Abiogenesis

In biology, abiogenesis (from Ancient Greek ἀ- (a-) 'not', βῐ́ος (bíos) 'life', and γένεσις (génesis) 'origin'), or the origin of life, is the natural process by which life has arisen from non-living matter, such as simple organic compounds. The prevailing scientific hypothesis is that the transition from non-living to living entities on Earth was not a single event, but a process of increasing complexity involving the formation of a habitable planet, the prebiotic synthesis of organic molecules, molecular self-replication, self-assembly, autocatalysis, and the emergence of cell membranes. The transition from non-life to life has never been observed experimentally, but many proposals have been made for different stages of the process.
The study of abiogenesis aims to determine how pre-life chemical reactions gave rise to life under conditions strikingly different from those on Earth today. It primarily uses tools from biology and chemistry, with more recent approaches attempting a synthesis of many sciences. Life functions through the specialized chemistry of carbon and water, and builds largely upon four key families of chemicals: lipids for cell membranes, carbohydrates such as sugars, amino acids for protein metabolism, and nucleic acid DNA and RNA for the mechanisms of heredity. Any successful theory of abiogenesis must explain the origins and interactions of these classes of molecules.
Many approaches to abiogenesis investigate how self-replicating molecules, or their components, came into existence. Researchers generally think that current life descends from an RNA world, although other self-replicating and self-catalyzing molecules may have preceded RNA. Other approaches ("metabolism-first" hypotheses) focus on understanding how catalysis in chemical systems on the early Earth might have provided the precursor molecules necessary for self-replication. The classic 1952 Miller–Urey experiment demonstrated that most amino acids, the chemical constituents of proteins, can be synthesized from inorganic compounds under conditions intended to replicate those of the early Earth. External sources of energy may have triggered these reactions, including lightning, radiation, atmospheric entries of micro-meteorites and implosion of bubbles in sea and ocean waves.
While the last universal common ancestor (LUCA) is thought to have been quite different from the origin of life, investigations into LUCA can guide research into early universal characteristics. A genomics approach has sought to characterise LUCA of modern organisms by identifying the genes shared by Archaea and Bacteria, members of the two major branches of life (with Eukaryotes included in the archaean branch in the two-domain system). 355 genes appear to be common to all life; their functions imply that the LUCA was anaerobic with the Wood–Ljungdahl pathway, deriving energy by chemiosmosis, and maintaining its hereditary material with DNA, the genetic code, and ribosomes. Although the LUCA lived over 4 billion years ago (4 Gya), researchers believe it was far from the first form of life. Earlier cells might have had a leaky membrane and been powered by a naturally occurring proton gradient near a deep-sea white smoker hydrothermal vent.
Earth remains the only place in the universe known to harbor life. Geochemical and fossil evidence from the Earth informs most studies of abiogenesis. The Earth was formed at 4.54 Gya, and the earliest evidence of life on Earth dates from at least 3.8 Gya from Western Australia. Fossil micro-organisms appear to have lived within hydrothermal vent precipitates dated 3.77 to 4.28 Gya from Quebec, soon after ocean formation 4.4 Gya during the Hadean.
Overview[edit]
Life consists of reproduction with (heritable) variations.[3] NASA defines life as "a self-sustaining chemical system capable of Darwinian [i.e., biological] evolution."[4] Such a system is complex; the last universal common ancestor (LUCA), presumably a single-celled organism which lived some 4 billion years ago, already had hundreds of genes encoded in the DNA genetic code that is universal today. That in turn implies a suite of cellular machinery including messenger RNA, transfer RNA, and ribosomes to translate the code into proteins. Those proteins included enzymes to operate its anaerobic respiration via the Wood–Ljungdahl metabolic pathway, and a DNA polymerase to replicate its genetic material.[5][6]
The challenge for abiogenesis (origin of life)[7][8][9] researchers is to explain how such a complex and tightly interlinked system could develop by evolutionary steps, as at first sight all its parts are necessary to enable it to function. For example, a cell, whether the LUCA or in a modern organism, copies its DNA with the DNA polymerase enzyme, which is in turn produced by translating the DNA polymerase gene in the DNA. Neither the enzyme nor the DNA can be produced without the other.[10] The evolutionary process could have involved molecular self-replication, self-assembly such as of cell membranes, and autocatalysis.[5][6][11] Nonetheless, the transition of non-life to life has never been observed experimentally.[12]
The precursors to the development of a living cell like the LUCA are clear enough, if disputed in their details: a habitable world is formed with a supply of minerals and liquid water. Prebiotic synthesis creates a range of simple organic compounds, which are assembled into polymers such as proteins and RNA. The process after the LUCA, too, is readily understood: biological evolution caused the development of a wide range of species with varied forms and biochemical capabilities. The derivation of living things such as the LUCA from simple components, however, is far from understood.[1]
Although Earth remains the only place where life is known,[13][14] the science of astrobiology seeks evidence of life on other planets. The 2015 NASA strategy on the origin of life aimed to solve the puzzle by identifying interactions, intermediary structures and functions, energy sources, and environmental factors that contributed to the diversity, selection, and replication of evolvable macromolecular systems,[2] and mapping the chemical landscape of potential primordial informational polymers. The advent of polymers that could replicate, store genetic information, and exhibit properties subject to selection was, it suggested, most likely a critical step in the emergence of prebiotic chemical evolution.[2] Those polymers derived, in turn, from simple organic compounds such as nucleobases, amino acids, and sugars that could have been formed by reactions in the environment.[15][8][16][17] A successful theory of the origin of life must explain how all these chemicals came into being.[18]
Pre-1960s conceptual history[edit]

Spontaneous generation[edit]
One ancient view of the origin of life, from Aristotle until the 19th century, is of spontaneous generation.[19] This theory held that "lower" animals were generated by decaying organic substances, and that life arose by chance.[20][21] This was questioned from the 17th century, in works like Thomas Browne's Pseudodoxia Epidemica.[22][23] In 1665, Robert Hooke published the first drawings of a microorganism. In 1676, Antonie van Leeuwenhoek drew and described microorganisms, probably protozoa and bacteria.[24] Van Leeuwenhoek disagreed with spontaneous generation, and by the 1680s convinced himself, using experiments ranging from sealed and open meat incubation and the close study of insect reproduction, that the theory was incorrect.[25] In 1668 Francesco Redi showed that no maggots appeared in meat when flies were prevented from laying eggs.[26] By the middle of the 19th century, spontaneous generation was considered disproven.[27][28]
