:05, 2 February 2013 (UTC)
 .
.
- Yes, and it achieves that by splitting the words, which reduces readability. Double sharp (talk) 14:17, 2 February 2013 (UTC)
- Yes you say: then please do not bring it up repeatingly. -DePiep (talk) 21:40, 2 February 2013 (UTC)
- Yes, and it achieves that by splitting the words, which reduces readability. Double sharp (talk) 14:17, 2 February 2013 (UTC)
Took a closer look. I now see two things to look at:
1. Cells' widths are not equal. Moreover, illogically unequal, not matching lengths of the elements' names. While it's OK on my laptop the way I normally view it (1600x900, Google Chrome), it gets much worse on lower resolutions. For example, if the word "osmium" can get split up ("os-mium"), "vanadium" somehow manages to stay unbroken (1024x768, Safari). In both versions, groups 8 and 9 are unexpectedly thin, esp. compared to 5 and 10.
2. I'd also suggest moving the abundance line a little higher, so it could be easier distinguished from the cell below. I initially failed to recognize ununoctium as synthetic just a few mins ago.
I'd also give the period nums just a little more space, but I don't insist. Pretty decent anyway.--R8R Gtrs (talk) 19:55, 22 February 2013 (UTC)
- Oh please. However good your intentions R8R, let it go. I claim that the sandbox, as it is today, is a great improvement to WP:periodic table. But both User:Double sharp and User:Edokter spoiled weeks of my designing and work just by saying "I don't like it" or "It is not what I am used to" or "a pixel too much on the left". Whatever I do, they keep finding a pixel wrong (without getting the picture). It still makes me angry. Just workshop-enclosed technicians who cannot understand that we need to communicate the PT to outsiders. Ten months I am a guest in this WP:ELEM community, and I learned this: those scientists do not know the diff between their own happy bathroom and the WP reader's mind. "I don't like it" as an argument. Sure. -DePiep (talk) 00:22, 23 February 2013 (UTC)
- Agreed, the thing's good. I already said that a few times and still feel so. If we replaced the current scheme with it just now, I'd be happy. I myself remember a few things you changed I was uncomfortable with (but kept aside discussions), but now wouldn't want back as I was unable to see your things were superior then but not now. I like your designing and stuff. I just think there's one step to perfection left. But if we're having a discussion of approval, I'd vote for it anyway. Just hoping you wouldn't forget to think about those if the decision were on your side.
- I like the lines (was originally thinking they were poorly designed, now think they're essential to your scheme's minimalism), I like how it suits into a screen (I think that unun- (break) octium initially reads better than if the cell didn't suit into the screen at all), I like that it is still simple and not ugly (still easy to read out info, amazing given it has much more).
- tl;dr we had a 6, you've made a 9. I think I know how to make it a 10, but 9 is a lot greater than 6 anyway.
- Opponents, please comment--R8R Gtrs (talk) 19:58, 23 February 2013 (UTC)
- I think it is OK now except for the occurrence line, which is hard to recognize. I still think that the basic PT should show the basic layout, but would not mind a change to this. (But we should really show both, perhaps with a click to flip between them if that is possible or allowed, just in case readers start thinking that every PT has names...) Double sharp (talk) 08:12, 24 February 2013 (UTC)
- Thank you, User talk:Double sharp, for this opening and inviting reply. As one could sense & read, I have refrained weeks & days from replying here. I will write more later on. -DePiep (talk) 23:26, 25 February 2013 (UTC)
- However clumsy I wrote it: appreciated. And we'll be ok, talk ahead. -DePiep (talk) 23:41, 25 February 2013 (UTC)
- I would be happy with it if only the occurrence lines were easier to distinguish. Can we turn it back to a border? Or at the very least border both the top and the bottom of the element cell? This way there won't be errors in readers' deciding which cell the border applies to (the one above, or the one below?) Of course they will come to a realization in the 1st and 7th periods, but that assumes they do look at the 1st and 7th periods. What if they are checking what element "Rh" is? It's far from the edges, and is in a sea of transition-metal colour (making borders even harder to distinguish, seeing as here external visual clues are few and far between). Double sharp (talk) 14:24, 23 March 2013 (UTC)
- I'll add the same line to the top of the cell. A vertical border (left, right) takes up pixels (width) in the horizontal direction, in which direction space is scarce. Ideas for an incomplete border, like horseshoe for example, have been demo'ed and discussed above in this talk. -DePiep (talk) 14:39, 23 March 2013 (UTC)
- Looks good. Double sharp (talk) 15:07, 23 March 2013 (UTC)
- Like a cutaway pie :) Don't want to insult anyone, just an association--R8R Gtrs (talk) 15:18, 23 March 2013 (UTC)
- Looks good. Double sharp (talk) 15:07, 23 March 2013 (UTC)
- I'll add the same line to the top of the cell. A vertical border (left, right) takes up pixels (width) in the horizontal direction, in which direction space is scarce. Ideas for an incomplete border, like horseshoe for example, have been demo'ed and discussed above in this talk. -DePiep (talk) 14:39, 23 March 2013 (UTC)
- I would be happy with it if only the occurrence lines were easier to distinguish. Can we turn it back to a border? Or at the very least border both the top and the bottom of the element cell? This way there won't be errors in readers' deciding which cell the border applies to (the one above, or the one below?) Of course they will come to a realization in the 1st and 7th periods, but that assumes they do look at the 1st and 7th periods. What if they are checking what element "Rh" is? It's far from the edges, and is in a sea of transition-metal colour (making borders even harder to distinguish, seeing as here external visual clues are few and far between). Double sharp (talk) 14:24, 23 March 2013 (UTC)
- However clumsy I wrote it: appreciated. And we'll be ok, talk ahead. -DePiep (talk) 23:41, 25 February 2013 (UTC)
- Thank you, User talk:Double sharp, for this opening and inviting reply. As one could sense & read, I have refrained weeks & days from replying here. I will write more later on. -DePiep (talk) 23:26, 25 February 2013 (UTC)
- I think it is OK now except for the occurrence line, which is hard to recognize. I still think that the basic PT should show the basic layout, but would not mind a change to this. (But we should really show both, perhaps with a click to flip between them if that is possible or allowed, just in case readers start thinking that every PT has names...) Double sharp (talk) 08:12, 24 February 2013 (UTC)
Need to fix the border in the legend at the bottom. YBG (talk) 19:11, 23 March 2013 (UTC)
- Will be done, promise. It's just we want
mythe great plan done: element name in the basic PT. ;-) -DePiep (talk) 23:23, 23 March 2013 (UTC)- Done. It even looks nice to me. See below for the use of "natural radio". -DePiep (talk) 17:56, 24 March 2013 (UTC)
- One more hiccup: in my Safari browser the widths do not line up well. So there are not 18 equal distances. That is bad. -DePiep (talk) 22:45, 2 April 2013 (UTC)
- Cell width problem looks solved (please check Chrome, Safari, ...). Some internal cleaunup left to do. -DePiep (talk) 10:41, 12 April 2013 (UTC)
- One more hiccup: in my Safari browser the widths do not line up well. So there are not 18 equal distances. That is bad. -DePiep (talk) 22:45, 2 April 2013 (UTC)
