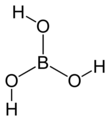
Boric acid
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Boric acid[1]
| |||
| Other names
Orthoboric acid, Boracic acid, Sassolite, Borofax, Trihydroxyborane, Boranetriol, Hydrogen borate, Acidum boricum
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.030.114 | ||
| EC Number |
| ||
| E number | E284 (preservatives) | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| BH3O3 | |||
| Molar mass | 61.83 g·mol−1 | ||
| Appearance | White crystalline solid | ||
| Density | 1.435 g/cm3 | ||
| Melting point | 170.9 °C (339.6 °F; 444.0 K) | ||
| Boiling point | 300 °C (572 °F; 573 K) | ||
| 2.52 g/100 mL (0 °C) 4.72 g/100 mL (20 °C) 5.7 g/100 mL (25 °C) 19.10 g/100 mL (80 °C) 27.53 g/100 mL (100 °C) | |||
| Solubility in other solvents | Soluble in lower alcohols moderately soluble in pyridine very slightly soluble in acetone | ||
| log P | -0.29[2] | ||
| Acidity (pKa) | 9.24 (first proton), 12.4 (second), 13.3 (complete) | ||
| Conjugate base | Borate | ||
| -34.1·10−6 cm3/mol | |||
| Structure | |||
| Trigonal planar | |||
| 0 D | |||
| Pharmacology | |||
| S02AA03 (WHO) D08AD (WHO) | |||
| Hazards | |||
| GHS labelling: | |||

| |||
| NFPA 704 (fire diamond) | |||
| Flash point | Nonflammable | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
2660 mg/kg, oral (rat) | ||
| Related compounds | |||
Related compounds
|
Boron trioxide Borax | ||
| Supplementary data page | |||
| Boric acid (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula B(OH)3. It may also be called hydrogen orthoborate, trihydroxidoboron or boracic acid.[3] It is usually encountered as colorless crystals or a white powder, that dissolves in water, and occurs in nature as the mineral sassolite. It is a weak acid that yields various borate anions and salts, and can react with alcohols to form borate esters.
Boric acid is often used as an antiseptic, insecticide, flame retardant, neutron absorber, or precursor to other boron compounds.
The term "boric acid" is also used generically for any oxoacid of boron, such as metaboric acid HBO2 and tetraboric acid H2B4O7.
History[edit]
Orthoboric acid was first prepared by Wilhelm Homberg (1652–1715) from borax, by the action of mineral acids, and was given the name sal sedativum Hombergi ("sedative salt of Homberg"). However boric acid and borates have been used since the time of the ancient Greeks for cleaning, preserving food, and other activities.[4]
Molecular and crystal structure[edit]
The three oxygen atoms form a trigonal planar geometry around the boron. The B-O bond length is 136 pm and the O-H is 97 pm. The molecular point group is C3h.[5]
Two crystalline forms of orthoboric acid are known: triclinic and hexagonal. The former is the most common; the second, which is a bit more stable thermodynamically, can be obtained with a special preparation method.[6]
Triclinic[edit]
The triclinic form of boric acid consists of layers of B(OH)3 molecules held together by hydrogen bonds with an O...O separation of 272 pm. The distance between two adjacent layers is 318 pm.[7]
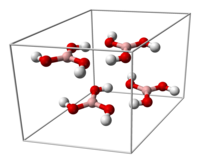 |
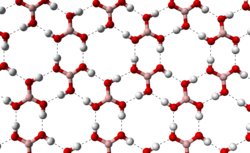 |
The unit cell of boric acid |
hydrogen bonding (dashed lines)
allows boric acid molecules to form parallel layers in the solid state |
Preparation[edit]
Boric acid may be prepared by reacting borax (sodium tetraborate decahydrate) with a mineral acid, such as hydrochloric acid:
- Na2B4O7·10H2O + 2 HCl → 4 B(OH)3 + 2 NaCl + 5 H2O
It is also formed as a by product of hydrolysis of boron trihalides and diborane:[8]
