Thromboxane
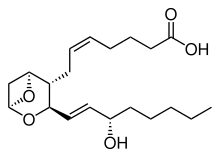

Thromboxane is a member of the family of lipids known as eicosanoids. The two major thromboxanes are thromboxane A2 and thromboxane B2. The distinguishing feature of thromboxanes is a 6-membered ether-containing ring.
Thromboxane is named for its role in blood clot formation (thrombosis).
Production[edit]


Thromboxane-A synthase, an enzyme found in platelets, converts the arachidonic acid derivative prostaglandin H2 to thromboxane.
People with asthma tend to have increased thromboxane production, and analogs of thromboxane act as bronchoconstrictors in patients with asthma.[1]
Mechanism[edit]
Thromboxane acts by binding to any of the thromboxane receptors, G-protein-coupled receptors coupled to the G protein Gq.[2]
Functions[edit]
Thromboxane is a vasoconstrictor and a potent hypertensive agent, and it facilitates platelet aggregation.
It is in homeostatic balance in the circulatory system with prostacyclin, a related compound. The mechanism of secretion of thromboxanes from platelets is still unclear. They act in the formation of blood clots and reduce blood flow to the site of a clot.
If the cap of a vulnerable plaque erodes or ruptures, as in myocardial infarction, platelets stick to the damaged lining of the vessel and to each other within seconds and form a plug. These "Sticky platelets" secrete several chemicals, including thromboxane A2 that stimulate vasoconstriction, reducing blood flow at the site.
Role of A2 in platelet aggregation[edit]
Thromboxane A2 (TXA2), produced by activated platelets, has prothrombotic properties, stimulating activation of new platelets as well as increasing platelet aggregation.
Platelet aggregation is achieved by mediating expression of the glycoprotein complex GP IIb/IIIa in the cell membrane of platelets. Circulating fibrinogen binds these receptors on adjacent platelets, further strengthening the clot.
Pathology[edit]
It is believed that the vasoconstriction caused by thromboxanes plays a role in Prinzmetal's angina. Omega-3 fatty acids are metabolized to produce higher levels of TxA3, which is relatively less potent than TxA2 and PGI3; therefore, there is a balance shift toward inhibition of vasoconstriction and platelet aggregation. It is believed that this shift in balance lowers the incidence of myocardial infarction (heart attack) and stroke. Vasoconstriction and, perhaps, various proinflammatory effects exerted by TxA on tissue microvasculature, is probable reason why the TxA is pathogenic in various diseases, such as ischemia-reperfusion injury.,[3] hepatic inflammatory processes,[4] acute hepatotoxicity [5] etc. TxB2, a stable degradation product of TxA2, plays a role in acute hepatoxicity induced by acetaminophen.[6][7]
Thromboxane inhibitors[edit]
Thromboxane inhibitors are broadly classified as either those that inhibit the synthesis of thromboxane, or those that inhibit the target effect of it.
Thromboxane synthesis inhibitors, in turn, can be classified regarding which step in the synthesis they inhibit:
- The widely used drug aspirin acts by inhibiting the ability of the COX enzyme to synthesize the precursors of thromboxane within platelets. Low-dose, long-term aspirin use irreversibly blocks the formation of thromboxane A2 in platelets, producing an inhibitory effect on platelet aggregation. This anticoagulant property makes aspirin useful for reducing the incidence of heart attacks.[8] 40 mg of aspirin a day is able to inhibit a large proportion of maximum thromboxane A2 release provoked acutely, with the prostaglandin I2 synthesis being little affected; however, higher doses of aspirin are required to attain further inhibition.[9]
- Thromboxane synthase inhibitors inhibit the final enzyme (thromboxane synthase) in the synthesis of thromboxane. Ifetroban is a potent and selective thromboxane receptor antagonist.[10] Dipyridamole antagonizes this receptor too, but has various other mechanisms of antiplatelet activity as well.
- High-dose naproxen can induce near-complete suppression of platelet thromboxane throughout the dosing interval and appears not to increase cardiovascular disease (CVD) risk, whereas other high-dose NSAID (non-steroidal-anti-inflammatory) regimens have only transient effects on platelet COX-1 and have been found to be associated "with a small but definite vascular hazard".[11]
The inhibitors of the target effects of thromboxane are the thromboxane receptor antagonist, including terutroban.