- Done. It even looks nice to me. See below for the use of "natural radio". -DePiep (talk) 17:56, 24 March 2013 (UTC)
- Final checks
Please check the {{Periodic table/sandbox}} once more. If there are no prohibitive issues, I suggest we put it live. Note that tweaking is always possible. -DePiep (talk) 13:50, 12 April 2013 (UTC)
- Cld 113 be shown as unun-trium rather than ununtr-ium? Sandbh (talk) 00:11, 13 April 2013 (UTC)
- Done. -DePiep (talk) 01:02, 13 April 2013 (UTC)
![]() Done. Element names in periodic table: live now. -DePiep (talk) 19:24, 14 April 2013 (UTC)
Done. Element names in periodic table: live now. -DePiep (talk) 19:24, 14 April 2013 (UTC)
Metalloid FA
I intend to nominate metalloid for FA. Does anyone have any comments before I proceed? Sandbh (talk) 11:26, 26 January 2013 (UTC)
- I would strongly suggest getting somebody outside of the project to copyedit it before putting it there. Things will be a lot smoother that way. Try with user:Sasata. Nergaal (talk) 14:57, 26 January 2013 (UTC)
- That worked well thank you Nergaal. I'm currently incorporating Sasata's feedback. Sandbh (talk) 11:30, 2 February 2013 (UTC)
- English variety. As part of climbing the FA nomination mountain, the article needs to use a consisent English language variety. The first non-stub version dated Sep 21, 2009 had the word 'behaviour' in it, which is a British (BrE) spelling. I've been more or less editing the article in Australian English (AusE) apart from being notably inconsistent with my use of –ise endings v –ize endings as in, for example, characterise v recognize (I introduced 'characterise' on Jul 4, 2011). Choices as I see it are American English, since much of the literature about metalloids is American; AusE, which is close to but not quite the same as BrE; or BrE. At the moment I've updated the article to be AusE-consistent e.g. colour instead of color; characterise, delocalised and recognised instead of their –ize variants; however it can easily be rendered in another variety. Sandbh (talk) 00:57, 17 February 2013 (UTC)
- Heh, I'm also very inconsistent about ise/ize. I haven't checked which I use more often in Alkali metal yet. ;-) Double sharp (talk) 02:22, 17 February 2013 (UTC)
- Copyediting all done, for now. I intend to review two FACs, before nominating metalloid. Sandbh (talk) 23:54, 1 March 2013 (UTC)
- I reviewed and contributed to the FACs for KFC and Arthur W. Radford. Metalloid FAC nomination to follow shortly. (I now find myself intently watching/analysing KFC commericals on TV, however.) Sandbh (talk) 04:54, 11 March 2013 (UTC)
- Well, I went ahead and nominated it. Sandbh (talk) 08:12, 11 March 2013 (UTC)
- I reviewed and contributed to the FACs for KFC and Arthur W. Radford. Metalloid FAC nomination to follow shortly. (I now find myself intently watching/analysing KFC commericals on TV, however.) Sandbh (talk) 04:54, 11 March 2013 (UTC)
- English variety. As part of climbing the FA nomination mountain, the article needs to use a consisent English language variety. The first non-stub version dated Sep 21, 2009 had the word 'behaviour' in it, which is a British (BrE) spelling. I've been more or less editing the article in Australian English (AusE) apart from being notably inconsistent with my use of –ise endings v –ize endings as in, for example, characterise v recognize (I introduced 'characterise' on Jul 4, 2011). Choices as I see it are American English, since much of the literature about metalloids is American; AusE, which is close to but not quite the same as BrE; or BrE. At the moment I've updated the article to be AusE-consistent e.g. colour instead of color; characterise, delocalised and recognised instead of their –ize variants; however it can easily be rendered in another variety. Sandbh (talk) 00:57, 17 February 2013 (UTC)
- That worked well thank you Nergaal. I'm currently incorporating Sasata's feedback. Sandbh (talk) 11:30, 2 February 2013 (UTC)
Predicted alkali metal colour
The current colour , while a reasonable choice, is not that similar to the basic alkali metal colour . zh.wikipedia uses , which is more clearly related, and still readily distinguishable from transition metal . Should we change it? Double sharp (talk) 05:25, 17 February 2013 (UTC)
- I have put
#ffa1a1for predicted alkali metalin the Wikipedia:WikiProject Elements/Legends/sandbox. Note: current color settings are at hand in project page Guidelines. -DePiep (talk) 11:41, 17 February 2013 (UTC)
| Proposal | Current | Current |
|---|---|---|
| Alkali metal #ff6666 |
Alkali metal #ff6666 |
Transition metal #ffc0c0 |
| predicted #ffa1a1 |
predicted #d8bcbc |
predicted #ffe2e2 |
-DePiep (talk) 12:02, 17 February 2013 (UTC)
- zh: uses the darker color, same as en:, here: zh:Uue. Where did you see the lighter one? -DePiep (talk) 12:17, 17 February 2013 (UTC)
- This is my opinion: better not change it. The proposed color indeed is more nicely associated with the red for alkali metal. But as a color, it is too close to the transition metal color. Remember it is a legend color, so the reader must be able to connect the color to the legend (explanation). Since these colors would be very similar, the reader might mistakingly end up at the wrong legend definition (with noone and nothing there to warn or prevent that). First and foremost, we must prevent such mistakes. -DePiep (talk) 12:24, 17 February 2013 (UTC)
- I found the lighter colour at zh:Template:擴展元素週期表. Double sharp (talk) 13:42, 17 February 2013 (UTC)
- I think you're too concerned with that 119 will be taken for a TM. An almost as red cell under a row of red cells is normally taken for continuation of the row, and the same is true for TMs. That said, if you can distinguish them, good. No, then no, okay, nobody will mind or have problems with that, I assume. Our current future (yeah) AM color is not any good. The Chinese choice is reasonable. (why do all East Asian Wikis use terrible fonts for Latin characters?)--R8R Gtrs (talk) 22:46, 30 March 2013 (UTC)
- zh: uses the darker color, same as en:, here: zh:Uue. Where did you see the lighter one? -DePiep (talk) 12:17, 17 February 2013 (UTC)
Electron "shells"
When did we arrive at a consensus to include simplistic diagrams like File:Electron shell 092 Uranium - no label.svg in infoboxes? Especially for the heavier elements these seem so highly misleading as to be counter-productive. I would accept these for lighter elements (e.g. sodium) in the body of the article with a warning. Not for anything beyond about argon though. What do others think? --John (talk) 11:54, 17 February 2013 (UTC)
- AFAIK it is a graphic of this. We could add the word "simplified" to it in the infoboxes. But how is it misleading and counter-productive? What warning do they need? -DePiep (talk) 12:12, 17 February 2013 (UTC)
- Well, electrons going in circles around the nucleus was the Bohr model of the atom. That was superseded by quantum mechanics in 1925, so the diagram is now 88 years out of date. Chris (talk) 12:27, 17 February 2013 (UTC)
- I agree it is somewhat misleading. But it is still a useful picture, despite being inaccurate. The quantum mechanical model has the subshells (1s, 2s/p, 3s/p/d, 4s/p/d/f, 5s/p/d/f/g, etc.) This picture doesn't show the subshells, only the shells (and shows these as orbits, which I'm sure you know is not true, but is nevertheless a useful mental picture). The quantum mechanical model is hard to show in an image in the same way this model can be shown. We do include the quantum mechanical model configuration in the same place, and even show it above the picture. (The picture has no label, so readers who don't want to count around 32 electrons will look at the text above it).