- B2H6 + 6 H2O → 2 B(OH)3 + 6 H2
- BX3 + 3 H2O → B(OH)3 + 3 HX (X = Cl, Br, I)
Reactions[edit]
Pyrolysis[edit]
When heated, orthoboric acid undergoes a three step dehydration. The reported transition temperatures vary substantially from source to source.[citation needed]
When heated above 140 °C, orthoboric acid yields metaboric acid (HBO2) with loss of one water molecule:[9][10]
- B(OH)3 → HBO2 + H2O
Heating metaboric acid above about 180 °C eliminates another water molecule forming tetraboric acid, also called pyroboric acid (H2B4O7):[9][10]
- 4 HBO2 → H2B4O7 + H2O
Further heating (to about 530 °C) leads to boron trioxide:[11][9][10]
- H2B4O7 → 2 B2O3 + H2O
Aqueous solution[edit]
When orthoboric acid is dissolved in water, it partially dissociates to give metaboric acid:
- B(OH)3 ⇌ HBO2 + H2O
The solution is mildly acidic due to ionization of the acids:
- B(OH)3 + H2O ⇌ [BO(OH)2]− + H3O+
- HBO2 + H2O ⇌ [BO2]− + H3O+
However, Raman spectroscopy of strongly alkaline solutions has shown the presence of [B(OH)4]− ions,[12] leading some to conclude that the acidity is exclusively due to the abstraction of OH− from water:[12]
- B(OH)3 + HO− ⇌ B(OH)−4
Equivalently,
- B(OH)3 + H2O ⇌ B(OH)−4 + H+ (K = 7.3×10−10; pK = 9.14)
Or, more properly,
- B(OH)3 + 2 H2O ⇌ B(OH)−4 + H3O+
This reaction occurs in two steps, with the neutral complex aquatrihydroxyboron B(OH)3(OH2) as an intermediate:[13]
- B(OH)3 + H2O → B(OH)3(OH2)
- B(OH)3(OH2) + H2O + HO− → [B(OH)4]− + H3O+
This reaction may be characterized as Lewis acidity of boron toward [HO]−, rather than as Brønsted acidity.[14][15][16] However, some of its behaviour towards some chemical reactions suggest it to be tribasic acid in the Brønsted sense as well.
Boric acid, mixed with borax Na2B4O7·10H2O (more properly Na2B4O5(OH)4·8H2O) in the weight ratio of 4:5, is highly soluble in water, though they are not so soluble separately.[17]
Sulfuric acid solution[edit]
Boric acid also dissolves in anhydrous sulfuric acid according to the equation:[7]
- B(OH)3 + 6 H2SO4 → [B(SO4H)4]− + 2 [HSO4]− + 3 H3O+
The product is an extremely strong acid, even stronger than the original oleum.[7]
Esterification[edit]
Boric acid reacts with alcohols to form borate esters, B(OR)3 where R is alkyl or aryl. The reaction is typically driven by a dehydrating agent, such as concentrated sulfuric acid:[18]
- B(OH)3 + 3 ROH → B(OR)3 + 3 H2O
With vicinal diols[edit]
The acidity of boric acid solutions is greatly increased in the presence of cis-vicinal diols (organic compounds containing similarly oriented hydroxyl groups in adjacent carbon atoms, (R1,R2)=C(OH)−C(OH)=(R3,R4)) such as glycerol and mannitol.[19][7][20]
The tetrahydroxyborate anion formed in the dissolution spontaneously reacts with these diols to form relatively stable anion esters containing one or two five-member −B−O−C−C−O− rings. For example, the reaction with mannitol H(HCOH)6H, whose two middle hydroxyls are in cis orientation, can be written as
- B(OH)3 + H2O ⇌ [B(OH)4]− + H+
- [B(OH)4]− + H(HCOH)6H ⇌ [B(OH)2(H(HCOH)2(HCO−)2(HCOH)2H)]− + 2 H2O
- [B(OH)2(H(HCOH)2(HCO−)2(HCOH)2H)]− + H(HCOH)6H ⇌ [B(H(HCOH)2(HCO−)2(HCOH)2H)2]− + 2 H2O
Giving the overall reaction
- B(OH)3 + 2 H(HCOH)6H ⇌ [B(H(HCOH)2(HCO−)2(HCOH)2H)2]− + 3 H2O + H+
The stability of these mannitoborate ester anions shifts the equilibrium of the right and thus increases the acidity of the solution by 5 orders of magnitude compared to that of pure boric oxide, lowering the pKa from 9 to below 4 for sufficient concentration of mannitol.[19][7][20] The resulting solution has been called mannitoboric acid.
