Vitronectin
Vitronectin (VTN or VN) is a glycoprotein of the hemopexin family which is synthesized and excreted by the liver, and abundantly found in serum, the extracellular matrix and bone.[5] In humans it is encoded by the VTN gene.[6][7]
Vitronectin binds to integrin alpha-V beta-3 and thus promotes cell adhesion and spreading. It also inhibits the membrane-damaging effect of the terminal cytolytic complement pathway and binds to several serpins (serine protease inhibitors). It is a secreted protein and exists in either a single chain form or a clipped, two chain form held together by a disulfide bond.[6] Vitronectin has been speculated to be involved in hemostasis[8] and tumor malignancy.[9][10]
Structure
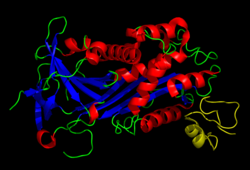
[edit]Vitronectin is a 54 kDa glycoprotein, consisting of 478 amino acid residues. About one-third of the protein's molecular mass is composed of carbohydrates. On occasion, the protein is cleaved after arginine 379, to produce two-chain vitronectin, where the two parts are linked by a disulfide bond. No high-resolution structure has been determined experimentally yet, except for the N-terminal domain.
The protein consists of three domains:
- The N-terminal Somatomedin B domain (1-39)
- A central domains with hemopexin homology (131-342)
- A C-terminal domain (residues 347-459) also with hemopexin homology.
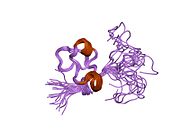
Several structures has been reported for the Somatomedin B domain. The protein was initially crystallized in complex with one of its physiological binding partners: the Plasminogen activator inhibitor-1 (PAI-1) and the structure solved for this complex.[11] Subsequently two groups reported NMR structures of the domain.[12][13]
The somatomedin B domain is a close-knit disulfide knot, with 4 disulfide bonds within 35 residues. Different disulfide configurations had been reported for this domain[14][15][16] but this ambiguity has been resolved by the crystal structure.[16]
Homology models have been built for the central and C-terminal domains.[16]
Function
[edit]The somatomedin B domain of vitronectin binds to plasminogen activator inhibitor-1 (PAI-1), and stabilizes it.[11] Thus vitronectin serves to regulate proteolysis initiated by plasminogen activation. In addition, vitronectin is a component of platelets and is, thus, involved in hemostasis. Vitronectin contains an RGD (45-47) sequence, which is a binding site for membrane-bound integrins, e.g., the vitronectin receptor, which serve to anchor cells to the extracellular matrix. The Somatomedin B domain interacts with the urokinase receptor, and this interaction has been implicated in cell migration and signal transduction. High plasma levels of both PAI-1 and the urokinase receptor have been shown to correlate with a negative prognosis for cancer patients. Cell adhesion and migration are directly involved in cancer metastasis, which provides a probable mechanistic explanation for this observation.
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000109072 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000017344 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Boron, Walter F. and Boulpaep, Emile L. "Medical Physiology". Saunders, 2012, p.1097.
- ^ a b "Entrez Gene: M Vitronectin".
- ^ Jenne D, Stanley KK (Oct 1987). "Nucleotide sequence and organization of the human S-protein gene: repeating peptide motifs in the "pexin" family and a model for their evolution". Biochemistry. 26 (21): 6735–42. doi:10.1021/bi00395a024. PMID 2447940.
- ^ Preissner KT, Seiffert D (Jan 1998). "Role of vitronectin and its receptors in haemostasis and vascular remodeling". Thrombosis Research. 89 (1): 1–21. doi:10.1016/S0049-3848(97)00298-3. PMID 9610756.
- ^ Felding-Habermann B, Cheresh DA (Oct 1993). "Vitronectin and its receptors". Current Opinion in Cell Biology. 5 (5): 864–8. doi:10.1016/0955-0674(93)90036-P. PMID 7694604.
- ^ Hurt, Elaine M.; Chan, King; Serrat, Maria Ana Duhagon; Thomas, Suneetha B.; Veenstra, Timothy D.; Farrar, William L. (2009). "Identification of Vitronectin as an Extrinsic Inducer of Cancer Stem Cell Differentiation and Tumor Formation". Stem Cells. 28 (3): 390–8. doi:10.1002/stem.271. PMC 3448441. PMID 19998373.
- ^ a b Zhou A, Huntington JA, Pannu NS, Carrell RW, Read RJ (Jul 2003). "How vitronectin binds PAI-1 to modulate fibrinolysis and cell migration". Nature Structural Biology. 10 (7): 541–4. doi:10.1038/nsb943. PMID 12808446. S2CID 26086796.
- ^ Kamikubo Y, De Guzman R, Kroon G, Curriden S, Neels JG, Churchill MJ, Dawson P, Ołdziej S, Jagielska A, Scheraga HA, Loskutoff DJ, Dyson HJ (Jun 2004). "Disulfide bonding arrangements in active forms of the somatomedin B domain of human vitronectin". Biochemistry. 43 (21): 6519–34. doi:10.1021/bi049647c. PMID 15157085.
- ^ Mayasundari A, Whittemore NA, Serpersu EH, Peterson CB (Jul 2004). "The solution structure of the N-terminal domain of human vitronectin: proximal sites that regulate fibrinolysis and cell migration". The Journal of Biological Chemistry. 279 (28): 29359–66. doi:10.1074/jbc.M401279200. PMID 15123712.
- ^ Kamikubo Y, Okumura Y, Loskutoff DJ (Jul 2002). "Identification of the disulfide bonds in the recombinant somatomedin B domain of human vitronectin". The Journal of Biological Chemistry. 277 (30): 27109–19. doi:10.1074/jbc.M200354200. PMID 12019263.
- ^ Horn NA, Hurst GB, Mayasundari A, Whittemore NA, Serpersu EH, Peterson CB (Aug 2004). "Assignment of the four disulfides in the N-terminal somatomedin B domain of native vitronectin isolated from human plasma". The Journal of Biological Chemistry. 279 (34): 35867–78. doi:10.1074/jbc.M405716200. PMID 15173163.
- ^ a b c Xu D, Baburaj K, Peterson CB, Xu Y (Aug 2001). "Model for the three-dimensional structure of vitronectin: predictions for the multi-domain protein from threading and docking". Proteins. 44 (3): 312–20. doi:10.1002/prot.1096. PMID 11455604. S2CID 24765480.
Further reading
[edit]- Singh B, Su YC, Riesbeck K (2010). "Vitronectin in bacterial pathogenesis: a host protein used in complement escape and cellular invasion". Mol. Microbiol. 78 (3): 545–60. doi:10.1111/j.1365-2958.2010.07373.x. PMID 20807208. S2CID 24516528.
- Singh B, Jalalvand F, Mörgelin M, Zipfel P, Blom AM, Riesbeck K (2011). "Haemophilus influenzae protein E recognizes the C-terminal domain of vitronectin and modulates the membrane attack complex". Mol. Microbiol. 81 (1): 80–98. doi:10.1111/j.1365-2958.2011.07678.x. PMID 21542857. S2CID 26484037.
- Su YC, Jalalvand F, Mörgelin M, Blom AM, Singh B, Riesbeck K (2013). "Haemophilus influenzae acquires vitronectin via the ubiquitous Protein F to subvert host innate immunity". Mol. Microbiol. 87 (6): 1245–66. doi:10.1111/mmi.12164. PMID 23387957. S2CID 26923730.
External links
[edit]- Vitronectin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)