Lanicemine
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
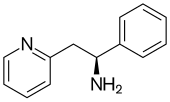
| Formula | C13H14N2 |
| Molar mass | 198.26 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lanicemine (AZD6765) is a low-trapping NMDA receptor antagonist developed by AstraZeneca,[1] which was being studied for the management of severe and treatment-resistant depression.[2] It was originally developed as a neuroprotective agent, but was redeveloped as an antidepressant following the observation that the NMDA receptor antagonist ketamine has potent antidepressant effects, but also has hallucinogenic side effects which make it unsuitable for use as an antidepressant in most circumstances. Lanicemine differs from ketamine in that it is a low-trapping NMDA receptor antagonist, showing similar rapid-acting antidepressant effects to ketamine in clinical trials but with little or no psychotomimetic side effects.[3] However, lanicemine did not meet study endpoints, and its development was terminated by AstraZeneca in 2013.[4]
See also
- AD-1211
- AV-101
- CERC-301
- Diphenidine
- Ephenidine
- Esketamine
- Lefetamine
- Memantine
- Methoxphenidine (MXP)
- MT-45
- NRX-1074
- Rapastinel
References
- ^ US 6518432, Melvyn E. Giles, "Process"
- ^ "AstraZeneca Annual Report and Form 20-F Information 2012" (PDF).
- ^ Zarate, C. A.; Mathews, D.; Ibrahim, L.; Chaves, J. F.; Marquardt, C.; Ukoh, I.; Jolkovsky, L.; Brutsche, N. E.; Smith, M. A.; Luckenbaugh, D. A. (2012). "A Randomized Trial of a Low-Trapping Nonselective N-Methyl-D-Aspartate Channel Blocker in Major Depression". Biological Psychiatry. 74 (4): 257–264. doi:10.1016/j.biopsych.2012.10.019. PMC 3594049. PMID 23206319.
- ^ Flowers, Sophie. "Return to growth: AstraZeneca's CEO Pascal Soriot says 2013 was year of "momentum" for the company". Retrieved 6 February 2014.