The addition of mannitol to an initially neutral solution containing boric acid or simple borates lowers its pH enough for it to be titrated by a strong base as NaOH, including with an automated a potentiometric titrator. This property is used in analytical chemistry to determine the borate content of aqueous solutions, for example to monitor the depletion of boric acid by neutrons in the water of the primary circuit of light-water reactor when the compound is added as a neutron poison during refueling operations.[7]
Toxicology[edit]
Based on mammalian median lethal dose (LD50) rating of 2,660 mg/kg body mass, boric acid is only poisonous if taken internally or inhaled in large quantities. The Fourteenth Edition of the Merck Index indicates that the LD50 of boric acid is 5.14 g/kg for oral dosages given to rats, and that 5 to 20 g/kg has produced death in adult humans. For a 70 kg adult, at the lower 5 g/kg limit, 350 g could produce death in humans. For comparison's sake, the LD50 of salt is reported to be 3.75 g/kg in rats according to the Merck Index. According to the Agency for Toxic Substances and Disease Registry, "The minimal lethal dose of ingested boron (as boric acid) was reported to be 2–3 g in infants, 5–6 g in children, and 15–20 g in adults. [...] However, a review of 784 human poisonings with boric acid (10–88 g) reported no fatalities, with 88% of cases being asymptomatic."[21]
Long-term exposure to boric acid may be of more concern, causing kidney damage and eventually kidney failure (see links below). Although it does not appear to be carcinogenic, studies in dogs have reported testicular atrophy after exposure to 32 mg/(kg⋅day) for 90 days. This level, were it applicable to humans at like dose, would equate to a cumulative dose of 202 g over 90 days for a 70 kg adult, not far lower than the above LD50.[22]
According to the CLH report for boric acid published by the Bureau for Chemical Substances Lodz, Poland, boric acid in high doses shows significant developmental toxicity and teratogenicity in rabbit, rat, and mouse fetuses, as well as cardiovascular defects, skeletal variations, and mild kidney lesions.[23] As a consequence in the 30th ATP to EU directive 67/548/EEC of August 2008, the European Commission decided to amend its classification as reprotoxic category 2 and to apply the risk phrases R60 (may impair fertility) and R61 (may cause harm to the unborn child).[24][25][26][27][28]
At a 2010 European Diagnostics Manufacturing Association (EDMA) Meeting, several new additions to the substance of very high concern (SVHC) candidate list in relation to the Registration, Evaluation, Authorisation and Restriction of Chemicals Regulations 2007 (REACH) were discussed. Following the registration and review completed as part of REACH, the classification of boric acid CAS 10043-35-3 / 11113-50-1 is listed from 1 December 2010 is H360FD (May damage fertility. May damage the unborn child).[29][30]
Uses[edit]
Industrial[edit]
The primary industrial use of boric acid is in the manufacture of monofilament fiberglass usually referred to as textile fiberglass. Textile fiberglass is used to reinforce plastics in applications that range from boats, to industrial piping to computer circuit boards.[31]
Boric Acid is used as a Nuclear Poison in modern PWR type Nuclear Reactors as it Reduce Fission Process by Reducing Neutrons Flux. It is used in PWR Nuclear Reactor's Coolant water for Controlling Reactor Power as well as to Perform Emergency Reactor Shutdown.
In the jewelry industry, boric acid is often used in combination with denatured alcohol to reduce surface oxidation and thus formation of firescale on metals during annealing and soldering operations.[citation needed]
Boric acid is used in the production of the glass in LCD flat panel displays.[citation needed]
In electroplating, boric acid is used as part of some proprietary formulas. One such known formula calls for about a 1 to 10 ratio of H
3BO
3 to NiSO
4, a very small portion of sodium lauryl sulfate and a small portion of H
2SO
4.
The solution of orthoboric acid and borax in 4:5 ratio is used as a fire retarding agent of wood by impregnation.[32]
It is also used in the manufacturing of ramming mass, a fine silica-containing powder used for producing induction furnace linings and ceramics.
