Methyldesorphine
Appearance
 | |
| Clinical data | |
|---|---|
| Other names | 3-Hydroxy-6,N-dimethyl- 4,5-epoxymorphin-6-en |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.036.474 |
| Chemical and physical data | |
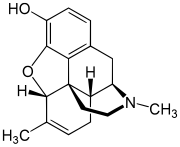
| Formula | C18H21NO2 |
| Molar mass | 283.36 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| | |
Methyldesorphine is an opioid analgesic. First synthesized in Germany in 1940 and patented in the USA in 1952,[1] it has a high potential for abuse as with any potent opioid agonist, and is sometimes found along with desomorphine as a component of the home-made opioid mixture known as "Krokodil" used in Russia and the neighboring former Soviet republics.[2] It is approximately 15 times more potent than morphine as an analgesic[3][4] but if the 6-7 bond is saturated, the β isomer is some 50 times more potent than morphine.
See also
References
- ^ US patent 2694068, Payne, G. B. et al., "Δ6-DESOXYMORPHINE COMPOUNDS AND PROCESS OF PRODUCING THE SAME", published 1952-08-05, issued 1954-09-11
- ^ Attention: This template ({{cite doi}}) is deprecated. To cite the publication identified by doi: 10.1007/s10809-008-4009-5 , please use {{cite journal}} (if it was published in a bona fide academic journal, otherwise {{cite report}} with
|doi= 10.1007/s10809-008-4009-5instead. - ^ Casy, A. F.; Parfitt, R. Y. (1986). Opioid Analgesics, Chemistry and Receptors. New York: Plenum Press. pp. 37–38. ISBN 0-306-42130-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Lenz, G. R.; Evans, S. M.; Walters, D. E.; Hopfinger, A. J. (1986). Opiates. Academic Press. p. 63. ISBN 978-0-12-443830-9.
{{cite book}}: CS1 maint: multiple names: authors list (link)
