Linsitinib
Appearance
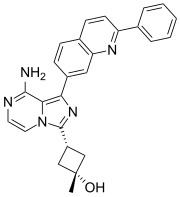
| |
| Names | |
|---|---|
| Preferred IUPAC name
(1s,3s)-3-[8-Amino-1-(2-phenylquinolin-7-yl)imidazo[1,5-a]pyrazin-3-yl]-1-methylcyclobutan-1-ol | |
| Other names
OSI-906
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C26H23N5O | |
| Molar mass | 421.504 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Linsitinib is an experimental drug candidate for the treatment of various types of cancer. It is an inhibitor of the insulin receptor and of the insulin-like growth factor 1 receptor (IGF-1R).[1] This prevents tumor cell proliferation and induces tumor cell apoptosis.[2]
Linsitinib was granted orphan drug designation for adrenocortical carcinoma in March 2012.[3]
Phase II clinical trials were initiated for multiple myeloma, ovarian cancer, hepatocellular carcinoma, and NSCLC, but subsatisfactory results caused research for these indications to be discontinued.[3] A phase III clinical trial found that linsitinib did not increase survival in patients with adrenocortical carcinoma.[4] As of 2017, no clinical trials were in progress.[3]
References
[edit]- ^ Mulvihill, Mark J; Cooke, Andrew; Rosenfeld-Franklin, Maryland; Buck, Elizabeth; Foreman, Ken; Landfair, Darla; O’Connor, Matthew; Pirritt, Caroline; Sun, Yingchaun; Yao, Yan; Arnold, Lee D; Gibson, Neil W; Ji, Qun-Sheng (September 2009). "Discovery of OSI-906: a selective and orally efficacious dual inhibitor of the IGF-1 receptor and insulin receptor". Future Medicinal Chemistry. 1 (6): 1153–1171. doi:10.4155/fmc.09.89. PMID 21425998.
- ^ "Linsitinib". NCI Drug Dictionary. National Cancer Institute. Retrieved October 16, 2012.
- ^ a b c "Linsitinib - AdisInsight". adisinsight.springer.com.
- ^ Fassnacht, Martin; Berruti, Alfredo; Baudin, Eric; Demeure, Michael J; Gilbert, Jill; Haak, Harm; Kroiss, Matthias; Quinn, David I; Hesseltine, Elizabeth; Ronchi, Cristina L; Terzolo, Massimo; Toni Choueiri; Poondru, Srinivasu; Fleege, Tanya; Rorig, Ramona; Chen, Jihong; Stephens, Andrew W; Worden, Francis; Hammer, Gary D (April 2015). "Linsitinib (OSI-906) versus placebo for patients with locally advanced or metastatic adrenocortical carcinoma: a double-blind, randomised, phase 3 study". The Lancet Oncology. 16 (4): 426–435. doi:10.1016/S1470-2045(15)70081-1. hdl:2318/1534804. PMID 25795408.
External links
[edit] Media related to Linsitinib at Wikimedia Commons
Media related to Linsitinib at Wikimedia Commons