Panspermia[edit]
Another ancient idea dating back to Anaxagoras in the 5th century BC is panspermia,[29] the idea that life exists throughout the universe, distributed by meteoroids, asteroids, comets[30] and planetoids.[31] It does not attempt to explain how life originated in itself, but shifts the origin of life on Earth to another heavenly body. The advantage is that life is not required to have formed on each planet it occurs on, but rather in a more limited set of locations (potentially even a single location), and then spread about the galaxy to other star systems via cometary or meteorite impact.[32]
"A warm little pond": primordial soup[edit]
The idea that life originated from non-living matter in slow stages appeared in Herbert Spencer's 1864–1867 book Principles of Biology, and in William Turner Thiselton-Dyer's 1879 paper "On spontaneous generation and evolution". On 1 February 1871 Charles Darwin wrote about these publications to Joseph Hooker, and set out his own speculation, suggesting that the original spark of life may have begun in a "warm little pond, with all sorts of ammonia and phosphoric salts, light, heat, electricity, &c., present, that a proteine compound was chemically formed ready to undergo still more complex changes." Darwin went on to explain that "at the present day such matter would be instantly devoured or absorbed, which would not have been the case before living creatures were formed."[33][34][35]
Alexander Oparin in 1924 and J. B. S. Haldane in 1929 proposed that the first molecules constituting the earliest cells slowly self-organized from a primordial soup, and this theory is called the Oparin–Haldane hypothesis.[36][37] Haldane suggested that the Earth's prebiotic oceans consisted of a "hot dilute soup" in which organic compounds could have formed.[21][38] J. D. Bernal showed that such mechanisms could form most of the necessary molecules for life from inorganic precursors.[39] In 1967, he suggested three "stages": the origin of biological monomers; the origin of biological polymers; and the evolution from molecules to cells.[40][41]
Miller–Urey experiment[edit]
In 1952, Stanley Miller and Harold Urey carried out a chemical experiment to demonstrate how organic molecules could have formed spontaneously from inorganic precursors under prebiotic conditions like those posited by the Oparin–Haldane hypothesis. It used a highly reducing (lacking oxygen) mixture of gases—methane, ammonia, and hydrogen, as well as water vapor—to form simple organic monomers such as amino acids.[42][43] Bernal said of the Miller–Urey experiment that "it is not enough to explain the formation of such molecules, what is necessary, is a physical-chemical explanation of the origins of these molecules that suggests the presence of suitable sources and sinks for free energy."[44] However, current scientific consensus describes the primitive atmosphere as weakly reducing or neutral,[45][46] diminishing the amount and variety of amino acids that could be produced. The addition of iron and carbonate minerals, present in early oceans, however, produces a diverse array of amino acids.[45] Later work has focused on two other potential reducing environments: outer space and deep-sea hydrothermal vents.[47][48][49]
Producing a habitable Earth[edit]
Origin of life timeline | ||||||||||||||||||||||||
−13 — – −12 — – −11 — – −10 — – −9 — – −8 — – −7 — – −6 — – −5 — – −4 — – −3 — – −2 — – −1 — – 0 — |
| |||||||||||||||||||||||
Evolutionary history[edit]
Early universe with first stars[edit]
Soon after the Big Bang, which occurred roughly 14 Gya, the only chemical elements present in the universe were hydrogen, helium, and lithium, the three lightest atoms in the periodic table. These elements gradually accreted and began orbiting in disks of gas and dust. Gravitational accretion of material at the hot and dense centers of these protoplanetary disks formed stars by the fusion of hydrogen.[50] Early stars were massive and short-lived, producing all the heavier elements through stellar nucleosynthesis. Element formation through stellar nucleosynthesis proceeds to its most stable element Iron-56. Heavier elements were formed during supernovae at the end of a stars lifecycle. Carbon, currently the fourth most abundant chemical element in the universe (after hydrogen, helium, and oxygen), was formed mainly in white dwarf stars, particularly those bigger than twice the mass of the sun.[51] As these stars reached the end of their lifecycles, they ejected these heavier elements, among them carbon and oxygen, throughout the universe. These heavier elements allowed for the formation of new objects, including rocky planets and other bodies.[52] According to the nebular hypothesis, the formation and evolution of the Solar System began 4.6 Gya with the gravitational collapse of a small part of a giant molecular cloud. Most of the collapsing mass collected in the center, forming the Sun, while the rest flattened into a protoplanetary disk out of which the planets, moons, asteroids, and other small Solar System bodies formed.[53]
Emergence of Earth[edit]
The age of the Earth is 4.54 Gya as found by radiometric dating of calcium-aluminium-rich inclusions in carbonaceous chrondrite meteorites, the oldest material in the Solar System.[54][55] The Hadean Earth (from its formation until 4 Gya) was at first inhospitable to any living organisms. During its formation, the Earth lost a significant part of its initial mass, and consequentially lacked the gravity to hold molecular hydrogen and the bulk of the original inert gases.[56] Soon after initial accretion of Earth at 4.48 Ga, its collision with Theia, a hypothesised impactor, is thought to have created the ejected debris that would eventually form the Moon.[57] This impact would have removed the Earth's primary atmosphere, leaving behind clouds of viscous silicates and carbon dioxide. This unstable atmosphere was short-lived and condensed shortly after to form the bulk silicate Earth, leaving behind an atmosphere largely consisting of water vapor, nitrogen, and carbon dioxide, with smaller amounts of carbon monoxide, hydrogen, and sulfur compounds.[58][59] The solution of carbon dioxide in water is thought to have made the seas slightly acidic, with a pH of about 5.5.[60]
Condensation to form liquid oceans is theorised to have occurred as early as the Moon-forming impact.[61][62] This scenario has found support from the dating of 4.404 Gya zircon crystals with high δ18O values from metamorphosed quartzite of Mount Narryer in Western Australia.[63][64] The Hadean atmosphere has been characterized as a "gigantic, productive outdoor chemical laboratory," similar to volcanic gases today which still support some abiotic chemistry. Despite the likely increased volcanism from early plate tectonics, the Earth may have been a predominantly water world between 4.4 and 4.3 Gya. It is debated whether or not crust was exposed above this ocean due to uncertainties of what early plate tectonics looked like. For early life to have developed, it is generally thought that a land setting is required, so this question is essential to determining when in Earth's history life evolved.[65] The post-Moon-forming impact Earth likely existed with little if any continental crust, a turbulent atmosphere, and a hydrosphere subject to intense ultraviolet light from a T Tauri stage Sun, from cosmic radiation, and from continued asteroid and comet impacts.[66] Despite all this, niche environments likely existed conducive to life on Earth in the Late-Hadean to Early-Archaean.