Boric acid is added to borax for use as welding flux by blacksmiths.[33]
Boric acid, in combination with polyvinyl alcohol (PVA) or silicone oil, is used to manufacture Silly Putty.[34]
Boric acid is also present in the list of chemical additives used for hydraulic fracturing (fracking) in the Marcellus Shale in Pennsylvania.[35] It is often used in conjunction with guar gum as cross-linking and gelling agent for controlling the viscosity and the rheology of the fracking fluid injected at high pressure in the well. It is important to control the fluid viscosity for keeping in suspension on long transport distances the grains of the propping agents aimed at maintaining the cracks in the shales sufficiently open to facilitate the gas extraction after the hydraulic pressure is relieved.[36][37][38] The rheological properties of borate cross-linked guar gum hydrogel mainly depend on the pH value.[39]
Boric acid is used in some expulsion-type electrical fuses as a de-ionization/extinguishing agent.[40] During an electrical fault in an expulsion-type fuse, a plasma arc is generated by the disintegration and rapid spring-loaded separation of the fusible element, which is typically a specialized metal rod that passes through a compressed mass of boric acid within the fuse assembly. The high-temperature plasma causes the boric acid to rapidly decompose into water vapor and boric anhydride, and in-turn, the vaporization products de-ionize the plasma, helping to interrupt the electrical fault.[41]
Medical[edit]
Boric acid can be used as an antiseptic for minor burns or cuts and is sometimes used in salves and dressings, such as boracic lint. Boric acid is applied in a very dilute solution as an eye wash. Boric acid vaginal suppositories can be used for recurrent candidiasis due to non-albicans candida as a second line treatment when conventional treatment has failed.[42][43] It is less effective than conventional treatment overall.[42] Boric acid largely spares lactobacilli within the vagina.[44] As TOL-463, it is under development as an intravaginal medication for the treatment for vulvovaginal candidiasis.[45][46][47]
As an antibacterial compound, boric acid can also be used as an acne treatment. It is also used as prevention of athlete's foot, by inserting powder in the socks or stockings. Various preparations can be used to treat some kinds of otitis externa (ear infection) in both humans and animals.[48] The preservative in urine sample bottles in the UK is boric acid.[49]
Boric acid solutions used as an eye wash or on abraded skin are known to be toxic, particularly to infants, especially after repeated use; this is because of its slow elimination rate.[50]
Boric acid is one of the most commonly used substances that can counteract the harmful effects of reactive hydrofluoric acid (HF) after an accidental contact with the skin. It works by forcing the free F− anions into the inert tetrafluoroborate anion. This process defeats the extreme toxicity of hydrofluoric acid, particularly its ability to sequester ionic calcium from blood serum which can lead to cardiac arrest and bone decomposition; such an event can occur from just minor skin contact with HF.[51][failed verification]
Insecticidal[edit]
Boric acid was first registered in the US as an insecticide in 1948 for control of cockroaches, termites, fire ants, fleas, silverfish, and many other insects. The product is generally considered to be safe to use in household kitchens to control cockroaches and ants. It acts as a stomach poison affecting the insects' metabolism, and the dry powder is abrasive to the insects' exoskeletons.[52][53][54] Boric acid also has the reputation as "the gift that keeps on killing" in that cockroaches that cross over lightly dusted areas do not die immediately, but that the effect is like shards of glass cutting them apart. This often allows a roach to go back to the nest where it soon dies. Cockroaches, being cannibalistic, eat others killed by contact or consumption of boric acid, consuming the powder trapped in the dead roach and killing them, too.[citation needed]
Preservation[edit]
In combination with its use as an insecticide, boric acid also prevents and destroys existing wet and dry rot in timbers. It can be used in combination with an ethylene glycol carrier to treat external wood against fungal and insect attack. It is possible to buy borate-impregnated rods for insertion into wood via drill holes where dampness and moisture is known to collect and sit. It is available in a gel form and injectable paste form for treating rot affected wood without the need to replace the timber. Concentrates of borate-based treatments can be used to prevent slime, mycelium, and algae growth, even in marine environments.[citation needed]
Boric acid is added to salt in the curing of cattle hides, calfskins, and sheepskins. This helps to control bacterial development, and helps to control insects.[citation needed]
pH buffer[edit]
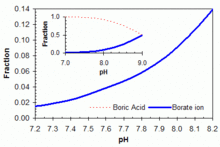
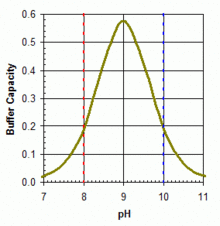
