MMDMA
From Wikipedia, the free encyclopedia
This is an old revision of this page, as edited by DMacks (talk | contribs) at 20:40, 19 June 2020 (Remove malformatted |molecular_weight= when infobox can autocalculate it, per Wikipedia talk:WikiProject Pharmacology#Molecular weights in drugboxes (via WP:JWB)). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 20:40, 19 June 2020 by DMacks (talk | contribs) (Remove malformatted |molecular_weight= when infobox can autocalculate it, per Wikipedia talk:WikiProject Pharmacology#Molecular weights in drugboxes (via WP:JWB))
The topic of this article may not meet Wikipedia's general notability guideline. Please help to demonstrate the notability of the topic by citing reliable secondary sources that are independent of the topic and provide significant coverage of it beyond a mere trivial mention. If notability cannot be shown, the article is likely to be merged, redirected, or deleted. Find sources: "MMDMA" – news · newspapers · books · scholar · JSTOR (November 2015) (Learn how and when to remove this message) |
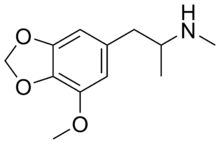 | |
| Clinical data | |
|---|---|
| Other names | 5-MeO-MDMA;3-methoxy-4,5-methylenedioxy-N-methylamphetamine[1] |
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C12H17NO3 |
| Molar mass | 223.272 g·mol−1 |
| 3D model (JSmol) | |
| |
5-Methoxy-3,4-methylenedioxymethamphetamine (MMDMA; 5-MeO-MDMA) is a designer drug of the substituted methylenedioxyphenethylamine (MDxx) class.[2] Little is known about its effects and it has not been formally studied in animals.
References
- ^ Bailey K, By A, Legault D, Verner D (1976). "Identification of the N-Methylated Analogs of the Hallucinogenic Amphetamines and Some Isomers". Journal of the Association of Official Analytical Chemists. 58 (1): 62–69.
- ^ Clark CR, Deruiter J, Noggle FT (1996). "Analysis of 1-(3-methoxy-4,5-methylenedioxyphenyl)-2-propanamine (MMDA) derivatives synthesized from nutmeg oil and 3-methoxy-4,5-methylenedioxybenzaldehyde". Journal of Chromatographic Science. 34 (1): 34–42. doi:10.1093/chromsci/34.1.34.
| Phenylalkyl- amines (other than cathinones) |
|
|---|---|
| Cyclized phenyl- alkylamines | |
| Cathinones | |
| Tryptamines | |
| Chemical classes | |
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
Retrieved from "https://en.wikipedia.org/w/index.php?title=MMDMA&oldid=963442409"
Hidden categories:
- Articles with topics of unclear notability from November 2015
- All articles with topics of unclear notability
- Drugs with non-standard legal status
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