- Also, if we're going to have the pictures on the lower elements, we should have them on all the elements. Otherwise readers will be wondering where they went. And after all, the electrons still do occupy these shells (see above about their representation as orbits), so it is not completely wrong, just somewhat misleading about how it conveys its information (and there's not much better for this kind of thing). Double sharp (talk) 14:04, 17 February 2013 (UTC)
- I agree, it is better not to have them at all. The text carries the same information more concisely anyway. I take it there was no consensus to add them in the first place and someone just did it? --John (talk) 19:34, 17 February 2013 (UTC)
- You agree with whom or what? -DePiep (talk) 20:31, 17 February 2013 (UTC)
- I agree with Double sharp when he says "if we're going to have the pictures on the lower elements, we should have them on all the elements" and also "somewhat misleading about how it conveys its information". Unless there's a strong consensus that these are worth keeping, I'm going to start removing them in 24 hours or so. They should not have been added without a strong consensus to do so. --John (talk) 20:49, 17 February 2013 (UTC)
- Double sharp adds the word somewhat as you quote, and you left a lot more out of the quote. For example: still a useful picture. And no, you cannot remove them on a basis of your self-constructed consensus criterium. For starters, as there is no consensus here, there will be no change. -DePiep (talk) 21:23, 17 February 2013 (UTC)
- Er, no. Unless you can show me a consensus where it was agreed to include these misleading primary-school graphics to our articles, or unless a strong one emerges very soon (I can hold off a day or two if you think it matters) I will start removing them. The onus is definitely on you to show good reason if you want them to be retained for some reason. --John (talk) 21:34, 17 February 2013 (UTC)
- It is up to you, John, to build consensus for any change. Imposing a deadline is not part of that. -DePiep (talk) 12:45, 18 February 2013 (UTC)
- I'd say these pictures are useful for a simple look at the way electrons fit into shells, as long as there's some way to indicate that the model is no longer the one used. And, I will say, the Bohr model is easier to depict as an image than the electron cloud model we use these days.King Jakob C2 21:53, 17 February 2013 (UTC)
- They were already in the infoboxes, just in very small size in the periodic table. DePiep added them with the electron configuration information a while ago. As for consensus, see Wikipedia talk:WikiProject Elements/Archive 12#Proposal: move the electron shells image in the infobox. You will notice that, in that discussion, nobody suggested removing the pictures (that were already there).Double sharp (talk) 11:56, 18 February 2013 (UTC)
- Yep. And still, we are not to "prove" consensus for what is here under threat of a deadline. If John or anyone else wants to propose and discuss an improvement, that is the route to go. Just build consensus for that change. Until now, in this thread, I get the impression that John is not engaging in the discussion he started himself. For example, already in the second post I asked some questions which are neglected. So far I feel not invited to think about improvements. -DePiep (talk) 12:45, 18 February 2013 (UTC)
- I am sorry if I have in some way hurt your feelings, DePiep. I see you were one of the instigators of the idea to make these diagrams enormous, and I am sure you must have had some good intentions in doing so. I am still however waiting for an answer to my initial question; "When did we arrive at a consensus to include simplistic diagrams"? They weren't always there. Sorry if that is a lot to ask. Or do I have to dig through hundreds of edits to find out? --John (talk) 17:38, 18 February 2013 (UTC)
- John, let me see if I can summarize your point of view:
- The electron shell diagrams are problematic
- You want to speedily delete them in the absense of a consensus otherwise
- This consensus should be reached in a matter of days
- You would also accept a reference to a past consensus
- The burden of proof is on those who want to retain the diagrams
- I'm not sure whether I have understood your view correctly, so some of my statements above may not be fully accurate. Please correct me if I have misunderstood you. I appreciate the fact that you chose to broach the question on this talk page instead of simply boldly making the change. However, please keep in mind that the fact that these diagrams have been around for a while (at least 7 months, if I understand the above discussion above). This, and the opposition your suggestion elicited, constitutes some sort of a consensus, although not a formal consensus in the WP sense of the term. May I suggest that best way to improve these infoboxes would be for you to agree not to make any bold changes until a (new?) consensus is reached, possibly in a new thread? Here is what I would suggest would be good topics to cover in a discussion:
- The probems with the current situation (Size, Bohr, ...)
- Potential alternatives (No diagram, New type of diagram, ...)
- Pros & cons of each alternative
- Hopefully such a discussion would either result in a change that would be an improvement over the present situation, or else a consensus that the present diagrams, while not perfect, are better than any of the alternatives considered. YBG (talk) 18:58, 18 February 2013 (UTC)
- re YBG: your description is finely to the point and appreciated. Note: these pictures were here long time before (in the infobox), but alas small sized. -DePiep (talk) 22:15, 18 February 2013 (UTC)
- John, let me see if I can summarize your point of view:
- I am sorry if I have in some way hurt your feelings, DePiep. I see you were one of the instigators of the idea to make these diagrams enormous, and I am sure you must have had some good intentions in doing so. I am still however waiting for an answer to my initial question; "When did we arrive at a consensus to include simplistic diagrams"? They weren't always there. Sorry if that is a lot to ask. Or do I have to dig through hundreds of edits to find out? --John (talk) 17:38, 18 February 2013 (UTC)
- Yep. And still, we are not to "prove" consensus for what is here under threat of a deadline. If John or anyone else wants to propose and discuss an improvement, that is the route to go. Just build consensus for that change. Until now, in this thread, I get the impression that John is not engaging in the discussion he started himself. For example, already in the second post I asked some questions which are neglected. So far I feel not invited to think about improvements. -DePiep (talk) 12:45, 18 February 2013 (UTC)
- They were already in the infoboxes, just in very small size in the periodic table. DePiep added them with the electron configuration information a while ago. As for consensus, see Wikipedia talk:WikiProject Elements/Archive 12#Proposal: move the electron shells image in the infobox. You will notice that, in that discussion, nobody suggested removing the pictures (that were already there).Double sharp (talk) 11:56, 18 February 2013 (UTC)
- Er, no. Unless you can show me a consensus where it was agreed to include these misleading primary-school graphics to our articles, or unless a strong one emerges very soon (I can hold off a day or two if you think it matters) I will start removing them. The onus is definitely on you to show good reason if you want them to be retained for some reason. --John (talk) 21:34, 17 February 2013 (UTC)
- Double sharp adds the word somewhat as you quote, and you left a lot more out of the quote. For example: still a useful picture. And no, you cannot remove them on a basis of your self-constructed consensus criterium. For starters, as there is no consensus here, there will be no change. -DePiep (talk) 21:23, 17 February 2013 (UTC)
- I agree with Double sharp when he says "if we're going to have the pictures on the lower elements, we should have them on all the elements" and also "somewhat misleading about how it conveys its information". Unless there's a strong consensus that these are worth keeping, I'm going to start removing them in 24 hours or so. They should not have been added without a strong consensus to do so. --John (talk) 20:49, 17 February 2013 (UTC)
- You agree with whom or what? -DePiep (talk) 20:31, 17 February 2013 (UTC)
- I agree, it is better not to have them at all. The text carries the same information more concisely anyway. I take it there was no consensus to add them in the first place and someone just did it? --John (talk) 19:34, 17 February 2013 (UTC)
- Well, electrons going in circles around the nucleus was the Bohr model of the atom. That was superseded by quantum mechanics in 1925, so the diagram is now 88 years out of date. Chris (talk) 12:27, 17 February 2013 (UTC)
- Good summary. A good alternative, and one I have already offered, is just to have the numbers. That tends to be what post-primary-school resources in the real world do, as it's less misleading. --John (talk) 19:39, 18 February 2013 (UTC)
- John, you write about hurt your [my] feelings and such: that is you getting personal. I was just asking topical questions. Invoking AGF after you imposed your non-discussed deadline is funny, sort of. -DePiep (talk) 22:07, 18 February 2013 (UTC)
- I was responding to your So far I feel not invited to think about improvements. If you don't want to talk about your feelings, don't talk about your feelings. That would suit me. I am just here to try to improve this mess we seem to have here.--John (talk) 20:56, 19 February 2013 (UTC)
- John, you write about hurt your [my] feelings and such: that is you getting personal. I was just asking topical questions. Invoking AGF after you imposed your non-discussed deadline is funny, sort of. -DePiep (talk) 22:07, 18 February 2013 (UTC)
- John, please tell us: which numbers? What numbers? Above you wrote this: I would accept these for lighter elements. So which element numbers would you agree on? Oh please, tell us what you want. -DePiep (talk) 22:24, 18 February 2013 (UTC)
- Which numbers? The electron arrangements of course; that's what we're talking about, isn't it? For uranium that would be [Rn] 5f3 6d1 7s2; 2, 8, 18, 32, 21, 9, 2. This is an appropriate and meaningful way to show the information, and it is what real-world sources would show. The Bohr diagram; well, what do you think it adds, since you are the one defending it? --John (talk) 20:56, 19 February 2013 (UTC)
(The text's getting too thin)
What they're trying to say is, "we don't know. Rather than wondering a million times, just 'dig through hundreds.' Anyway, we're ready to talk, but don't you push it." It's a doable thing: there is some way to keyword-search only in Project's archives (I don't know how, but there are technical help pages on Wiki, should be easy to find out). Or contact the people who inserted the thing (look at templates' histories, pic creators, etc), ask'em. If after doing this no info comes up, then ask for a change, offer a discussion. Any rational idea can make it through a discussion. May sound like shit (no feeling like I wanna!). Still, the fact you work on your position much increases your chances to be listened to (not necessarily agreed with).
Oh well, you were just offered a discussion above. Go 'head and discuss. Don't pressurize people for no reason -- the hate it.