The Late Heavy Bombardment hypothesis posits that a period of intense impact occurred at ~3.9 Gya during the Hadean.[67][68] A cataclysmic impact event would have had the potential to sterilise all life on Earth by volatilising liquid oceans and blocking the Sun needed for photosynthesising primary producers, pushing back the earliest possible emergence of life to after Late Heavy Bombardment.[69] Recent research questions both the intensity of the Late Heavy Bombardment as well as its potential for sterilisation. Uncertainties as to whether Late Heavy Bombardment was one giant impact or a period of greater impact rates greatly changed the implication of its destructive power.[70][71] The 3.9 Ga date arises from dating of Apollo mission sample returns collected mostly near the Imbrium Basin, biasing the age of recorded impacts.[72] Impact modelling of the lunar surface reveals that rather than a cataclysmic event at 3.9 Ga, multiple small-scale, short-lived periods of bombardment likely occurred.[73] Terrestrial data backs this idea by showing multiple periods of ejecta in the rock record both before and after the 3.9 Ga marker, suggesting that the early Earth was subject to continuous impacts that would not have had as great an impact on extinction as previously thought.[74] If the Late Heavy Bombardment did not take place, this allows for the emergence of life to have taken place far before 3.9 Ga.
If life evolved in the ocean at depths of more than ten meters, it would have been shielded both from late impacts and the then high levels of ultraviolet radiation from the sun. Geothermically heated oceanic crust could have yielded far more organic compounds through deep hydrothermal vents than the Miller–Urey experiments indicated.[75] The available energy is maximized at 100–150 °C, the temperatures at which hyperthermophilic bacteria and thermoacidophilic archaea live.[76]
Earliest evidence of life[edit]
The exact timing at which life emerged on Earth is unknown. Minimum age estimates are based on evidence from the geologic rock record. The earliest physical evidence of life so far found consists of microbialites in the Nuvvuagittuq Greenstone Belt of Northern Quebec, in banded iron formation rocks at least 3.77 and possibly as old as 4.32 Gya. The micro-organisms lived within hydrothermal vent precipitates, soon after the 4.4 Gya formation of oceans during the Hadean. The microbes resembled modern hydrothermal vent bacteria, supporting the view that abiogenesis began in such an environment.[77]
Biogenic graphite has been found in 3.7 Gya metasedimentary rocks from southwestern Greenland[78] and in microbial mat fossils from 3.49 Gya cherts in the Pilbara region of Western Australia.[79] Evidence of early life in rocks from Akilia Island, near the Isua supracrustal belt in southwestern Greenland, dating to 3.7 Gya, have shown biogenic carbon isotopes.[80] In other parts of the Isua supracrustal belt, graphite inclusions trapped within garnet crystals are connected to the other elements of life: oxygen, nitrogen, and possibly phosphorus in the form of phosphate, providing further evidence for life 3.7 Gya.[81] In the Pilbara region of Western Australia, compelling evidence of early life was found in pyrite-bearing sandstone in a fossilized beach, with rounded tubular cells that oxidized sulfur by photosynthesis in the absence of oxygen.[82][83] Carbon isotope ratios on graphite inclusions from the Jack Hills zircons suggest that life could have existed on Earth from 4.1 Gya.[84]
The Pilbara region of Western Australia contains the Dresser Formation with rocks 3.48 Gya, including layered structures called stromatolites. Their modern counterparts are created by photosynthetic micro-organisms including cyanobacteria.[85] These lie within undeformed hydrothermal-sedimentary strata; their texture indicates a biogenic origin. Parts of the Dresser formation preserve hot springs on land, but other regions seem to have been shallow seas.[86] A molecular clock analysis suggests the LUCA emerged prior to the Late Heavy Bombardment (3.9 Gya).[87]
-
Stromatolites in the Siyeh Formation, Glacier National Park, dated 3.5 Gya, placing them among the earliest life-forms
-
Modern stromatolites in Shark Bay, created by photosynthetic cyanobacteria
Producing molecules: prebiotic synthesis[edit]
All chemical elements except for hydrogen and helium derive from stellar nucleosynthesis. The basic chemical ingredients of life – the carbon-hydrogen molecule (CH), the carbon-hydrogen positive ion (CH+) and the carbon ion (C+) – were produced by ultraviolet light from stars.[88] Complex molecules, including organic molecules, form naturally both in space and on planets.[89] Organic molecules on the early Earth could have had either terrestrial origins, with organic molecule synthesis driven by impact shocks or by other energy sources, such as ultraviolet light, redox coupling, or electrical discharges; or extraterrestrial origins (pseudo-panspermia), with organic molecules formed in interstellar dust clouds raining down on to the planet.[90][91]
Observed extraterrestrial organic molecules[edit]
An organic compound is a chemical whose molecules contain carbon. Carbon is abundant in the Sun, stars, comets, and in the atmospheres of most planets.[92] Organic compounds are relatively common in space, formed by "factories of complex molecular synthesis" which occur in molecular clouds and circumstellar envelopes, and chemically evolve after reactions are initiated mostly by ionizing radiation.[89][93][94] Purine and pyrimidine nucleobases including guanine, adenine, cytosine, uracil, and thymine have been found in meteorites. These could have provided the materials for DNA and RNA to form on the early Earth.[95] The amino acid glycine was found in material ejected from comet Wild 2; it had earlier been detected in meteorites.[96] Comets are encrusted with dark material, thought to be a tar-like organic substance formed from simple carbon compounds under ionizing radiation. A rain of material from comets could have brought such complex organic molecules to Earth.[97][98][59] It is estimated that during the Late Heavy Bombardment, meteorites may have delivered up to five million tons of organic prebiotic elements to Earth per year.[59]
PAH world hypothesis[edit]

Green areas show regions where radiation from hot stars collided with large molecules and small dust grains called "polycyclic aromatic hydrocarbons" (PAHs), causing them to fluoresce. Spitzer Space Telescope, 2018.
