Calcium chloride

| |
| Names | |
|---|---|
| IUPAC name
Calcium chloride
| |
| Other names
Calcium(II) chloride, calcium dichloride, E509
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.030.115 |
| EC Number |
|
| E number | E509 (acidity regulators, ...) |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CaCl2 | |
| Molar mass | 110.98 g·mol−1 |
| Appearance | White powder, hygroscopic |
| Odor | Odorless |
| Density |
|
| Melting point | 772–775 °C (1,422–1,427 °F; 1,045–1,048 K) anhydrous[5] 260 °C (500 °F; 533 K) monohydrate, decomposes 175 °C (347 °F; 448 K) dihydrate, decomposes 45.5 °C (113.9 °F; 318.6 K) tetrahydrate, decomposes[5] 30 °C (86 °F; 303 K) hexahydrate, decomposes[1] |
| Boiling point | 1,935 °C (3,515 °F; 2,208 K) anhydrous[1] |
| Anhydrous: 74.5 g/100 mL (20 °C)[2] Hexahydrate: 49.4 g/100 mL (−25 °C) 59.5 g/100 mL (0 °C) 65 g/100 mL (10 °C) 81.1 g/100 mL (25 °C)[1] 102.2 g/100 mL (30.2 °C) α-Tetrahydrate: 90.8 g/100 mL (20 °C) 114.4 g/100 mL (40 °C) Dihydrate: 134.5 g/100 mL (60 °C) 152.4 g/100 mL (100 °C)[3] | |
| Solubility | |
| Solubility in ethanol |
|
| Solubility in methanol |
|
| Solubility in acetone | 0.1 g/kg (20 °C)[4] |
| Solubility in pyridine | 16.6 g/kg[4] |
| Acidity (pKa) |
|
| −5.47·10−5 cm3/mol[1] | |
Refractive index (nD)
|
1.52 |
| Viscosity |
|
| Structure | |
| |
| |
| |
a = 6.259 Å, b = 6.444 Å, c = 4.17 Å (anhydrous, 17 °C)[6] α = 90°, β = 90°, γ = 90°
| |
| Octahedral (Ca2+, anhydrous) | |
| Thermochemistry | |
Heat capacity (C)
|
|
Std molar
entropy (S⦵298) |
108.4 J/mol·K[1][5] |
Std enthalpy of
formation (ΔfH⦵298) |
|
Gibbs free energy (ΔfG⦵)
|
−748.81 kJ/mol[1][5] |
| Pharmacology | |
| A12AA07 (WHO) B05XA07 (WHO), G04BA03 (WHO) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| GHS labelling: | |
 [7] [7]
| |
| Warning | |
| H319[7] | |
| P305+P351+P338[7] | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1,000-1,400 mg/kg (rats, oral)[8] |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Calcium chloride is an inorganic compound, a salt with the chemical formula CaCl2. It is a colorless crystalline solid at room temperature, highly soluble in water.