Also, just food for thoughts: Newton physics is false (it's just an approximation of what's really going on, and anytime our bodies may break apart 'cause our atoms may, and the screen you're reading this on may vaporize away, but the probabilities are neglectible), but ask RSA, or NASA, or ESA, or whoever if it's useful.--R8R Gtrs (talk) 19:17, 18 February 2013 (UTC)
- What is this? Is this really R8R? -DePiep (talk) 22:18, 18 February 2013 (UTC)
- I guess I'm too tired IRL, have little time to sleep, a lotta work to do, etc. In such conditions, it freaks me out when people are wasting others' time asking questions an answer for which is searchable, and forcing others to ask for some alternative suggestions (I'd write "I don't know what it could be like instead, maybe X?", so a not so stressed talk begins). Nothing in person, John-- after all, when I have a few days off, I'll be at least not so picky.
- Also, DePiep: decent work on adding elements' names to the PT. The thing doesn't look overcrowded. Don't know what you even coud improve. Still somehow want to get an ability to click and have a large/short table displayed, but if it's not possible, then fine.--R8R Gtrs (talk) 19:29, 19 February 2013 (UTC)
- I get it. Let's say I was getting concerned ;-). The names-in-PT I will address elsewhere, it is not dead yet. -DePiep (talk) 23:25, 19 February 2013 (UTC)
- One month on, and no reason advanced for using these over-simplistic graphics. I've removed them from the infobox pending a reasoned consensus that they add something to the articles. I wouldn't be against using them in certain articles, eg the alkali metals, but not in the infobox please. This is not Simple Wikipedia. --John (talk) 21:02, 21 March 2013 (UTC)
- I reverted. No consensus so no change. -DePiep (talk) 10:31, 22 March 2013 (UTC)
- That wasn't very clever. Please see WP:DRNC. Saying "no consensus" is not a reason to add this dumbed-down and misleading graphic to our articles. Another 24 hours to come up with a better reason? I'm almost tempted to swing by WP:AIV but I won't as I know your intentions are good. Please have a think about this. --John (talk) 17:50, 22 March 2013 (UTC)
- As for DRNC you point to: it says ... merely .... Given this thread, that is not at hand. -DePiep (talk) 15:57, 23 March 2013 (UTC)
- John, I'd like to give some counter-arguments. Will I be listened to?
- I remember myself being 14 years old and started to get chemistry lessons. In the very first year (in the end of it), we made it to the Bohr model. And hat was the point when chemistry finally got me. Not that I was bored at lessons, and back then we looked in the Internet for the way to explode something. But when my teacher (not a really good one) told me at first about the Bohr model, this was the moment when I realized: it's so cool, not ever gonna stop doing it. This was a very easy way to understand how chemistry works. The whole science became so easy to understand. And you want to cut it off? I assume that there are thousand or millions people who were/are/will be thinking like me. There are reasons why this was/is/will be taught in schools.
- (Sorry, I already mentioned that) Newtonian physics is also outdated, it's an approximation of what's going on in the real world. 108 years out of date. Should we give it up as well?
- But if we had an instrument as useful so the change were easier to make (not limit, but alter our info on offer), then I'm probably on your side. But do we?
- A quote of yours: "...For uranium that would be [Rn] 5f3 6d1 7s2; 2, 8, 18, 32, 21, 9, 2. This is an appropriate and meaningful way to show the information, and it is what real-world sources would show. The Bohr diagram; well, what do you think it adds..."
- It's visualizing. To understand what's going on in the 2, 8, 18, 32, 21, 9, 2 notation, you should know that it means that two electrons revolve around the nucleus on the first energy level, 8 on the second one, and so on. But what if you don't?
- Remember how you were taught physics. Air resistance became vital only in the uni, even if it's not the real world without the air.--R8R Gtrs (talk) 19:32, 22 March 2013 (UTC)
- You appear to not like the diagrams because they are not an accurate portrayal of what is going on. That is very true, and is a reasonable stand if we are writing a work for people who already have a strong background in chemistry already. However, we are not writing solely for such people. In spite of their major flaws, the Bohr model does have very good pedagogical value. If you don't give a diagram of the info, you may well render it incomprehensible to those who do not already know what the string of numbers 2,8,18,19,9,2 means, much less the string of symbols [Xe]4f15d16s2 which Sam Kean described as "what looked for all the world like computer error messages" in The Disappearing Spoon. But with the diagram, you have a visual representation of the shells. It is not an accurate one, but it is a representation no less. In the Poincaré disc model, straight lines in the hyperbolic plane are represented as looking curved, and the entire infinite hyperbolic plane is compressed into a circle. They do not really look like that; if you were wandering around in a hyperbolic plane, you wouldn't see this. But it is still a very good representation of hyperbolic geometry, and is very commonly used. Similarly, if you could see what is going on at the atomic scale, you would not see electrons orbiting the nucleus in shells. But these electrons do have distinct energy levels, and the shells are just a way to represent it visually. Of course not everything can be represented well with a diagram: just try drawing a quantum-mechanics-included model of a cerium or uranium atom. (It would most likely end in a mass of messy scribbles.) But the Bohr model is not too far off (although things are not represented as they really are, the main points are sound and the representations have a consistent relation to reality). Finally, the pedagogical value of such a simplified (but still correct in a weaker way) model isn't to be underestimated. Ever wondered why teachers start with such models before progressing to accurate quantum-mechanical models? These are easier to understand and the basics "click" into place neatly. When the time comes that they have accumulated enough understanding to move on, their basics are sound, and now they can be told what really happens. And if the model is good, the difference between reality and the model is often just changing of details (like how things are represented) and not changing of main points (if I introduced a model where the nucleus revolves around an electron cloud, I doubt it would be accepted widely, simply because it is not a good model – the entire basic points are different from reality. But in the Bohr model, only how things are represented are different.). This way, the students do not unlearn, just learn more stuff. But if you remove the entire basic model, they now are plunging into the deep end, and can reasonably expected to come out of it not having a clue what they have just seen or read depicted. This is not really a desirable outcome, to say the least.
- tl;dr: It's fine to remove the diagram for people who already know what we are talking about. Yet not everyone does, and we should help them with a more basic simplified diagram. When such a diagram is only different from reality in how it represents things, and not its concepts, then it serves as a good entry point to learning reality. The Bohr model is one such diagram. For those who already know what we are talking about, the electron configuration is right there above the diagram. Having the diagram does not hurt anyone; not having it does. Double sharp (talk) 14:17, 23 March 2013 (UTC)
- Great, Double sharp. Thank you for clarifying. -DePiep (talk) 00:08, 24 March 2013 (UTC)
- Even better: already the first google page for The Disappearing Spoon Madness Periodic Elements (sic) made me laugh. "Why did Gandhi hate iodine (I, 53)?" -DePiep (talk) 21:22, 24 March 2013 (UTC)
- It's a rumour, which was perpetrated because the introduction of iodized salt to India (spurred on by the West) was regarded in India as having a whiff of colonialism (cf. the salt tax). Details in the book. Double sharp (talk) 12:54, 28 March 2013 (UTC)
- That wasn't very clever. Please see WP:DRNC. Saying "no consensus" is not a reason to add this dumbed-down and misleading graphic to our articles. Another 24 hours to come up with a better reason? I'm almost tempted to swing by WP:AIV but I won't as I know your intentions are good. Please have a think about this. --John (talk) 17:50, 22 March 2013 (UTC)
Main decay modes of isotopes
Previously I have noted that there is some contradiction in the Isotopes of ruthenium on the main decay modes of isotopes. The text states that lighter isotopes are mainly decay via electron capture, while the tables states no or only some EC and mostly beta-plus decay. Now it seems that this is a more general issue affecting several other articles (starting with Isotopes of scandium, and based on a quick search it occurs frequently). I am not sure what is the source of either information, but the table looks more reliable to me. Any thoughts on this? Szaszicska (talk) 09:16, 23 February 2013 (UTC)
- Quick explanation: whenever you have β+, you have EC. But having EC is not a guarantee for having β+. So whenever there is β+, we mention it alone without EC (which is implied). Only when there is EC but no β+ do we write "EC". We do this so that the table in the infobox doesn't get too large and also because since most isotopes have enough decay energy to go through both decay modes, it's quite uninteresting to write both all the time. It's easiest to explain it in a central article – although Positron emission is probably not the best possible choice.