Polycyclic aromatic hydrocarbons (PAH) are the most common and abundant polyatomic molecules in the observable universe, and are a major store of carbon.[92][99][100][101] They seem to have formed shortly after the Big Bang,[102][100][101] and are associated with new stars and exoplanets.[92] They are a likely constituent of Earth's primordial sea.[102][100][101] PAHs have been detected in nebulae,[103] and in the interstellar medium, in comets, and in meteorites.[92]
The PAH world hypothesis posits PAHs as precursors to the RNA world.[104] A star, HH 46-IR, resembling the sun early in its life, is surrounded by a disk of material which contains molecules including cyanide compounds, hydrocarbons, and carbon monoxide. PAHs in the interstellar medium can be transformed through hydrogenation, oxygenation, and hydroxylation to more complex organic compounds used in living cells.[105]
Nucleobases and Nucleotides[edit]
The majority of organic compounds introduced on Earth by interstellar dust particles have helped to form complex molecules, thanks to their peculiar surface-catalytic activities.[106][107] Studies of the 12C/13C isotopic ratios of organic compounds in the Murchison meteorite suggest that the RNA component uracil and related molecules, including xanthine, were formed extraterrestrially.[108] NASA studies of meteorites suggest that all four DNA nucleobases (adenine, guanine and related organic molecules) have been formed in outer space.[106][109][110] The cosmic dust permeating the universe contains complex organics ("amorphous organic solids with a mixed aromatic–aliphatic structure") that could be created rapidly by stars.[111] Glycolaldehyde, a sugar molecule and RNA precursor, has been detected in regions of space including around protostars and on meteorites.[112][113]
Laboratory synthesis[edit]
As early as the 1860s, experiments demonstrated that biologically relevant molecules can be produced from interaction of simple carbon sources with abundant inorganic catalysts. The spontaneous formation of complex polymers from abiotically generated monomers under the conditions posited by the "soup" theory is not straightforward. Besides the necessary basic organic monomers, compounds that would have prohibited the formation of polymers were also formed in high concentration during the Miller–Urey and Joan Oró experiments.[114] Biology uses essentially 20 amino acids for its coded protein enzymes, representing a very small subset of the structurally possible products. Since life tends to use whatever is available, an explanation is needed for why the set used is so small.[115] Formamide is attractive as a medium that potentially provided a source of amino acid derivatives from simple aldehyde and nitrile feedstocks.[116]
Sugars[edit]

Alexander Butlerov showed in 1861 that the formose reaction created sugars including tetroses, pentoses, and hexoses when formaldehyde is heated under basic conditions with divalent metal ions like calcium. R. Breslow proposed that the reaction was autocatalytic in 1959.[117]
Nucleobases[edit]
Nucleobases, such as guanine and adenine, can be synthesized from simple carbon and nitrogen sources, such as hydrogen cyanide (HCN) and ammonia.[118] Formamide produces all four ribonucleotides when warmed with terrestrial minerals. Formamide is ubiquitous in the Universe, produced by the reaction of water and HCN. It can be concentrated by the evaporation of water.[119][120] HCN is poisonous only to aerobic organisms (eukaryotes and aerobic bacteria), which did not yet exist. It can play roles in other chemical processes such as the synthesis of the amino acid glycine.[59]
DNA and RNA components including uracil, cytosine and thymine can be synthesized under outer space conditions, using starting chemicals such as pyrimidine found in meteorites. Pyrimidine may have been formed in red giant stars or in interstellar dust and gas clouds.[121] All four RNA-bases may be synthesized from formamide in high-energy density events like extraterrestrial impacts.[122]
Other pathways for synthesizing bases from inorganic materials have been reported.[123] Freezing temperatures are advantageous for the synthesis of purines, due to the concentrating effect for key precursors such as hydrogen cyanide.[124] However, while adenine and guanine require freezing conditions for synthesis, cytosine and uracil may require boiling temperatures.[125] Seven amino acids and eleven types of nucleobases formed in ice when ammonia and cyanide were left in a freezer for 25 years.[126][127] S-triazines (alternative nucleobases), pyrimidines including cytosine and uracil, and adenine can be synthesized by subjecting a urea solution to freeze-thaw cycles under a reductive atmosphere, with spark discharges as an energy source.[128] The explanation given for the unusual speed of these reactions at such a low temperature is eutectic freezing, which crowds impurities in microscopic pockets of liquid within the ice, causing the molecules to collide more often.[129]
Peptides[edit]
Prebiotic peptide synthesis is proposed to have occurred through a number of possible routes. Some center on high temperature/concentration conditions in which condensation becomes energetically favorable, while others focus on the availability of plausible prebiotic condensing agents.[130][further explanation needed]