Calcium chloride is commonly encountered as a hydrated solid with generic formula CaCl2(H2O)x, where x = 0, 1, 2, 4, and 6. These compounds are mainly used for de-icing and dust control. Because the anhydrous salt is hygroscopic, it is used as a desiccant.[10]
Uses
De-icing and freezing-point depression

By depressing the freezing point of water, calcium chloride is used to prevent ice formation and is used to de-ice. This application consumes the greatest amount of calcium chloride. Calcium chloride is relatively harmless to plants and soil. As a de-icing agent, it is much more effective at lower temperatures than sodium chloride. When distributed for this use, it usually takes the form of small, white spheres a few millimeters in diameter, called prills. Solutions of calcium chloride can prevent freezing at temperature as low as −52 °C (−62 °F), making it ideal for filling agricultural implement tires as a liquid ballast, aiding traction in cold climates.[11]
It is also used in domestic and industrial chemical air dehumidifiers.[12]
Road surfacing

The second largest application of calcium chloride exploits hygroscopic properties and the tackiness of its hydrates. A concentrated solution keeps a liquid layer on the surface of dirt roads, which suppresses formation of dust. It keeps the finer dust particles on the road, providing a cushioning layer. If these are allowed to blow away, the large aggregate begins to shift around and the road breaks down. Using calcium chloride reduces the need for grading by as much as 50% and the need for fill-in materials as much as 80%.[13]
Water treatment
Calcium chloride is used to increase the water hardness in swimming pools. This process reduces the erosion of the concrete in the pool. By Le Chatelier's principle and the common-ion effect, increasing the concentration of calcium in the water reduces the dissolution of calcium compounds essential to the structure of concrete.[citation needed]
Food
The average intake of calcium chloride as food additives has been estimated to be 160–345 mg/day.[14] Calcium chloride is permitted as a food additive in the European Union for use as a sequestrant and firming agent with the E number E509. It is considered as generally recognized as safe (GRAS) by the U.S. Food and Drug Administration.[15] Its use in organic crop production is generally prohibited under the US National Organic Program.[16]
In marine aquariums, calcium chloride is one way to introduce bioavailable calcium for calcium carbonate-shelled animals such as mollusks and some cnidarians. Calcium hydroxide (kalkwasser mix) or a calcium reactor can also be used.
As a firming agent, calcium chloride is used in canned vegetables, in firming soybean curds into tofu and in producing a caviar substitute from vegetable or fruit juices.[17] It is commonly used as an electrolyte in sports drinks and other beverages, including bottled water. The extremely salty taste of calcium chloride is used to flavor pickles without increasing the food's sodium content. Calcium chloride's freezing-point depression properties are used to slow the freezing of the caramel in caramel-filled chocolate bars. Also, it is frequently added to sliced apples to maintain texture.
In brewing beer, calcium chloride is sometimes used to correct mineral deficiencies in the brewing water. It affects flavor and chemical reactions during the brewing process, and can also affect yeast function during fermentation.
In cheesemaking, calcium chloride is sometimes added to processed (pasteurized/homogenized) milk to restore the natural balance between calcium and protein in casein. It is added before the coagulant.
Calcium chloride is used to prevent cork spot and bitter pit on apples by spraying on the tree during the late growing season.[18]
Laboratory and related drying operations
Drying tubes are frequently packed with calcium chloride. Kelp is dried with calcium chloride for use in producing sodium carbonate. Anhydrous calcium chloride has been approved by the FDA as a packaging aid to ensure dryness (CPG 7117.02).[19]
Medicine
It is injected to treat internal hydrofluoric acid burns. It can be used to treat magnesium intoxication. Calcium chloride injection may improve the appearance of an electrocardiogram. It can help to protect the myocardium from dangerously high levels of serum potassium in hyperkalemia. Calcium chloride can be used to quickly treat calcium channel blocker toxicity, from the side effects of drugs such as diltiazem.[20]
Cardiac arrest
While intravenous calcium has been used to treat cardiac arrest, its general use is not recommended.[21] Cases of cardiac arrest in which it is still recommended include high blood potassium, low blood calcium such as may occur following blood transfusions, and calcium channel blocker overdose.[21] There is the potential that general use could worsen outcomes.[21] If calcium is used, calcium chloride is generally the recommended form.[21]
Science
Aqueous calcium chloride is used in genetic transformation of cells by increasing the cell membrane permeability, inducing competence for DNA uptake (allowing DNA fragments to enter the cell more readily).
Miscellaneous applications
Calcium chloride is used in concrete mixes to accelerate the initial setting, but chloride ions lead to corrosion of steel rebar, so it should not be used in reinforced concrete.[22] The anhydrous form of calcium chloride may also be used for this purpose and can provide a measure of the moisture in concrete.[23]
Calcium chloride is included as an additive in plastics and in fire extinguishers, in wastewater treatment as a drainage aid, in blast furnaces as an additive to control scaffolding (clumping and adhesion of materials that prevent the furnace charge from descending), and in fabric softener as a thinner.