- If you want to know more about the relationship between β+ and EC, read the last paragraph of the section Positron emission#Emission mechanism. Double sharp (talk) 11:27, 23 February 2013 (UTC)
- Just a thought: if it's not obvious to anyone, let's state it somehow. Plus if EC is implied, do the energies of decays match? And a mode half-time? It bears too many questions to the unaware to simply imply it.--R8R Gtrs (talk) 19:25, 23 March 2013 (UTC)
- Different DE, AFAIK. Otherwise this wouldn't be much of a problem.
- Half-life should be the same, but I'm not sure.
- But you're going to have a hard time searching for a source that gives both... Double sharp (talk) 02:23, 24 March 2013 (UTC)
- Writing a disclaimer "we're missing EC" would be fair at very least.--R8R Gtrs (talk) 15:35, 24 March 2013 (UTC)
- And yet that is also not really accurate: when EC is the only decay mode, we include it, so we are not completely missing EC. It would have to be something like "when β+ occurs, so does EC/ε" (and same for the much rarer pair β+β+ / εε). (What's the equivalent EC-style symbol for double electron capture?) Double sharp (talk) 12:52, 28 March 2013 (UTC)
- "We may be missing EC?", then? (I think there's no ECEC)--R8R Gtrs (talk) 14:50, 8 April 2013 (UTC)
- And yet that is also not really accurate: when EC is the only decay mode, we include it, so we are not completely missing EC. It would have to be something like "when β+ occurs, so does EC/ε" (and same for the much rarer pair β+β+ / εε). (What's the equivalent EC-style symbol for double electron capture?) Double sharp (talk) 12:52, 28 March 2013 (UTC)
- Writing a disclaimer "we're missing EC" would be fair at very least.--R8R Gtrs (talk) 15:35, 24 March 2013 (UTC)
- Just a thought: if it's not obvious to anyone, let's state it somehow. Plus if EC is implied, do the energies of decays match? And a mode half-time? It bears too many questions to the unaware to simply imply it.--R8R Gtrs (talk) 19:25, 23 March 2013 (UTC)
Strontium
I think we can go to a B-Class here. Please have a look and give a hint what is still missing to GA.--Stone (talk)
- A 30 sec look:
- even more reactive toward water than calcium -- to what extent? what if I know nothing about calcium? It can't be the only ref point. reacts on contact -- how else can anything react?
- Is it dense?
- 89Sr is a short-lived artificial radioisotope that is used in the treatment of bone cancer. In circumstances where cancer patients have widespread and painful bony metastases (secondaries), the administration of 89Sr results in the delivery of radioactive emissions (beta particles in this case) directly to the area of bony problem (where calcium turnover is greatest). The 89Sr is manufactured as the chloride salt (which is soluble), and when dissolved in normal saline can be injected intravenously. Typically, cancer patients will be treated with a dose of 150 MBq. Patients must take precautions following this because their urine becomes contaminated with radioactivity, so they must sit to urinate and double-flush the toilet. The beta particles travel about 3.5 mm in bone (energy 0.583 MeV) and 6.5 mm in tissue, so there is no requirement to isolate patients having been treated, except to say they should not have any one (especially young children) sitting in their laps for 10–40 days[citation needed]. The variation in time results from the variable clearing time for 89Sr, which depends on renal function and the number of bony metastases. With a lot of bony metastases, the entire 89Sr dose can be taken up into bone and so the radioactivity is retained to decay over a 50.5-day half-life. It takes about 10 half-lives or about 500 days for 99.9% of the radioactive strontium to decay. However, where there are few bony metastases, the large proportion of 89Sr not taken up by the bone will be filtered by the kidney, so that the effective half-life (a combination of the physical and biological half-life) will be much shorter. -- belongs to applications, plus should be shortened
- pyrotechnics is interesting. My head begins to generate question like "how much %% and g/kg/lb Sr(Whevr)2 can be there to remain safe?" BTW, the whole thing doesn't say anything about how to stay safe if you're breathed in the metal dust or stuff like that.
--R8R Gtrs (talk) 17:23, 17 March 2013 (UTC)
- My real life as rocket scientist will keep me busy, but I will work on it.--Stone (talk) 21:32, 18 March 2013 (UTC)
- Can we have more citations for the content in the "Isotopes" section? (Per R8R, much of it should also be moved.) Double sharp (talk) 06:09, 21 March 2013 (UTC)
- My real life as rocket scientist will keep me busy, but I will work on it.--Stone (talk) 21:32, 18 March 2013 (UTC)
Chalcogen FA
Anyone think chalcogen is ready for FA?King Jakob C2 13:25, 17 March 2013 (UTC)
- I personally have given it a quick look as of now and found some style problems. I haven't read it yet (can't judge the quality of the text), but style needs to be polished anyway prior to FAC. Like why "oxygen-16, oxygen-17, oxygen-18" but H2SO4, H2SO3, H2SeO4, and H2TeO4? Why does the reader need to know them all when he's got a graphic right under this para: "Ag, As, Bi, Cd, Cu, Ga, Ge, Hg, In, Pb, Po, S, Sb, Se, Sn, Te, Tl, and Zn." Vital ≠ nontoxic, we all die even in 100% O2, so it can't be an antithesis (this can be a better one: good in small quantities, bad in large), etc, etc.
- I extremely recommend a peer review.--R8R Gtrs (talk) 19:58, 22 March 2013 (UTC)
- FA? Get ready to be whacked. ;-) I would extremely recommend everything R8R extremely recommends, and would also recommend looking at FAs of other groups (noble gas, although you may also consider looking at alkali metal for what to include, although it is not done as best as it could be there) and chalcogens (just O at the moment, unfortunately). Double sharp (talk) 13:53, 30 March 2013 (UTC)
- Yeah, focus at the Characteristics section for while, it seems to be the most problematic (however, to be honest, I simply didn't make it any further yet), and alkali metal and noble gas are worth comparisons. I wanted to give some short constructive criticism, Atomic and physical lacks way too much info: density, color, hardness, mp, bp, etc. Isotopes lacks info about meta states, decay modes, etc. And it's not really important how many radioisotopes you have, the scientists may synthesize another one in a day, but nothing will change, most of them are not useful for anything. Chemical has too many formulas standing for words (way too many, write with words), and prose isn't genuine: "This oxidation number is found in sulfates, selenates, tellurates, and H2SO4 [hydrogen sulfate] or Na2SeO4. [sodium selenate] [and polonium?] [also, why not oxygen? tell the people]" etc, etc. It may be a torture, but it will hopefully pay you back.
- And you're describing what separate elements have, while this article is supposed to show similarities. So rather than saying "oxygen has ..., sulfur has ...," say "chalcogenes commonly have ..., with polonium (say) being an exception due to..." as a way to write better texts. Treat the whole article as such.
- And watch out for where you may need sources.--R8R Gtrs (talk) 14:18, 30 March 2013 (UTC)
- Ah, this is the problem I pointed out with regard to our current period articles, which are really bad. Why are they bad? They don't focus on what should be their topic, the period itself: they spend way too much time about the elements. If I want to know about a specific element in the group, I go to the article on that specific element. Readers go to the group article because they want to know about the group as a whole, as more than just the sum of its parts, its elements. So you should give that to them.
- (BTW, your organization doesn't quite work at times: for example, why are you mentioning the nonmetal-to-metal trend in the "Atomic and physical [characteristics]" section? It seems rather chemical to me!) Double sharp (talk) 16:06, 30 March 2013 (UTC)
- (Before going on: I sincerely apologize for not being very organized in my comments, but that's probably because I just skimmed through the article. And yet I found this many things to comment on. O_O)
- On Dmitri Mendeleev table: note how the elements are alternating to the left and right to each group across periods? That's the A group/B group divide. You should at least mention this; currently you are giving the impression that the A group/B group was a later development. It's not; it's just not marked explicitly here.
- You should also explain why chalcogenide does not usually include oxides. (Chemists usually don't group S with O mentally – their behaviour is just too different. The mental grouping is S/Se/Te/(Po if they can afford it). You need to explain something about their different behaviour. You do not make it strong enough. And yet "chalcogen" refers to the whole group 16.)