Boric acid in equilibrium with its conjugate base the borate ion is widely used (in the concentration range 50–100 ppm boron equivalents) as a primary or adjunct pH buffer system in swimming pools. Boric acid is a weak acid, with pKa (the pH at which buffering is strongest because the free acid and borate ion are in equal concentrations) of 9.24 in pure water at 25 °C. But apparent pKa is substantially lower in swimming pool or ocean waters because of interactions with various other molecules in solution. It will be around 9.0 in a salt-water pool. No matter which form of soluble boron is added, within the acceptable range of pH and boron concentration for swimming pools, boric acid is the predominant form in aqueous solution, as shown in the accompanying figure. The boric acid – borate system can be useful as a primary buffer system (substituting for the bicarbonate system with pKa1 = 6.0 and pKa2 = 9.4 under typical salt-water pool conditions) in pools with salt-water chlorine generators that tend to show upward drift in pH from a working range of pH 7.5–8.2. Buffer capacity is greater against rising pH (towards the pKa around 9.0), as illustrated in the accompanying graph. The use of boric acid in this concentration range does not allow any reduction in free HOCl concentration needed for pool sanitation, but it may add marginally to the photo-protective effects of cyanuric acid and confer other benefits through anti-corrosive activity or perceived water softness, depending on overall pool solute composition.[55]
Lubrication[edit]
Colloidal suspensions of nanoparticles of boric acid dissolved in petroleum or vegetable oil can form a remarkable lubricant on ceramic or metal surfaces[56] with a coefficient of sliding friction that decreases with increasing pressure to a value ranging from 0.10 to 0.02. Self-lubricating B(OH)3 films result from a spontaneous chemical reaction between water molecules and B2O3 coatings in a humid environment. In bulk-scale, an inverse relationship exists between friction coefficient and Hertzian contact pressure induced by applied load.[citation needed]
Boric acid is used to lubricate carrom and novuss boards, allowing for faster play.[57]
Nuclear power[edit]
Boric acid is used in some nuclear power plants as a neutron poison. The boron in boric acid reduces the probability of thermal fission by absorbing some thermal neutrons. Fission chain reactions are generally driven by the probability that free neutrons will result in fission and is determined by the material and geometric properties of the reactor. Natural boron consists of approximately 20% boron-10 and 80% boron-11 isotopes. Boron-10 has a high cross-section for absorption of low energy (thermal) neutrons. By increasing boric acid concentration in the reactor coolant, the probability that a neutron will cause fission is reduced. Changes in boric acid concentration can effectively regulate the rate of fission taking place in the reactor. During normal at power operation, boric acid is used only in pressurized water reactors (PWRs), whereas boiling water reactors (BWRs) employ control rod pattern and coolant flow for power control, although BWRs can use an aqueous solution of boric acid and borax or sodium pentaborate for an emergency shutdown system if the control rods fail to insert. Boric acid may be dissolved in spent fuel pools used to store spent fuel elements. The concentration is high enough to keep neutron multiplication at a minimum. Boric acid was dumped over Reactor 4 of the Chernobyl nuclear power plant after its meltdown to prevent another reaction from occurring.[citation needed]
Pyrotechnics[edit]
Boron is used in pyrotechnics to prevent the amide-forming reaction between aluminium and nitrates. A small amount of boric acid is added to the composition to neutralize alkaline amides that can react with the aluminium.
Boric acid can be used as a colorant to make fire green. For example, when dissolved in methanol it is popularly used by fire jugglers and fire spinners to create a deep green flame much stronger than copper sulfate.[58]
Agriculture[edit]
Boric acid is used to treat or prevent boron deficiencies in plants. It is also used in preservation of grains such as rice and wheat.[59]
References[edit]
- ^ "Boric acid".
- ^ "boric_msds".
- ^ Entry "boracic acid" in the online Merriamm-Webster Dictionary. Gives the first use as 1790. Retrieved 2022-06-24.
- ^ Ronald Eisler (2007). Eisler's Encyclopedia of Environmentally Hazardous Priority Chemicals. Elsevier. p. 59. ISBN 978-0-08-054707-7.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 1291. ISBN 978-0-08-037941-8.
- ^ Andrei Rotaru (2017): "Thermal and kinetic study of hexagonal boric acid versus triclinic boric acid in air flow." Journal of Thermal Analysis and Calorimetry, volume 127, pages 755–763. doi:10.1007/s10973-016-5583-7
- ^ a b c d e f Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Housecroft, C. E.; Sharpe, A. G. (2008). "Chapter 13: The Group 13 Elements". Inorganic Chemistry (3rd ed.). Pearson. p. 340. ISBN 978-0-13-175553-6.