Experimental evidence for the formation of peptides in uniquely concentrated environments is bolstered by work suggesting that wet-dry cycles and the presence of specific salts can greatly increase spontaneous condensation of glycine into poly-glycine chains.[131] Other work suggests that while mineral surfaces, such as those of pyrite, calcite, and rutile catalyze peptide condensation, they also catalyze their hydrolysis. The authors suggest that additional chemical activation or coupling would be necessary to produce peptides at sufficient concentrations. Thus, mineral surface catalysis, while important, is not sufficient alone for peptide synthesis.[132]
Many prebiotically plausible condensing/activating agents have been identified, including the following: cyanamide, dicyanamide, dicyandiamide, diaminomaleonitrile, urea, trimetaphosphate, NaCl, CuCl2, (Ni,Fe)S, CO, carbonyl sulfide (COS), carbon disulfide (CS2), SO2, and diammonium phosphate (DAP).[130]
More recently, a 2024 article used a substrate with a web of thin cracks under a heat flow, similar to the environment of deep-ocean vents, as a mechanism to separate and concentrate prebiotically relevant building blocks from a dilute mixture, purifying their concentration by up to three orders of magnitude. The authors propose this as a plausible model for the origin of complex biopolymers.[133] This presents another physical process that allows for concentrated peptide precursors to combine in the right conditions. A similar role of increasing amino acid concentration has been suggested for clays as well.[134]
Producing suitable vesicles[edit]

The largest unanswered question in evolution is how simple protocells first arose and differed in reproductive contribution to the following generation, thus initiating the evolution of life. The lipid world theory postulates that the first self-replicating object was lipid-like.[135][136] Phospholipids form lipid bilayers in water while under agitation—the same structure as in cell membranes. These molecules were not present on early Earth, but other amphiphilic long-chain molecules also form membranes. These bodies may expand by insertion of additional lipids, and may spontaneously split into two offspring of similar size and composition. Lipid bodies may have provided sheltering envelopes for information storage, allowing the evolution and preservation of polymers like RNA that store information. Only one or two types of amphiphiles have been studied which might have led to the development of vesicles.[137] There is an enormous number of possible arrangements of lipid bilayer membranes, and those with the best reproductive characteristics would have converged toward a hypercycle reaction,[138][139] a positive feedback composed of two mutual catalysts represented by a membrane site and a specific compound trapped in the vesicle. Such site/compound pairs are transmissible to the daughter vesicles leading to the emergence of distinct lineages of vesicles, which would have allowed natural selection.[140]
A protocell is a self-organized, self-ordered, spherical collection of lipids proposed as a stepping-stone to the origin of life.[137] A functional protocell has (as of 2014) not yet been achieved in a laboratory setting.[141][142][143] Self-assembled vesicles are essential components of primitive cells.[137] The theory of classical irreversible thermodynamics treats self-assembly under a generalized chemical potential within the framework of dissipative systems.[144][145][146] The second law of thermodynamics requires that overall entropy increases, yet life is distinguished by its great degree of organization. Therefore, a boundary is needed to separate ordered life processes from chaotic non-living matter.[147]
Irene Chen and Jack W. Szostak suggest that elementary protocells can give rise to cellular behaviors including primitive forms of differential reproduction, competition, and energy storage.[142] Competition for membrane molecules would favor stabilized membranes, suggesting a selective advantage for the evolution of cross-linked fatty acids and even the phospholipids of today.[142] Such micro-encapsulation would allow for metabolism within the membrane and the exchange of small molecules, while retaining large biomolecules inside. Such a membrane is needed for a cell to create its own electrochemical gradient to store energy by pumping ions across the membrane.[148][149] Fatty acid vesicles in conditions relevant to alkaline hydrothermal vents can be stabilized by isoprenoids which are synthesized by the formose reaction; the advantages and disadvantages of isoprenoids incorporated within the lipid bilayer in different microenvironments might have led to the divergence of the membranes of archaea and bacteria.[150]
Laboratory experiments have shown that vesicles can undergo an evolutionary process under pressure cycling conditions.[151] Simulating the systemic environment in tectonic fault zones within the Earth's crust, pressure cycling leads to the periodic formation of vesicles.[152] Under the same conditions, random peptide chains are being formed, which are being continuously selected for their ability to integrate into the vesicle membrane. A further selection of the vesicles for their stability potentially leads to the development of functional peptide structures,[153][154][155] associated with an increase in the survival rate of the vesicles.