- I think you are focusing too much on the isotopes themselves. What do they imply? Chalcogens are all even-Z, doesn't this have an effect on their abundance? Why is O so abundant? (This level of talking about the individual elements is permissible, but try not to go overboard. Going overboard would be like discussing every element separately when there is no reason to. Hilarious example: March 2011 revision of alkali metal. And to think that I thought at that time that that would pass GA...O blessed naïvety!)
- Another point that is missing: why are Se and Te so rare in the crust? They're not so rare in the Solar System. You should explain why in the abundance section, even more so as this applies to the group somewhat (it's due to their volatile hydrides).
- Also: "Tellurium is not generally highly toxic. As little as 10 micrograms of tellurium per cubic meter of air can cause notoriously unpleasant breath, described as smelling like rotten garlic. Acute tellurium poisoning can cause vomiting, gut inflammation, internal bleeding, and respiratory failure. Extended, low-level exposure to tellurium causes tiredness and indigestion. Sodium tellurite (Na2TeO3) is lethal in amounts of around 2 grams." You mention so many severe effects of Te poisoning and start the paragraph with "Tellurium is not generally highly toxic"? To an uninformed reader, this will look weird, to say the least. I know what you're trying to get at, but all the same, you should try to fix this. Mention how much Te you need for acute Te poisoning, at least.
- On the very next paragraph: surely there are more isotopes of Po than just Po-210? I know that's the famous and most common one (Litvinenko) but there are more isotopes. Po-208 and Po-209 are even longer-lived, surely there must have been some studies done with them? Even if not, at least mention them somehow. Double sharp (talk) 16:24, 30 March 2013 (UTC)
- To add to your pain, the image of the CdTe array belongs to the U.S. Department of Energy contract-operator of the National Renewable Energy Laboratory and is therefore probably not a "work of the federal government" and is therefore probably not in the public domain. Unless you can establish that it is, it will get bounced during the FAC process. See here. Sandbh (talk) 22:13, 30 March 2013 (UTC)
- I commented out the file for now. King Jakob C2 22:26, 30 March 2013 (UTC)
- If it is from the DoE, isn't it covered under
{{PD-USGov-DOE}}? Double sharp (talk) 14:03, 2 April 2013 (UTC)- Only if it was taken by DoE employee as opposed to an employee of a DOE contractor-operator. Sandbh (talk) 09:27, 3 April 2013 (UTC)
- If it is from the DoE, isn't it covered under
- I commented out the file for now. King Jakob C2 22:26, 30 March 2013 (UTC)
- To add to your pain, the image of the CdTe array belongs to the U.S. Department of Energy contract-operator of the National Renewable Energy Laboratory and is therefore probably not a "work of the federal government" and is therefore probably not in the public domain. Unless you can establish that it is, it will get bounced during the FAC process. See here. Sandbh (talk) 22:13, 30 March 2013 (UTC)
- FA? Get ready to be whacked. ;-) I would extremely recommend everything R8R extremely recommends, and would also recommend looking at FAs of other groups (noble gas, although you may also consider looking at alkali metal for what to include, although it is not done as best as it could be there) and chalcogens (just O at the moment, unfortunately). Double sharp (talk) 13:53, 30 March 2013 (UTC)
Where the article stands
Done
- Remove the chemical formulas from wikilinks (not counting isotopes)
- Move the section on classification to chemical properties
- Add info on isomers and decay trends
- Address the comments R8R Gtrs left in the article (some of them)
- Get rid of the mention about all chalcogens having even atomic number-useless and obvious
- Find a citation for livermorium's configuration
- See if I can find anything on polonium-208 or polonium-209 toxicity
- Fix the inconsistencies on tellurium's toxicity
To do still
- Ease the primary focus to the actual element group. I have a bad feeling that this requires a substantial rewrite.
 Doing..., but really slowly.
Doing..., but really slowly. - Explain "chalcogenide" not including oxides
King Jakob C2 17:53, 2 April 2013 (UTC)
Yay, I fixed almost all the issues (but the biggest one still remains, I'll save it for last). Questions/comments/etc welcome. King Jakob C2 13:30, 10 April 2013 (UTC)
- You have fixed the inconsistency on Te's toxicity, but have not actually added the data. You mention acute Te poisoning, and only give a figure (which is not even stated explicitly to be an LD50 or toxic threshold, only implied very weakly) for annoying effects of Te. A reader will probably be more concerned with toxicity than annoying effects, so give them what they want.
- Lv's status as a chalcogen is not certain, and you should not talk about it assuming it was one of them without comment. Give a section of extensions talking about what the chalcogens beyond livermorium are predicted to be (by various people!) and state some stuff about their properties. The papers I posted links to (and Haire's book) in the "Metalloids" section above are very good sources.
- I would not call the fact that all chalcogens have even atomic number useless or even obvious. It does have rather deeper consequences – for example, their abundancy (and this is something you should write about, as I stated above. Talk about why O is so common and why Se and Te are so rare in the Earth's crust). Don't stop with what I tell you here. It is not really a good thing to dump the content of an entire article into a comment. Find out more. At the very least, read the articles on the individual chalcogens very closely and hunt for info. (Please do more than what group 4 element does. That is just the sort of article you will have to write when there is not much homology or info. There is a lot more on the chalcogens.)
- Chemists do not lump O with the rest of the chalcogens in their mental categories. This is why you will not usually see the term "chalcogenides" encompassing oxides in the absence of a special reason to do so. You should thus explain why.
- Here is a really useful ref. Mostly O and S, and doesn't take into acount FOOF (O2F2) in its discussion of oxygen halides, but very good as a source to mine. (I've been mining their alkali metal page for info for two years and it still hasn't completely run dry of info!)
- If you want my help with anything, ping me on my talkpage. Double sharp (talk) 11:31, 11 April 2013 (UTC)
- Also: start a separate section on chemistry. You can cover the main points in the Chemical characteristics subsection and save the details about chalcogen chemistry in this new section. Double sharp (talk) 14:52, 13 April 2013 (UTC)
- Presume you mean like this? King Jakob C2 15:08, 13 April 2013 (UTC)
- Yes, something like that. Double sharp (talk) 16:17, 13 April 2013 (UTC)
- Presume you mean like this? King Jakob C2 15:08, 13 April 2013 (UTC)
- I am currently working on Chalcogen#Chemical actually about chalcogens. It's getting there.King Jakob C2 18:31, 19 April 2013 (UTC)
- It's still kind of disorganized and doesn't follow compounds in a structured sequence. You could talk about their most basic compounds first like I do in alkali metal (I perhaps wrote too much on them, but you should consider giving each one paragraph at most – give it some coverage before you jump into the deep end of thioketones and chalcogenols that the average Wikipedian probably knows nothing about. Start with the simplest stuff.) Double sharp (talk) 11:55, 23 April 2013 (UTC)
About easing the focus to the primary group: you can keep most of the material you already have. But you should explicitly link it somehow back to the main topic, or we may go off course, or at least appear to, which is not good. If a reader wants to go off course, that's what hyperlinks are for: this is hypertext, after all. I talk about a lot of peripheral things in alkali metal in addition to the core info everyone is looking for. That's OK though, since I explicitly tie it back to the main topic and show how it is relevant. Double sharp (talk) 12:01, 23 April 2013 (UTC)
Group templates coloring: simplicity vs. complication
Look at our pnictogen group template just as an example.
We are stating in every cell that an element is a metalloid (antimony's case) or whatever else. And duplicate that with color, which, according to the legend, means essentially the same. The color info is only duplicating what we are saying without using it.
Do we need the color legend? Do we need colors at all?
See a draft of how it looks w/o colors.
Not perfect either. It has colors still in the small top table. We could remove them. Or keep them, we don't say what most of 'em mean anyway.
But there's a designing trick called multiplying by two, which basically states "make me more complicated, but so that anyone could understand me." Is it the case? (Just to confuse you further, there's also division by two, but something makes me think this is almost certainly not the case. may be wrong.)