- ^ a b c Gurwinder Kaur, Shagun Kainth, Rohit Kumar, Piyush Sharma and O. P. Pandey (2021): "Reaction kinetics during non-isothermal solid-state synthesis of boron trioxide via boric acid dehydration." Reaction Kinetics, Mechanisms and Catalysis, volume 134, pages 347–359. doi:10.1007/s11144-021-02084-8
- ^ a b c Siavash Aghili, Masoud Panjepour, and Mahmood Meratian (2018): "Kinetic analysis of formation of boron trioxide from thermal decomposition of boric acid under non-isothermal conditions." Journal of Thermal Analysis and Calorimetry, volume 131, pages 2443–2455. doi:10.1007/s10973-017-6740-3
- ^ Balci, Suna; Sezgi, Naime; Eren, Esin (2012). "Boron Oxide Production Kinetics Using Boric Acid as Raw Material". Industrial & Engineering Chemistry Research. 51 (34): 11091–11096. doi:10.1021/ie300685x.
- ^ a b Jolly, W. L. (1984). Modern Inorganic Chemistry. McGraw-Hill. p. 198.
- ^ Masanori Tachikawa (2004): "A density functional study on hydrated clusters of orthoboric acid, B(OH)3(H2O)n (n=1–5)". Journal of Molecular Structure: THEOCHEM, volume 710, issues 1–3, pages 139-150. doi:10.1016/j.theochem.2004.09.008
- ^ Housecroft, C.E.; Sharpe, A.G. (2005). Inorganic Chemistry (2nd ed.). Pearson Prentice-Hall. pp. 314–5.
- ^ MHE. Comprehensive Chemistry for JEE Advanced 2014. Tata McGraw-Hill Education. p. 15.5. ISBN 978-1-259-06426-5 – via Google Books.
- ^ Darpan, Pratiyogita (1 May 2000). Competition Science Vision. Pratiyogita Darpan – via Internet Archive.
- ^ Tsuyumoto, I.; Oshio, T.; Katayama, K. (2007). "Preparation of highly concentrated aqueous solution of sodium borate". Inorganic Chemistry Communications. 10 (1): 20–22. doi:10.1016/j.inoche.2006.08.019.
- ^ Brown, Herbert C.; Mead, Edward J.; Shoaf, Charles J. (1956). "Convenient procedures for the preparation of alkyl borate esters". J. Am. Chem. Soc. 78 (15): 3613–3614. doi:10.1021/ja01596a015.
- ^ a b Mendham, J.; Denney, R. C.; Barnes, J. D.; Thomas, M. J. K. (2000), Vogel's Quantitative Chemical Analysis (6th ed.), New York: Prentice Hall, p. 357, ISBN 0-582-22628-7
- ^ a b NIST Special Publication. U.S. Government Printing Office. 1969.
- ^ "Toxicological Profile for Boron" (PDF). Centers for Disease Control. November 2010. p. 11.
- ^ "Report of the Food Quality Protection Act (FQPA) Tolerance Reassessment Eligibility Decision (TRED) for Boric Acid/Sodium Borate Salts" (PDF). United States Environmental Protection Agency. 2006. Archived from the original (PDF) on 6 October 2006. Retrieved 21 April 2008.
- ^ "CLH report for boric acid – Proposal for Harmonised Classification and Labelling". Lodz, Poland: Bureau for Chemical Substances. 23 April 2018. Retrieved 18 October 2018.
- ^ "Boric acid, ACC# 03260 MSDS" (PDF). 11 February 2008. Archived from the original (PDF) on 16 December 2011. Retrieved 24 September 2009.
- ^ Ishii, Y.; Fujizuka, N.; Takahashi, T.; et al. (1993). "A fatal case of acute boric acid poisoning". Clinical Toxicology. 31 (2): 345–352. doi:10.3109/15563659309000402. PMID 8492348.
- ^ Restuccio, A.; Mortensen, M. E.; Kelley, M. T. (1992). "Fatal ingestion of boric acid in an adult". American Journal of Emergency Medicine. 10 (6): 545–547. doi:10.1016/0735-6757(92)90180-6. PMID 1388380.
- ^ Duldner, J. E. (30 January 2009). "Boric acid poisoning". A.D.A.M. Medical Encyclopedia. MedLine Plus.
