UWA-101
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
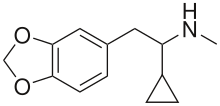
| Formula | C13H17NO2 |
| Molar mass | 219.284 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
UWA-101 (also known as α-cyclopropyl-MDMA) is a phenethylamine derivative researched as a potential treatment for Parkinson's disease. Its chemical structure is very similar to that of the illegal drug MDMA, the only difference being the replacement of the α-methyl group with an α-cyclopropyl group. MDMA has been found in animal studies and reported in unauthorised human self-experiments to be effective in the short-term relief of side-effects of Parkinson's disease therapy, most notably levodopa-induced dyskinesia.[1][2][3][4] However the illegal status of MDMA and concerns about its potential for recreational use, neurotoxicity and potentially dangerous side effects mean that it is unlikely to be investigated for medical use in this application, and so alternative analogues were investigated.[5]
Replacing the α-methyl with a cyclopropyl dramatically reduces affinity for the noradrenaline transporter and 5-HT2A receptor, while retaining high serotonin transporter affinity and markedly increasing affinity for the dopamine transporter (and as such, it is one of the few selective SDRIs or serotonin-dopamine reuptake inhibitors). This change causes UWA-101 to lack cytotoxicity and MDMA-like behavioral effects in animals, while retaining similar or slightly improved antidyskinetic effectiveness when compared to MDMA.[6] This research was a continuation of earlier work from the same team led by medicinal chemist Matthew Piggott, at the University of Western Australia, which showed that replacing the α-methyl group of MDMA with larger aromatic ring systems produced compounds which lacked psychoactivity and neurotoxicity, but had potent anti-cancer effects against Burkitt's lymphoma cells in vitro.[7][8]
UWA-121 is the (R)-enantiomer of UWA-101 and the (S)-enantiomer is UWA-122.[9] Both are active monoamine reuptake inhibitors.[9] UWA-121 is an SDRI with 10-fold preference for inhibition of dopamine reuptake over serotonin reuptake while UWA-122 is a highly selective serotonin reuptake inhibitor.[9]
Another relative is UWA-104 ("α-isopropyl-MDMA"), which is also active.[6] It is a selective dopamine reuptake inhibitor, with greater than 30-fold selectivity over the other monoamine transporters or the serotonin 5-HT2A receptor.[6] Other analogues include UWA-091 ("α-propyl-MDMA"), UWA-102 ("α-tert-butyl-MDMA"), and UWA-001 ("α-phenyl-MDMA").[6] The in vitro pharmacological activities of UWA-101, UWA-104, and UWA-001 have been reported.[6]
See also
[edit]- MBDB
- Methyl-K (UWA-091)
- Isohexylone
- UWA-001
- Zylofuramine
- RTI-83 - another drug which selectively increases dopamine and serotonin levels without affecting noradrenaline
References
[edit]- ^ Schmidt WJ, Mayerhofer A, Meyer A, Kovar KA (September 2002). "Ecstasy counteracts catalepsy in rats, an anti-parkinsonian effect?". Neuroscience Letters. 330 (3): 251–4. doi:10.1016/s0304-3940(02)00823-6. PMID 12270640. S2CID 41609012.
- ^ Iravani MM, Jackson MJ, Kuoppamäki M, Smith LA, Jenner P (October 2003). "3,4-methylenedioxymethamphetamine (ecstasy) inhibits dyskinesia expression and normalizes motor activity in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated primates". The Journal of Neuroscience. 23 (27): 9107–15. doi:10.1523/JNEUROSCI.23-27-09107.2003. PMC 6740822. PMID 14534244.
- ^ Lebsanft HB, Kohles T, Kovar KA, Schmidt WJ (March 2005). "3,4-Methylenedioxymethamphetamine counteracts akinesia enantioselectively in rat rotational behavior and catalepsy". Synapse. 55 (3): 148–55. doi:10.1002/syn.20102. PMID 15602749. S2CID 24601744.
- ^ Huot P, Johnston TH, Lewis KD, Koprich JB, Reyes MG, Fox SH, et al. (May 2011). "Characterization of 3,4-methylenedioxymethamphetamine (MDMA) enantiomers in vitro and in the MPTP-lesioned primate: R-MDMA reduces severity of dyskinesia, whereas S-MDMA extends duration of ON-time". The Journal of Neuroscience. 31 (19): 7190–8. doi:10.1523/JNEUROSCI.1171-11.2011. PMC 6703214. PMID 21562283.
- ^ Jerome I (Spring 2008). "MDMA and Parkinson's: Lots of Research, Few Practical Answers" (PDF). MAPS. XVI (1): 16–18. Archived (PDF) from the original on 2011-09-15. Retrieved 2012-04-09.
- ^ a b c d e Johnston TH, Millar Z, Huot P, Wagg K, Thiele S, Salomonczyk D, et al. (May 2012). "A novel MDMA analogue, UWA-101, that lacks psychoactivity and cytotoxicity, enhances L-DOPA benefit in parkinsonian primates". FASEB Journal. 26 (5): 2154–63. doi:10.1096/fj.11-195016. PMID 22345403. S2CID 37589231.
- ^ Gandy MN, McIldowie M, Lewis K, Wasik AM, Salomonczyk D, Wagg K, et al. (2010). "Redesigning the designer drug ecstasy: nonpsychoactive MDMA analogues exhibiting Burkitt's lymphoma cytotoxicity". MedChemComm. 1 (4): 287–293. doi:10.1039/c0md00108b.
- ^ Wasik AM, Gandy MN, McIldowie M, Holder MJ, Chamba A, Challa A, et al. (August 2012). "Enhancing the anti-lymphoma potential of 3,4-methylenedioxymethamphetamine ('ecstasy') through iterative chemical redesign: mechanisms and pathways to cell death" (PDF). Investigational New Drugs. 30 (4): 1471–83. doi:10.1007/s10637-011-9730-5. PMID 21850491. S2CID 20880580. Archived (PDF) from the original on 2020-03-10. Retrieved 2019-09-18.
- ^ a b c Huot P, Johnston TH, Lewis KD, Koprich JB, Reyes MG, Fox SH, et al. (July 2014). "UWA-121, a mixed dopamine and serotonin re-uptake inhibitor, enhances L-DOPA anti-parkinsonian action without worsening dyskinesia or psychosis-like behaviours in the MPTP-lesioned common marmoset". Neuropharmacology. 82: 76–87. doi:10.1016/j.neuropharm.2014.01.012. PMID 24447715. S2CID 37160397.
External links
[edit]- Do neurologists dance? A personal experience of Parkinson's disease & MDMA, Tim Lawrence, 2003
