Naloxegol
 | |
| Clinical data | |
|---|---|
| Trade names | Movantik, Moventig |
| Other names | NKTR-118 |
| AHFS/Drugs.com | movantik |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | ~4.2% |
| Metabolism | Liver (CYP3A) |
| Elimination half-life | 6–11 h |
| Excretion | Feces (68%), urine (16%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C34H53NO11 |
| Molar mass | 651.794 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Naloxegol (INN; PEGylated naloxol;[4] trade names Movantik and Moventig) is a peripherally acting μ-opioid receptor antagonist developed by AstraZeneca, licensed from Nektar Therapeutics, for the treatment of opioid-induced constipation.[5] It was approved in 2014 in adult patients with chronic, non-cancer pain.[6] Doses of 25 mg were found safe and well tolerated for 52 weeks.[7] When given concomitantly with opioid analgesics, naloxegol reduced constipation-related side effects, while maintaining comparable levels of analgesia.[8]
The most common side effects are abdominal pain, diarrhea, nausea, flatulence, vomiting, and headache.[9]
Naloxegol was previously a Schedule II drug in the United States because of its chemical similarity to opium alkaloids. It was officially decontrolled in January 2015. It was reclassified as a prescription drug after the FDA and DEA concluded that the impermeability of the blood–brain barrier to this compound made it non-habit-forming, and so without the potential for abuse.[10]
Medical use
[edit]Naloxegol is indicated for the treatment of opioid-induced constipation (OIC) in people with chronic non-cancer pain.[9][11]
Side effects
[edit]The most common side effects are abdominal pain, diarrhea, nausea, flatulence, vomiting, and headache.[9]
Pharmacodynamic properties
[edit]Naloxegol inhibits opioid binding in μ-opioid receptors in the gastrointestinal tract, thus decreasing the constipating effects (slowing of gastrointestinal motility and transit, hypertonicity, increased fluid reabsorption) associated with opioids.[12]
If naloxegol is coadministered with other opioid antagonists, there is a potential for additive effect and increased risk of opioid withdrawal.[9]
Mechanism of action
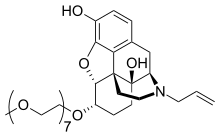
[edit]Chemically, naloxegol is a pegylated (polyethylene glycol-modified) derivative of α-naloxol. Specifically, the 6-α-hydroxyl group of α-naloxol is connected via an ether linkage to the free hydroxyl group of a monomethoxy-terminated n=7 oligomer of PEG, shown extending at the lower left of the molecule image at right. The "n=7" defines the number of two-carbon ethylenes, and so the chain length, of the attached PEG chain, and the "monomethoxy" indicates that the terminal hydroxyl group of the PEG is "capped" with a methyl group.[13] The pegylation of the 6-α-hydroxyl side chain of naloxol prevents the drug from crossing the blood–brain barrier (BBB).[8]
References
[edit]- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Moventig EPAR". European Medicines Agency (EMA). 8 December 2014. Retrieved 28 September 2024.
- ^ Seifert R, Wieland T, Mannhold R, Kubinyi H, Folkers G (17 July 2006). G Protein-Coupled Receptors as Drug Targets: Analysis of Activation and Constitutive Activity. John Wiley & Sons. p. 227. ISBN 978-3-527-60695-5. Retrieved 14 May 2012.
- ^ "Nektar | R&D Pipeline | Products in Development | CNS/Pain | Oral Naloxegol (NKTR-118) and Oral NKTR-119". Archived from the original on 13 February 2012. Retrieved 14 May 2012.
- ^ "FDA approves MOVANTIK™ (naloxegol) Tablets C-II for the treatment of opioid-induced constipation in adult patients with chronic non-cancer pain". 16 September 2014. Archived from the original on 10 May 2015.
- ^ Webster L, Chey WD, Tack J, Lappalainen J, Diva U, Sostek M (October 2014). "Randomised clinical trial: the long-term safety and tolerability of naloxegol in patients with pain and opioid-induced constipation" (PDF). Alimentary Pharmacology & Therapeutics. 40 (7): 771–9. doi:10.1111/apt.12899. PMID 25112584. S2CID 34286557.
- ^ a b Garnock-Jones KP (March 2015). "Naloxegol: a review of its use in patients with opioid-induced constipation". Drugs. 75 (4): 419–25. doi:10.1007/s40265-015-0357-2. PMID 25666542. S2CID 207488539.
- ^ a b c d "Movantik prescribing information highlights" (PDF). Retrieved 14 August 2019.
- ^ "Schedules of Controlled Substances: Removal of Naloxegol From Control". www.deadiversion.usdoj.gov. Archived from the original on 9 March 2016. Retrieved 27 February 2016.
- ^ "Naloxegol for Opioid-Induced Constipation in Patients with Noncancer Pain" (PDF).
- ^ Garnock-Jones KP (March 2015). "Naloxegol: a review of its use in patients with opioid-induced constipation". Drugs. 75 (4): 419–25. doi:10.1007/s40265-015-0357-2. PMID 25666542. S2CID 207488539.
- ^ Technically, the molecule that is attached via the ether link is O-methyl-heptaethylene glycol [that is, methoxyheptaethylene glycol, CH3OCH2CH2O(CH2CH2O)5CH2CH2OH], molecular weight 340.4, CAS number 4437-01-8. See "Compound Summary for CID 526555, Pubchem Compound 4437-01". PubChem Compound Database. Bethesda, MD: NCBI, U.S. NLM. 2016. Archived from the original on 5 February 2016. Retrieved 28 January 2016.