- ^ NSW Food Authority. "Borax and boric acid". Australia: New South Wales Government. Archived from the original on 15 October 2009. Retrieved 24 September 2009.
- ^ "Boric acid as a substance of very high concern because of its CMR properties" (PDF). ECHA Document library. Archived from the original (PDF) on 18 May 2016. Retrieved 28 May 2017.
- ^ Regulation (EC) No 1272/2008 of the European Parliament and of the Council, 16 December 2008
- ^ Kistler, R. B.; Helvaci, C. (1994). "Boron and Borates". In Carr, D. D. (ed.). Industrial Minerals and Rocks (6th ed.). Littleton, CO: SME. pp. 171–186.
- ^ Tsuyumoto, I.; Oshio, T. (2009). "Development of fire resistant laminated wood using concentrated sodium polyborate aqueous solution". Journal of Wood Chemistry and Technology. 29 (4): 277–285. doi:10.1080/02773810903033721. S2CID 98730912.
- ^ Dempsey, Jock (2009) [1998]. "Borax". Dempsey's Forge. Retrieved 23 July 2010.
- ^ Prager, Felice. "Science Becomes a Toy – Silly Putty". Loti.com. Rewind the Fifites. Archived from the original on 12 May 2013. Retrieved 7 June 2013.
- ^ "Chemicals used by hydraulic fracturing companies in pennsylvania for surface and hydraulic fracturing activities" (PDF). Pennsylvania Department of Environmental Protection, Bureau of Oil and Gas Management. 30 June 2010.
- ^ Fink, Johannes (2015). "Fracturing fluids". Petroleum Engineer's Guide to Oil Field Chemicals and Fluids. pp. 567–651. doi:10.1016/B978-0-12-803734-8.00017-5. ISBN 978-0-12-803734-8.
- ^ Bishop, Maximilienne; Shahid, Naureen; Yang, Jianzhong; Barron, Andrew R. (2004). "Determination of the mode and efficacy of the cross-linking of guar by borate using MAS11B NMR of borate cross-linked guar in combination with solution11B NMR of model systems". Dalton Trans. (17): 2621–2634. doi:10.1039/B406952H. ISSN 1477-9226. PMID 15514744.
- ^ "European Patent EP3004279A1. Concentrated borate crosslinking solutions for use in hydraulic fracturing operations". European Patent Office. Retrieved 27 October 2019.
- ^ Wang, Shibin; Tang, Hongbiao; Guo, Jianchun; Wang, Kunjie (2016). "Effect of pH on the rheological properties of borate crosslinked hydroxypropyl guar gum hydrogel and hydroxypropyl guar gum". Carbohydrate Polymers. 147: 455–463. doi:10.1016/j.carbpol.2016.04.029. ISSN 0144-8617. PMID 27178952.
- ^ Electrical Sector Solutions Product Overview (PDF). Vol. 14: Fuses. Eaton Corporation. 2011.
- ^ Strom, A. P.; Rawlins, H. L. (December 1932). "The Boric Acid Fuse". Transactions of the American Institute of Electrical Engineers. 51 (4): 1020–1025. doi:10.1109/T-AIEE.1932.5056215. ISSN 0096-3860. S2CID 51650608.
- ^ a b Iavazzo C, Gkegkes ID, Zarkada IM, Falagas ME (August 2011). "Boric acid for recurrent vulvovaginal candidiasis: the clinical evidence". J Womens Health (Larchmt). 20 (8): 1245–55. doi:10.1089/jwh.2010.2708. PMID 21774671.
- ^ Abercrombie, P. (2010). "Vaginitis". In Maizes, V.; Low Dog, T. (eds.). Integrative Women's Health. New York, NY: Oxford University Press. p. 192. doi:10.1093/med/9780190214791.003.0013. ISBN 978-0-19-537881-8.
- ^ Sobel JD, Sobel R (August 2021). "Current and emerging pharmacotherapy for recurrent bacterial vaginosis". Expert Opin Pharmacother. 22 (12): 1593–1600. doi:10.1080/14656566.2021.1904890. PMID 33750246. S2CID 232325625.
- ^ "TOL 463 – AdisInsight".
- ^ Nyirjesy P, Brookhart C, Lazenby G, Schwebke J, Sobel JD (April 2022). "Vulvovaginal Candidiasis: A Review of the Evidence for the 2021 Centers for Disease Control and Prevention of Sexually Transmitted Infections Treatment Guidelines". Clin Infect Dis. 74 (Suppl_2): S162–S168. doi:10.1093/cid/ciab1057. PMID 35416967.