Picotamide has activity both as a thromboxane synthase inhibitor and as a thromboxane receptor antagonist.[12]
Ridogrel is another example.[13]
References[edit]
- ^ Chung, Kian Fan; Barnes, Peter J. (2009). "Mediator Antagonists". Asthma and COPD: 655–662. doi:10.1016/B978-0-12-374001-4.00052-3. ISBN 9780123740014. Retrieved 20 January 2023.
- ^ Rat kidney thromboxane receptor: molecular cloning, signal ...
- ^ Ito Y (2003). "Effects of selective cyclooxygenase inhibitors on ischemia/reperfusion-induced hepatic microcirculatory dysfunction in mice". Eur Surg Res. 35 (5): 408–16. doi:10.1159/000072174. PMID 12928598. S2CID 40932766.
- ^ Katagiri H (2004). "Role of thromboxane derived from COX-1 and -2 in hepatic microcirculatory dysfunction during endotoxemia in mice". Hepatology. 39 (1): 139–150. doi:10.1002/hep.20000. PMID 14752832. S2CID 27252438.
- ^ Yokoyama Y (2005). "Role of thromboxane in producing hepatic injury during a hepatic stress disorder". Arch. Surg. 140 (8): 801–7. doi:10.1001/archsurg.140.8.801. PMID 16103291.
- ^ Cavar I (2011). "Anti-thromboxane B2 antibodies protect against acetaminophen-induced liver injury in mice". Journal of Xenobiotics. 1 (1): 38–44. doi:10.4081/xeno.2011.e8.
- ^ Cavar I (2010). "The role of prostaglandin E2 in acute acetaminophen hepatotoxicity in mice". Histol Histopathol. 25 (7): 819–830. PMID 20503171.
- ^ [1] American Heart Association: Aspirin in Heart Attack and Stroke Prevention "The American Heart Association recommends aspirin use for patients who've had a myocardial infarction (heart attack), unstable angina, ischemic stroke (caused by blood clot) or transient ischemic attacks (TIAs or "little strokes"), if not contraindicated. This recommendation is based on sound evidence from clinical trials showing that aspirin helps prevent the recurrence of such events as heart attack, hospitalization for recurrent angina, second strokes, etc. (secondary prevention). Studies show aspirin also helps prevent these events from occurring in people at high risk (primary prevention)." [2]
- ^ Tohgi, H; S Konno; K Tamura; B Kimura; K Kawano (1992). "Effects of low-to-high doses of aspirin on platelet aggregability and metabolites of thromboxane A2 and prostacyclin". Stroke. 23 (10): 1400–1403. doi:10.1161/01.STR.23.10.1400. PMID 1412574. S2CID 14177039.
- ^ Dockens, RC; Santone, KS; Mitroka, JG; Morrison, RA; Jemal, M; Greene, DS; Barbhaiya, RH (Aug 2000). "Disposition of radiolabeled ifetroban in rats, dogs, monkeys, and humans". Drug Metabolism and Disposition. 28 (8): 973–80. PMID 10901709.
- ^ Coxib and traditional NSAID Trialists' (CNT) Collaboration (30 May 2013). "Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials". Lancet. 382 (9894): 769–79. doi:10.1016/S0140-6736(13)60900-9. PMC 3778977. PMID 23726390.
- ^ Ratti, S; Quarato, P; Casagrande, C; Fumagalli, R; Corsini, A (1998). "Picotamide, an antithromboxane agent, inhibits the migration and proliferation of arterial myocytes". European Journal of Pharmacology. 355 (1): 77–83. doi:10.1016/S0014-2999(98)00467-1. PMID 9754941.
- ^ Di Perri, T.; Notari, M.; Assogna, G. (October 1991). "[Ridogrel, a new platelet antiaggregant molecule with a double mechanism of action. A pharmacological and clinical profile]". Recenti Progressi in Medicina. 82 (10): 533–540. ISSN 0034-1193. PMID 1759039.
External links[edit]
- Thromboxanes at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