Producing biology[edit]
Energy and entropy[edit]
Life requires a loss of entropy, or disorder, as molecules organize themselves into living matter. At the same time, the emergence of life is associated with the formation of structures beyond a certain threshold of complexity[156]. The emergence of life with increasing order and complexity does not contradict the second law of thermodynamics, which states that overall entropy never decreases, since a living organism creates order in some places (e.g. its living body) at the expense of an increase of entropy elsewhere (e.g. heat and waste production).[157][158][159]
Multiple sources of energy were available for chemical reactions on the early Earth. Heat from geothermal processes is a standard energy source for chemistry. Other examples include sunlight, lightning,[59] atmospheric entries of micro-meteorites,[160] and implosion of bubbles in sea and ocean waves.[161] This has been confirmed by experiments[162][163] and simulations.[164] Unfavorable reactions can be driven by highly favorable ones, as in the case of iron-sulfur chemistry. For example, this was probably important for carbon fixation.[a] Carbon fixation by reaction of CO2 with H2S via iron-sulfur chemistry is favorable, and occurs at neutral pH and 100 °C. Iron-sulfur surfaces, which are abundant near hydrothermal vents, can drive the production of small amounts of amino acids and other biomolecules.[59]
Chemiosmosis[edit]

In 1961, Peter Mitchell proposed chemiosmosis as a cell's primary system of energy conversion. The mechanism, now ubiquitous in living cells, powers energy conversion in micro-organisms and in the mitochondria of eukaryotes, making it a likely candidate for early life.[165][166] Mitochondria produce adenosine triphosphate (ATP), the energy currency of the cell used to drive cellular processes such as chemical syntheses. The mechanism of ATP synthesis involves a closed membrane in which the ATP synthase enzyme is embedded. The energy required to release strongly bound ATP has its origin in protons that move across the membrane.[167] In modern cells, those proton movements are caused by the pumping of ions across the membrane, maintaining an electrochemical gradient. In the first organisms, the gradient could have been provided by the difference in chemical composition between the flow from a hydrothermal vent and the surrounding seawater,[149] or perhaps meteoric quinones that were conducive to the development of chemiosmotic energy across lipid membranes if at a terrestrial origin.[168]

The RNA world[edit]
The RNA world hypothesis describes an early Earth with self-replicating and catalytic RNA but no DNA or proteins.[169] Many researchers concur that an RNA world must have preceded the DNA-based life that now dominates.[170] However, RNA-based life may not have been the first to exist.[171][172] Another model echoes Darwin's "warm little pond" with cycles of wetting and drying.[173]
RNA is central to the translation process. Small RNAs can catalyze all the chemical groups and information transfers required for life.[172][174] RNA both expresses and maintains genetic information in modern organisms; and the chemical components of RNA are easily synthesized under the conditions that approximated the early Earth, which were very different from those that prevail today. The structure of the ribosome has been called the "smoking gun", with a central core of RNA and no amino acid side chains within 18 Å of the active site that catalyzes peptide bond formation.[175][171][176]
The concept of the RNA world was proposed in 1962 by Alexander Rich,[177] and the term was coined by Walter Gilbert in 1986.[172][178] There were initial difficulties in the explanation of the abiotic synthesis of the nucleotides cytosine and uracil.[179] Subsequent research has shown possible routes of synthesis; for example, formamide produces all four ribonucleotides and other biological molecules when warmed in the presence of various terrestrial minerals.[119][120]
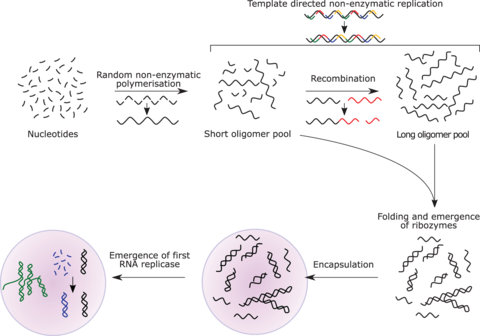
RNA replicase can function as both code and catalyst for further RNA replication, i.e. it can be autocatalytic. Jack Szostak has shown that certain catalytic RNAs can join smaller RNA sequences together, creating the potential for self-replication. The RNA replication systems, which include two ribozymes that catalyze each other's synthesis, showed a doubling time of the product of about one hour, and were subject to natural selection under the experimental conditions.[180][181][171] If such conditions were present on early Earth, then natural selection would favor the proliferation of such autocatalytic sets, to which further functionalities could be added.[182][183][184] Self-assembly of RNA may occur spontaneously in hydrothermal vents.[185][186][187] A preliminary form of tRNA could have assembled into such a replicator molecule.[188]
Possible precursors to protein synthesis include the synthesis of short peptide cofactors or the self-catalysing duplication of RNA. It is likely that the ancestral ribosome was composed entirely of RNA, although some roles have since been taken over by proteins. Major remaining questions on this topic include identifying the selective force for the evolution of the ribosome and determining how the genetic code arose.[189]
Eugene Koonin has argued that "no compelling scenarios currently exist for the origin of replication and translation, the key processes that together comprise the core of biological systems and the apparent pre-requisite of biological evolution. The RNA World concept might offer the best chance for the resolution of this conundrum but so far cannot adequately account for the emergence of an efficient RNA replicase or the translation system."[190]
From RNA to Directed Protein Synthesis[edit]
In line with the RNA world hypothesis, much of modern biology's templated protein biosynthesis is done by RNA molecules—namely tRNAs and the ribosome (consisting of both protein and rRNA components). The most central reaction of peptide bond synthesis is understood to be carried out by base catalysis by the 23S rRNA domain V.[191] Experimental evidence has demonstrated successful di- and tripeptide synthesis with a system consisting of only aminoacyl phosphate adaptors and RNA guides, which could be a possible stepping stone between and RNA world and modern protein synthesis.[191][192] Aminoacylation ribozymes that can charge tRNAs with their cognate amino acids have also been selected in in vitro experimentation.[193] The authors also extensively mapped fitness landscapes within their selection to find that chance emergence of active sequences was more important that sequence optimization.[193]
Early Functional Peptides[edit]
The first proteins would have had to arise without a fully-fledged system of protein biosynthesis. As discussed above, numerous mechanisms for the prebiotic synthesis of polypeptides exist. However, these random sequence peptides would not have likely had biological function. Thus, significant study has gone into exploring how early functional proteins could have arisen from random sequences. First, some evidence on hydrolysis rates shows that abiotically plausible peptides likely contained significant "nearest-neighbor" biases.[194] This could have had some effect on early protein sequence diversity. In other work by Anthony Keefe and Jack Szostak, mRNA display selection on a library of 6*1012 80-mers was used to search for sequences with ATP binding activity. They concluded that approximately 1 in 1011 random sequences had ATP binding function.[195] While this is a single example of functional frequency in the random sequence space, the methodology can serve as a powerful simulation tool for understanding early protein evolution.[196]
Phylogeny and LUCA[edit]
Starting with the work of Carl Woese from 1977, genomics studies have placed the last universal common ancestor (LUCA) of all modern life-forms between Bacteria and a clade formed by Archaea and Eukaryota in the phylogenetic tree of life. It lived over 4 Gya.[197][198] A minority of studies have placed the LUCA in Bacteria, proposing that Archaea and Eukaryota are evolutionarily derived from within Eubacteria;[199] Thomas Cavalier-Smith suggested in 2006 that the phenotypically diverse bacterial phylum Chloroflexota contained the LUCA.[200]
-
Phylogenetic tree showing the last universal common ancestor (LUCA) at the root. The major clades are the Bacteria on one hand, and the Archaea and Eukaryota on the other.