The exothermic dissolution of calcium chloride is used in self-heating cans and heating pads.
In the oil industry, calcium chloride is used to increase the density of solids-free brines. It is also used to provide inhibition of swelling clays in the water phase of invert emulsion drilling fluids.
CaCl2 acts as flux material (decreasing melting point) in the Davy process for the industrial production of sodium metal, through the electrolysis of molten NaCl.
Similarly, CaCl2 is used as a flux and electrolyte in the FFC Cambridge process for titanium production, where it ensures the proper exchange of calcium and oxygen ions between the electrodes.
Calcium chloride is also used in the production of activated charcoal.
Calcium chloride is also an ingredient used in ceramic slipware. It suspends clay particles so that they float within the solution making it easier to use in a variety of slipcasting techniques.
Animal sterilization
Calcium chloride dihydrate (20% by weight) dissolved in ethanol (95% ABV) has been used as a sterilant for male animals. The solution is injected into the testes of the animal. Within 1 month, necrosis of testicular tissue results in sterilization.[24][25]
Hazards
Calcium chloride can act as an irritant by desiccating moist skin. Solid calcium chloride dissolves exothermically, and burns can result in the mouth and esophagus if it is ingested. Ingestion of concentrated solutions or solid products may cause gastrointestinal irritation or ulceration.[26]
Consumption of calcium chloride can lead to hypercalcemia.[27]
Properties

Calcium chloride dissolves in water, producing chloride and the aquo complex [Ca(H2O)6]2+. In this way, these solutions are sources of "free" calcium and free chloride ions. This description is illustrated by the fact that these solutions react with phosphate sources to give a solid precipitate of calcium phosphate:
- 3 CaCl2 + 2 PO3−
4 → Ca3(PO4)2 + 6 Cl−
Calcium chloride has a very high enthalpy change of solution, indicated by considerable temperature rise accompanying dissolution of the anhydrous salt in water. This property is the basis for its largest-scale application.
Molten calcium chloride can be electrolysed to give calcium metal and chlorine gas:
- CaCl2 → Ca + Cl2
Preparation
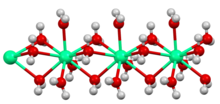
In much of the world, calcium chloride is derived from limestone as a by-product of the Solvay process:[10] North American consumption in 2002 was 1,529,000 tonnes (3.37 billion pounds).[28]
- 2 NaCl + CaCO3 → Na2CO3 + CaCl2
In the US, most of calcium chloride is obtained by purification from brine. A Dow Chemical Company manufacturing facility in Michigan houses about 35% of the total U.S. production capacity for calcium chloride.[29]
As with most bulk commodity salt products, trace amounts of other cations from the alkali metals and alkaline earth metals (groups 1 and 2) and other anions from the halogens (group 17) typically occur, but the concentrations are trifling.
Occurrence
Calcium chloride occurs as the rare evaporite minerals sinjarite (dihydrate) and antarcticite (hexahydrate). The related minerals chlorocalcite (potassium calcium chloride, KCaCl3) and tachyhydrite (calcium magnesium chloride, CaMg2Cl6·12H2O) are also very rare.
See also
References
- ^ a b c d e f g h i Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ "CALCIUM CHLORIDE (ANHYDROUS)". ICSC. International Programme on Chemical Safety and the European Commission.
- ^ Seidell, Atherton; Linke, William F. (1919). Solubilities of Inorganic and Organic Compounds (2nd ed.). New York: D. Van Nostrand Company. p. 196.
- ^ a b c d e f Anatolievich, Kiper Ruslan. "Properties of substance: calcium chloride". chemister.ru. Retrieved 7 July 2014.
- ^ a b c d e f Pradyot, Patnaik (2003). Handbook of Inorganic Chemicals. The McGraw-Hill Companies, Inc. p. 162. ISBN 0-07-049439-8.