Your thoughts, guys?--R8R Gtrs (talk) 19:14, 23 March 2013 (UTC)
- I don't mind. I think I prefer the coloured template because the colour in the cells matches the colour in the small top table. So the cells are a blown up version of the small top table. Hence would expect to see the colours in the blown up version too. Also the progression of colours is congruent with metallic character increasing going down the group. Sandbh (talk) 23:05, 23 March 2013 (UTC)
- (edit conflict)agree. R8R annouced this nicely on my talk page here. The proposal is: do no show background color for metalishness ("post-transition", "non-metal"). I support. We have a diff, please compare: {{Periodic table (pnictogens)}} versus {{ Periodic table (pnictogens)/sandbox}}. These same pictures look soooo much more brightly! -DePiep (talk) 23:09, 23 March 2013 (UTC)
- Happy to have the colours removed (the pictures do look better). Sandbh (talk) 23:25, 23 March 2013 (UTC)
- Fine with me either way. Double sharp (talk) 02:07, 24 March 2013 (UTC)
- Happy to have the colours removed (the pictures do look better). Sandbh (talk) 23:25, 23 March 2013 (UTC)
- (edit conflict)agree. R8R annouced this nicely on my talk page here. The proposal is: do no show background color for metalishness ("post-transition", "non-metal"). I support. We have a diff, please compare: {{Periodic table (pnictogens)}} versus {{ Periodic table (pnictogens)/sandbox}}. These same pictures look soooo much more brightly! -DePiep (talk) 23:09, 23 March 2013 (UTC)
Changed basic template {{Element cell image}} [5]. The color is {{element color}} "table mark", that is a light grey: #f0f0f0. One can still use |bg=#ab0823 to overrule this color (set bg color). Note: bg colors in {{Periodic table (with pictures)}} are gone too. If someone wants them back there, please say so.
- Not a simple edit any more: legend lines removed from each PT group infobox [6], expect 18 times. -DePiep (talk) 18:04, 24 March 2013 (UTC)
![]() Done -DePiep (talk) 17:43, 24 March 2013 (UTC)
Done -DePiep (talk) 17:43, 24 March 2013 (UTC)
Occurrence 'natural radio'?
Many of our periodic tables show the occurrence of an element (in the border pattern). The wording "natural radio" seems incorrect. My question is: are all the texts and links correct?
- From decay link: Trace radioisotope
- Also used: natural radio (to set this border type). There is no link to Natural radioactivity, which does Redirect to Background radiation.
- It looks like "natural radio" is a shortcut, and is not the right word. To set the border this may be OK, but the text also appears in the title (mousehoover) text. Should we abandon this, and use from decay only?
- Adding: let's link to Element from decay, that now redirects to Trace radioisotope and can evolve into whatever. Redirects are cheap (and useful, this way).
The two other options are less problematic:
- Primordial link: Primordial element, R to Primordial nuclide
- Should we link directly to page Primordial nuclide?
- Synthetic link to Synthetic element
-DePiep (talk) 14:12, 24 March 2013 (UTC) Added the Element from decay link as part of the proposal. -DePiep (talk) 17:01, 24 March 2013 (UTC)
- Here's what I think of when I hear "natural radio." "From decay" is better.--R8R Gtrs (talk) 15:27, 24 March 2013 (UTC)
- Thanks for making me laugh. I expected this. -DePiep (talk) 16:47, 24 March 2013 (UTC)
I have added new page Element from decay as a logical link, and for now it can redirect. The name fits the pattern a bit with the other two links. This is what redirects are good for. -DePiep (talk) 17:01, 24 March 2013 (UTC)
- See Alkaline earth metal: radioactive natural element links to Radioactive decay. All fine? -DePiep (talk) 23:12, 2 April 2013 (UTC)
No. I think "natural radio" probably refers to naturally occuring VLF radio waves from lightning strikes. What makes whistlers in HAM radio. [7]. Probably should redirect to lightning as WP has no article on the natural production of radio waves by terrestrial sources, other than the note in lightning that lightning makes a huge amount of ELF and VLF radio waves. SBHarris 01:10, 3 April 2013 (UTC)
- "Natural radio": we should also consider it meaning radio as it was meant to be, being an elementary part of the success of radio broadcast. We could link to here. -DePiep (talk) 09:32, 5 April 2013 (UTC)
Radon picture
There is a discussion here on our radon image which I participated in (and it got me to go around looking for a radon picture again. My findings are listed there.) Double sharp (talk) 12:49, 28 March 2013 (UTC)
'Ask the historian'
If you ever had the pleasure of reading one of Bill Jensen's 'Ask the historian' columns in The Journal of Chemical Education, e.g. 'Why Helium Ends in "-ium" '; 'Why Tungsten Instead of Wolfram?', he has published them all (and a few that didn't make it into print) in a free e-book. Sandbh (talk) 22:24, 28 March 2013 (UTC)
- Yay! (Why didn't I notice this sooner?) Double sharp (talk) 03:19, 29 March 2013 (UTC)
- LOL! Ditto! Sandbh (talk) 06:31, 29 March 2013 (UTC)
- Time to mine these juicy sources. ;-) Double sharp (talk) 02:38, 31 March 2013 (UTC)
- Please stop spoiling my fun. Already Double sharp told the Ghandi thing. Book has not arrived yet. -DePiep (talk) 23:57, 2 April 2013 (UTC)
- Time to mine these juicy sources. ;-) Double sharp (talk) 02:38, 31 March 2013 (UTC)
- LOL! Ditto! Sandbh (talk) 06:31, 29 March 2013 (UTC)
Nuclides at Wikidata
Did anybody discuss a transfer of nuclides data to Wikidata? This should be done sooner or later, and there are not so many nuclides to deny one Wikidata entry for each of them. Since Lua can read from Wikidata, it would put the end to clumsy tables in “isotopes of … ” articles. Incnis Mrsi (talk) 12:01, 6 April 2013 (UTC)
- I dunno. I would prefer having the tables here, actually. We need to give this sort of info about the isotopes here – it's rather important data for an isotope, isn't it? Also, there is way more content that could be put in the "Isotopes of X" articles. They should be more than just lists. The table should be there for reference, but there should also be content about the isotopes of X in general and some individual important isotopes of X. Double sharp (talk) 03:38, 7 April 2013 (UTC)
{{Cite doi}}
For our articles this template seems to be really nice. Nergaal (talk) 23:09, 9 April 2013 (UTC)
Etymology
I have added etymology=... and etymology link=... to the standard {{infobox element}}. See Gold for a first example.
On can set the link: etymology link=pageX (to produce: [[{{{etymology link}}}|Etymology]]. The etymology could be a verbose entry, not just a simple fact. -DePiep (talk) 02:00, 14 April 2013 (UTC) tried to clarify -DePiep (talk) 02:04, 14 April 2013 (UTC)
- Nice! Double sharp (talk) 02:56, 14 April 2013 (UTC)
- Sweet.--R8R Gtrs (talk) 09:20, 14 April 2013 (UTC)
- But not neat yet ;-) Infobox ununennium. I think we can find a better link & phrasing. -DePiep (talk) 09:45, 14 April 2013 (UTC)
Which pattern(s) to use?