In 2016, a set of 355 genes likely present in the LUCA was identified. A total of 6.1 million prokaryotic genes from Bacteria and Archaea were sequenced, identifying 355 protein clusters from among 286,514 protein clusters that were probably common to the LUCA. The results suggest that the LUCA was anaerobic with a Wood–Ljungdahl (reductive Acetyl-CoA) pathway, nitrogen- and carbon-fixing, thermophilic. Its cofactors suggest dependence upon an environment rich in hydrogen, carbon dioxide, iron, and transition metals. Its genetic material was probably DNA, requiring the 4-nucleotide genetic code, messenger RNA, transfer RNA, and ribosomes to translate the code into proteins such as enzymes. LUCA likely inhabited an anaerobic hydrothermal vent setting in a geochemically active environment. It was evidently already a complex organism, and must have had precursors; it was not the first living thing.[10][201] The physiology of LUCA has been in dispute.[202][203][204]
-
LUCA systems and environment included the Wood–Ljungdahl pathway.[10]
Leslie Orgel argued that early translation machinery for the genetic code would be susceptible to error catastrophe. Geoffrey Hoffmann however showed that such machinery can be stable in function against "Orgel's paradox".[205][206][207] Metabolic reactions that have also been inferred in LUCA are the incomplete reverse Krebs cycle, gluconeogenesis, the pentose phosphate pathway, glycolysis, reductive amination, and transamination.[208][209]
Suitable geological environments[edit]
A variety of geologic and environmental settings have been proposed for an origin of life. These theories are often in competition with one another as there are many differing views of prebiotic compound availability, geophysical setting, and early life characteristics. The first organism on Earth likely looked different from LUCA. Between the first appearance of life and where all modern phylogenies began branching, an unknown amount of time passed, with unknown gene transfers, extinctions, and evolutionary adaptation to various environmental niches.[210] One major shift is believed to be from the RNA world to an RNA-DNA-protein world. Modern phylogenies provide more pertinent genetic evidence about LUCA than about its precursors.[211]
The most popular hypotheses for settings for the origin of life are deep sea hydrothermal vents and surface bodies of water. Surface waters can be classified into hot springs, moderate temperature lakes and ponds, and cold settings.
Deep sea hydrothermal vents[edit]
Hot fluids[edit]

Early micro-fossils may have come from a hot world of gases such as methane, ammonia, carbon dioxide, and hydrogen sulfide, toxic to much current life.[212] Analysis of the tree of life places thermophilic and hyperthermophilic bacteria and archaea closest to the root, suggesting that life may have evolved in a hot environment.[213] The deep sea or alkaline hydrothermal vent theory posits that life began at submarine hydrothermal vents.[214][215] William Martin and Michael Russell have suggested "that life evolved in structured iron monosulphide precipitates in a seepage site hydrothermal mound at a redox, pH, and temperature gradient between sulphide-rich hydrothermal fluid and iron(II)-containing waters of the Hadean ocean floor. The naturally arising, three-dimensional compartmentation observed within fossilized seepage-site metal sulphide precipitates indicates that these inorganic compartments were the precursors of cell walls and membranes found in free-living prokaryotes. The known capability of FeS and NiS to catalyze the synthesis of the acetyl-methylsulphide from carbon monoxide and methylsulphide, constituents of hydrothermal fluid, indicates that pre-biotic syntheses occurred at the inner surfaces of these metal-sulphide-walled compartments".[216]
These form where hydrogen-rich fluids emerge from below the sea floor, as a result of serpentinization of ultra-mafic olivine with seawater and a pH interface with carbon dioxide-rich ocean water. The vents form a sustained chemical energy source derived from redox reactions, in which electron donors (molecular hydrogen) react with electron acceptors (carbon dioxide); see iron–sulfur world theory. These are exothermic reactions.[214][b]
Chemiosmotic gradient[edit]
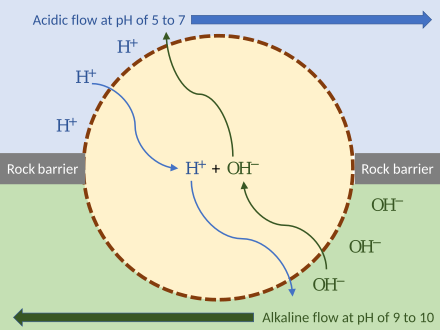
Russell demonstrated that alkaline vents created an abiogenic proton motive force chemiosmotic gradient,[216] ideal for abiogenesis. Their microscopic compartments "provide a natural means of concentrating organic molecules," composed of iron-sulfur minerals such as mackinawite, endowed these mineral cells with the catalytic properties envisaged by Günter Wächtershäuser.[217] This movement of ions across the membrane depends on a combination of two factors:
- Diffusion force caused by concentration gradient—all particles including ions tend to diffuse from higher concentration to lower.
- Electrostatic force caused by electrical potential gradient—cations like protons H+ tend to diffuse down the electrical potential, anions in the opposite direction.