- ^ Marrazzo JM, Dombrowski JC, Wierzbicki MR, Perlowski C, Pontius A, Dithmer D, Schwebke J (February 2019). "Safety and Efficacy of a Novel Vaginal Anti-infective, TOL-463, in the Treatment of Bacterial Vaginosis and Vulvovaginal Candidiasis: A Randomized, Single-blind, Phase 2, Controlled Trial". Clin Infect Dis. 68 (5): 803–809. doi:10.1093/cid/ciy554. PMC 6376090. PMID 30184181.
- ^ Adriztina, I.; Adenin, L. I.; Lubis, Y. M. (January 2018). "Efficacy of Boric Acid as a Treatment of Choice for Chronic Suppurative Otitis Media and Its Ototoxicity". Korean J Fam Med. 39 (1): 2–9. doi:10.4082/kjfm.2018.39.1.2. PMC 5788841. PMID 29383205.
- ^ UK standards for microbiology investigations (PDF). Public Health England. 7 August 2017.
- ^ Harvey, S. C. (1980). "Antiseptics and Disinfectants; Fungicides; Ectoparasiticides". In Gilman, A. G.; Goodman, L. S.; Gilman, A. (eds.). Goodman & Gillman's The Pharmacological Basis of Therapeutics (6th ed.). Macmillan. p. 971. ISBN 978-0-02-344720-4.
- ^ "Method 3052 microwave assisted acid digestion of siliceous and organically based matrices" (PDF). US EPA. 22 June 2015.
- ^ "Borates in Pesticides | AMERICAN BORATE COMPANY".
- ^ Boone, C.; Bond, C.; Stone, D. (2012). "Boric Acid General Fact Sheet". National Pesticide Information Center, Oregon State University Extension Services.
- ^ "R.E.D. Facts – Boric Acid" (PDF). United States Environmental Protection Agency. Archived (PDF) from the original on 23 December 2021. Retrieved 2 April 2022.
- ^ Birch, Robert G (2013). "Boric acid as a swimming pool buffer" (PDF). The University of Queensland. Retrieved 30 November 2013.
- ^ Düzcükoğlu, H.; Acaroğlu, M. (2009). "Lubrication Properties of Vegetable Oils Combined with Boric Acid and Determination of Their Effects on Wear". Energy Sources, Part A: Recovery, Utilization, and Environmental Effects. 32 (3): 275–285. doi:10.1080/15567030802606053. S2CID 97537085.
- ^ Singh, Harpreet. "Standard equipments". Punjab State Carrom Association. Archived from the original on 14 March 2007. Retrieved 24 September 2009.
- ^ Weingart, George (1947). Pyrotechnics. Chemical Publishing Company. ISBN 978-0-8206-0112-0.
- ^ "Use of Boric Acid and Borax in Food". cfs.gov.hk. Retrieved 22 May 2019.
Further reading[edit]
- Jolly, W. L. (1991). Modern Inorganic Chemistry (2nd ed.). New York: McGraw-Hill. ISBN 978-0-07-112651-9.
- Goodman, L.; Gilman, A.; Brunton, L.; Lazo, J.; Parker, K. (2006). Goodman & Gilman's The Pharmacological Basis of Therapeutics. New York: McGraw Hill.
- Cordia JA, Bal EA, Mak WA and Wils ERJ (2003), Determination of some physico-chemical properties of Optibor EP. Rijswijk, The Netherlands: TNO Prins Maurits Laboratory, report PML 2002-C42rr, GLP, Unpublished, confidential data provided by Bor ax Europe Limited
External links[edit]
 Media related to Boric acid at Wikimedia Commons
Media related to Boric acid at Wikimedia Commons- Boric Acid Technical Fact Sheet – National Pesticide Information Center
- Boric Acid General Fact Sheet – National Pesticide Information Center
- International Chemical Safety Card 0991
- US EPA Pesticide Reregistration Eligibility Decision
- National Pollutant Inventory – Boron and compounds
- Boric acid at ChemicalLand21
- European Chemicals Agency (ECHA)"New Public Consultation on Eight Potential Substances of Very High Concern" – includes Boric Acid. Closes 22 April 2010
- ChemSub Online: Boric acid