- ^ a b c d
Müller, Ulrich (2006). Inorganic Structural Chemistry (2nd ed.). England: John Wiley & Sons Ltd. p. 33. ISBN 978-0-470-01864-4.
{{cite book}}:|work=ignored (help) - ^ a b c Sigma-Aldrich Co., Calcium chloride. Retrieved on 2014-07-07.
- ^ Donald E. Garrett (2004). Handbook of Lithium and Natural Calcium Chloride. p. 379. ISBN 0080472907. Retrieved 29 August 2018.
Its toxicity upon ingestion, is indicated by the test on rats: oral LD50 (rat) is 1.0–1.4 g/kg (the lethal dose for half of the test animals, in this case rats...)
- ^ "MSDS of Calcium chloride". fishersci.ca. Fisher Scientific. Retrieved 7 July 2014.
- ^ a b Robert Kemp, Suzanne E. Keegan "Calcium Chloride" in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a04_547
- ^ "Binary Phase diagram: The Calcium Chloride – water system". Aqueous Solutions Aps. October 2016. Retrieved 20 April 2017.
- ^ "humantouchofchemistry.com Keeping Things Dry". Retrieved 23 October 2014.
- ^ "Dust: Don't Eat It! Control It!". Road Management & Engineering Journal. US Roads (TranSafety Inc.). 1 June 1998. Retrieved 9 August 2006.
- ^ Calcium Chloride SIDS Initial Assessment Profile, UNEP Publications, SIAM 15, Boston, 22–25 October 2002, pp. 13–14.
- ^ 21 CFR § 184.1193
- ^ 7 CFR § 205.602
- ^ "Apple Caviar Technique". StarChefs Studio. StarChefs.com. April 2004. Retrieved 9 August 2006.
- ^ "Cork Spot and Bitter Pit of Apples", Richard C. Funt and Michael A. Ellis, Ohioline.osu.edu/factsheet/plpath-fru-01
- ^ "CPG 7117.02". FDA Compliance Articles. US Food and Drug Administration. March 1995. Retrieved 3 December 2007.
- ^ "Calcium chloride Prescribing Information". Hospira, Inc. November 2009. Retrieved 10 June 2011.
- ^ a b c d "Calcium Salts". The American Society of Health-System Pharmacists. Retrieved 8 January 2017.
- ^ "Accelerating Concrete Set Time". Federal Highway Administration. 1 June 1999. Retrieved 16 January 2007.
- ^ National Research Council (U.S.). Building Research Institute (1962). Adhesives in Building: Selection and Field Application; Pressure-sensitive Tapes. National Academy of Science-National Research Council. pp. 24–5.
- ^ Koger, Nov 1977, "Calcium Chloride, Practical Necrotizing Agent", Journal of the American Association of Bovine Practitioners (USA), (Nov 1977), v. 12, p. 118–119
- ^ Jana, K.; Samanta, P.K. (2011). "Clinical evaluation of non-surgical sterilization of male cats with single intra-testicular injection of calcium chloride". BMC Vet. Res. 7: 39. doi:10.1186/1746-6148-7-39. PMC 3152893. PMID 21774835.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Product Safety Assessment (PSA): Calcium Chloride". Dow Chemical Company. 2 May 2006.
- ^ "Calcium Chloride Possible Side Affects".
- ^ Calcium Chloride SIDS Initial Assessment Profile, UNEP Publications, SIAM 15, Boston, 22–25 October 2002, page 11.
- ^ Calcium Chloride Chemical Profile, The Innovation Group, www.the-innovation-group.com, printed 9 September 2005.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
External links
- International Chemical Safety Card 1184
- Product and Application Information (Formerly Dow Chemical Calcium Chloride division)
- Report on steel corrosion by chloride including CaCl2
- Collection of calcium chloride reports and articles
- Calcium chloride, Anhydrous MSDS
- Difusivity of calcium chloride
- Centers for Disease Control and Prevention, National Institutes of Occupational Safety and Health, "Calcium Chloride (anhydrous)"