- Historical, translated
- (h1): Krypton ← from [κρυπτός kryptos] Error: {{Lang-xx}}: text has italic markup (help) "the hidden one" (uses {{lang-el}}; currently in the lead)
- (h2): Krypton ← from [κρυπτός kryptos] Error: {{Lang-xx}}: text has italic markup (help) meaning "the hidden one" (uses {{lang-el}})
- (h3): Krypton ← from κρυπτός kryptos, Error: {{language with name/for}}: missing language tag or language name (help) (uses {{Language with name/for}})
- (h4): Krypton ← κρυπτός kryptos, Error: {{language with name/for}}: missing language tag or language name (help) (uses {{Language with name/for}})
- From places
- (p1): Berkelium ← after Berkely, California where it was discovered (Lawrence Berkeley National Laboratory) (no language template used)
- (p2): Berkelium ← after Berkely, California
- (p3): Berkelium ← after Berkely, California where it was discovered
- From names
- (n1): Einsteinium ← after Albert Einstein
- From systematic element name
- (s1): Ununennium ← IUPAC systematic element name
- (s2): Ununennium ← Systematic element name
- (s3): Ununennium ← "one-one-nine-ium" as per IUPAC Systematic element name
- Other
- (o1):
- Talks
(I fixed changed the numbering from "#" into stable "(h1)" etc.) -DePiep (talk) 22:04, 15 April 2013 (UTC) (added details, -DePiep (talk) 22:39, 15 April 2013 (UTC))
Both language templates show: only the non-English words are italics. -DePiep (talk) 10:43, 14 April 2013 (UTC)
- I like the wording of (h1), but the language template mucks up the formatting. Better IMO to use normal formatting:
- from Greek κρυπτός kryptos "the hidden one"
- No template covering the Greek text, not even fwow? And I miss the word meaning (or equivalent). Even in an infobox. -DePiep (talk) 22:30, 15 April 2013 (UTC)
- from Greek κρυπτός kryptos "the hidden one"
- I like (s1) better than (s2), as it's more explicit. For (p1), I would drop the parenthetical, since that info's in the article anyway, and it's a bit long with in the box. — kwami (talk) 05:18, 15 April 2013 (UTC)
Infobox data
Is there currently a centralised way of monitoring changes to element inforbox data? I generated a feed, which may be useful.--Gilderien Chat|List of good deeds 00:33, 20 April 2013 (UTC)
- There is template {{WikiProject Elements page lists}}, that is on various WP:ELEM pages. Clicking the right RELC shows edits to all PT templates (not just infoboxes), plus their talkpages. It uses [8]. -DePiep (talk) 11:17, 20 April 2013 (UTC)
Standard PT legends
I am working on standard legends for all of the periodic tables we have. At the moment, they have different appearences. I aim to give them the same look-and-feel. In the near future, on a PT page we can choose from a list of legend themes (category, occurrence, valence, ...). Also, the presence of fields like "Unknown" is optional. One example is in {{Periodic table/sandbox}} (compare {{Periodic table}}). The all-up list is at User:DePiep/sandbox1, with various variants demo in User:DePiep/sandbox1. Any remarks? -DePiep (talk) 11:42, 20 April 2013 (UTC)
Reignition
What happened in the intervening months between when I last came here (as Freywa) and now? First of all, I became a brony. I took a new pseudonym, Parcly Taxel, which is a play on the mitotic inhibitor paclitaxel. Once I slipped into the fandom, I created accounts on a string of websites, first deviantART, then Twitter and Reddit and Tumblr, all using the new pseudonym. Eventually I went full circle, and changed my username here. I have come back – as an alicorn.
I was the one who applied for our Signpost interview way back in June 2011. Since I left the project has fallen into relative inactivity, although the periodic table has been restored to its former glory after 7¾ years. Where are we now, and what articles need work? I suggest that we work to promote one of our five A-class articles to FA, and I think the most appropriate one of them is berkelium. There are some uses for it, though not a lot, so patching the gaps should be rather easy. The time has come for reignition. Princess Parcly Taxel 02:27, 21 April 2013 (UTC)
- Our "relative inactivity" is not helped by my tendency to experience a severe case of wikiwanderlust when things slow down a little :-). I will finish Po shortly for GA. I left Bk a while ago but can return to it (see R8R Gtrs' comments, untouched for some time). I am waiting for R8R to finish commenting on Hs before I start on it in earnest.
- On our A-class articles, R8R will not lay down his typing fingers indefinitely (though he may take long breaks from it) until he gets an FA. That much is clear. He is also working on At and 117 (the latter with me). TCO seems to have disappeared from us for now, but come back he will, I would think. Would that they were here, for we seem to be in need of them! Sandbh is working on the metalloids. Bk was suggested by R8R and I accepted (though have taken a long break from it). Therefore it looks like the A-class articles are all taken (and no, I would think it impolite to steal an article someone is working on away from them before asking them.)
- Despite chalcogen being a GA, it is in need of serious work, I deem. I would advise King jakob c to really read and mine his sources not in isolation from each other, but together. That way he can understand the subject he is writing on better and can write the whole article much better. (To him: Sincere apologies for talking of you while you are not here, but in this context it is rather unavoidable.) I learnt this myself in alkali metal some years ago. You can check out B+'s arsenic and boron group. The latter is actually not bad, but for its writing style (and its omission of a major section on periodic trends). Double sharp (talk) 13:00, 21 April 2013 (UTC)
- Also: we need to resolve that huge "metalloids" discussion above. It's already lasted over half a year here and people are still discussing. That is not a bad thing; let us not rush into a choice. Yet, we really should tie up some of the loose ends. Double sharp (talk) 13:01, 21 April 2013 (UTC)
- BTW, please don't use the Unicode fractions. The more commonly used ones are already hard enough to see and distinguish and as for the rest, let's not go there, shall we? Double sharp (talk) 15:00, 21 April 2013 (UTC)
- Indeed. MOS:FRAC says we should use {{frac}}, so that is for all of WP. -DePiep (talk) 18:15, 21 April 2013 (UTC)
- BTW, please don't use the Unicode fractions. The more commonly used ones are already hard enough to see and distinguish and as for the rest, let's not go there, shall we? Double sharp (talk) 15:00, 21 April 2013 (UTC)
Well, Parcly Taxel, Freywa, this is what happened while you were away. First of all, Double sharp and R8R and Sandbh and ST11 and me and others took care of The Elements. None escaped! But we were very concerned about you. For example me, I was longing for your edits -- cannot imagine how I survived without. Already your re-introduction here is a WP joy (self-serving, but alas). The very fact that you did not see a diff between your departure and now, already says you are indispensable. Glad you are back. Now do a contributive edit or shut up. -DePiep (talk) 23:24, 22 April 2013 (UTC)
- Well, this about summarizes the difference between the articles now and two years ago. Nergaal (talk) 13:16, 23 April 2013 (UTC)
References: new error messages
The {{cite book}} and {{cite web}} templates have been changed into Lua script. Many error messages are added. They show up below, in the reference section (like here, before I corrected). All our articles might need a check & cleanup. -DePiep (talk) 14:24, 21 April 2013 (UTC)
- Please change this overview table: set a page
aq=B(to green) when you have checked & cleared any ref errors. - Note: errors may be suppressed, depending on your skin setting. See Help:CS1_errors#Controlling_error_message_display. -DePiep (talk) 15:08, 21 April 2013 (UTC)
- blue: see below
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| infobox element (128x) |
(Note: Groups 6 through 9 have 0 citations, not even a single one, group 10 has 1 (a bare url), group 11 has two. --R8R Gtrs (talk) 15:48, 21 April 2013 (UTC))
(Note: the script barks at the non-systematic unused_data parameter. Articles with it remain orange. All elements have been checked.--R8R Gtrs (talk) 16:33, 21 April 2013 (UTC))
- Do you mean: all those elements still orange have error(s)? -DePiep (talk) 17:00, 21 April 2013 (UTC)
- Yes.--R8R Gtrs (talk) 17:17, 21 April 2013 (UTC)
- Do you mean: all those elements still orange have error(s)? -DePiep (talk) 17:00, 21 April 2013 (UTC)
- Note: an error can also be in the infobox. -DePiep (talk) 16:38, 21 April 2013 (UTC)
Todo: Rubidium has an incorrect doi, with no hints left. -DePiep (talk) 23:18, 21 April 2013 (UTC)
- I suggest deleting the ref (can't be completed as there are no correct info about it, there's another ref at the very same place in the article). That's a lazy way, but it seems to be appropriate in this case.--R8R Gtrs (talk) 16:30, 23 April 2013 (UTC)
I must say, I liked it better in the past when the code was more forgiving. Double sharp (talk) 10:12, 22 April 2013 (UTC)
- These errors mostly are ambiguous info, if not plain wrong. Esp. for outside connections (doi, authors, COinS, ISBN, ...) these corrections are an improvement. And solving them helps me understand the cite-templates & how to cite correctly. -DePiep (talk) 12:41, 22 April 2013 (UTC)
Much of the sixth period has been fixed. For lead one of the errors was in a {{cite doi}} template. Princess Parcly Taxel 11:17, 24 April 2013 (UTC)