RB-120
 | |
| Identifiers | |
|---|---|
| |
| PubChem CID | |
| Chemical and physical data | |
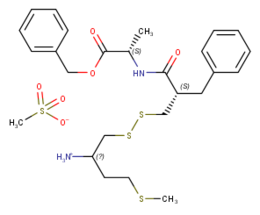
| Formula | C25H34N2O3S3 |
| Molar mass | 506.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
RB-120 (benzyl (2S)-2-{[(2S)-2-({[(2S)-2-amino-4-methylsulfanylbutyl]disulfanyl}methyl)-3-phenylpropanoyl]amino}propanoate) is an orally active analog of the drug RB-101.[1] It acts as an enkephalinase inhibitor, which is used in scientific research. Via intravenous administration, it is approximately three times as potent as RB-101 or twice as potent as the isolated (S,S) isomer of RB101. However, via i.p. administration it is approximately twice as potent as racemic RB-101 and about as potent as the isolated (S,S) isomer of RB101. During i.v. administration RB120 is approximately twice as weak as morphine in terms of analgesia, however it is 16x weaker during i.p. and p.o. administration.[1]
References
- ^ a b Noble, Florence; Smadja, Claire; Valverde, Olga; Maldonado, Rafaël; Coric, Pascale; Turcaud, Serge; Fournié-Zaluski, Marie-Claude; Roques, Bernard P. (1997). "Pain-suppressive effects on various nociceptive stimuli (thermal, chemical, electrical and inflammatory) of the first orally active enkephalin-metabolizing enzyme inhibitor RB 120". Pain. 73 (3): 383–391. doi:10.1016/S0304-3959(97)00125-5. PMID 9469529.