These two gradients taken together can be expressed as an electrochemical gradient, providing energy for abiogenic synthesis. The proton motive force can be described as the measure of the potential energy stored as a combination of proton and voltage gradients across a membrane (differences in proton concentration and electrical potential).[149]
The surfaces of mineral particles inside deep-ocean hydrothermal vents have catalytic properties similar to those of enzymes and can create simple organic molecules, such as methanol (CH3OH) and formic, acetic, and pyruvic acids out of the dissolved CO2 in the water, if driven by an applied voltage or by reaction with H2 or H2S.[218][219]
The research reported by Martin in 2016 supports the thesis that life arose at hydrothermal vents,[220][221] that spontaneous chemistry in the Earth's crust driven by rock–water interactions at disequilibrium thermodynamically underpinned life's origin[222][223] and that the founding lineages of the archaea and bacteria were H2-dependent autotrophs that used CO2 as their terminal acceptor in energy metabolism.[224] Martin suggests, based upon this evidence, that the LUCA "may have depended heavily on the geothermal energy of the vent to survive".[10] Pores at deep sea hydrothermal vents are suggested to have been occupied by membrane-bound compartments which promoted biochemical reactions.[225][226] Metabolic intermediates in the Krebs cycle, gluconeogenesis, amino acid bio-synthetic pathways, glycolysis, the pentose phosphate pathway, and including sugars like ribose, and lipid precursors can occur non-enzymatically at conditions relevant to deep-sea alkaline hydrothermal vents.[227]
If the deep marine hydrothermal setting was the site for the origin of life, then abiogenesis could have happened as early as 4.0-4.2 Gya. If life evolved in the ocean at depths of more than ten meters, it would have been shielded both from impacts and the then high levels of ultraviolet radiation from the sun. The available energy in hydrothermal vents is maximized at 100–150 °C, the temperatures at which hyperthermophilic bacteria and thermoacidophilic archaea live.[228][229] Arguments against a hydrothermal origin of life state that hyperthermophily was a result of convergent evolution in bacteria and archaea, and that a mesophilic environment would have been more likely.[230][231] This hypothesis, suggested in 1999 by Galtier, was proposed one year before the discovery of the Lost City Hydrothermal Field, where white-smoker hydrothermal vents average ~45-90 °C.[232] Moderate temperatures and alkaline seawater at Lost City are now the favoured hydrothermal vent setting in contrast to acidic, high temperature (~350 °C) black-smokers.
Arguments against a vent setting[edit]
Production of prebiotic organic compounds at hydrothermal vents is estimated to be 1x108 kg yr−1.[233] While a large amount of key prebiotic compounds, such as methane, are found at vents, they are in far lower concentrations than estimates of a Miller-Urey Experiment environment. In the case of methane, the production rate at vents is around 2-4 orders of magnitude lower than predicted amounts in a Miller-Urey Experiment surface atmosphere.[233][234]
Other arguments against an oceanic vent setting for the origin of life include the inability to concentrate prebiotic materials due to strong dilution from seawater. This open-system cycles compounds through minerals that make up vents, leaving little residence time to accumulate.[235] All modern cells rely on phosphates and potassium for nucleotide backbone and protein formation respectively, making it likely that the first life forms also shared these functions. These elements were not available in high quantities in the Archaean oceans as both primarily come from the weathering of continental rocks on land, far from vent settings. Submarine hydrothermal vents are not conducive to condensation reactions needed for polymerisation to form macromolecules.[236][237] Further, these polymers were encapsulated in vesicles after condensation, which would not happen in saltwater because of the high concentrations of ions.[238]
Surface bodies of water[edit]
Surface bodies of water provide environments able to dry out and be rewetted. Continued wet-dry cycles allow the concentration of prebiotic compounds and condensation reactions to polymerise macromolecules. Moreover, lake and ponds on land allow for detrital input from the weathering of continental rocks which contain apatite, the most common source of phosphates needed for nucleotide backbones. The amount of exposed continental crust in the Hadean is unknown, but models of early ocean depths and rates of ocean island and continental crust growth make it plausible that there was exposed land.[239] Another line of evidence for a surface start to life is the requirement for UV for organism function. UV is necessary for the formation of the U+C nucleotide base pair by partial hydrolysis and nucleobase loss.[240] Simultaneously, UV can be harmful and sterilising to life, especially for simple early lifeforms with little ability to repair radiation damage. Radiation levels from a young Sun were likely greater, and, with no ozone layer, harmful shortwave UV rays would reach the surface of Earth. For life to begin, a shielded environment with influx from UV-exposed sources is necessary to both benefit and protect from UV. Shielding under ice, liquid water, mineral surfaces (e.g. clay) or regolith is possible in a range of surface water settings. While deep sea vents may have input from raining down of surface exposed materials, the likelihood of concentration is lessened by the ocean's open system.[241]
Hot springs[edit]
Most branching phylogenies are thermophilic or hyperthermophilic, making it possible that the Last universal common ancestor (LUCA) and preceding lifeforms were similarly thermophilic. Hot springs are formed from the heating of groundwater by geothermal activity. This intersection allows for influxes of material from deep penetrating waters and from surface runoff that transports eroded continental sediments. Interconnected groundwater systems create a mechanism for distribution of life to wider area.[242]
Mulkidjanian and co-authors argue that marine environments did not provide the ionic balance and composition universally found in cells, or the ions required by essential proteins and ribozymes, especially with respect to high K+/Na+ ratio, Mn2+, Zn2+ and phosphate concentrations. They argue that the only environments that mimic the needed conditions on Earth are hot springs similar to ones at Kamchatka.[243] Mineral deposits in these environments under an anoxic atmosphere would have suitable pH (while current pools in an oxygenated atmosphere would not), contain precipitates of photocatalytic sulfide minerals that absorb harmful ultraviolet radiation, have wet-dry cycles that concentrate substrate solutions to concentrations amenable to spontaneous formation of biopolymers[244][245] created both by chemical reactions in the hydrothermal environment, and by exposure to UV light during transport from vents to adjacent pools that would promote the formation of biomolecules.[246] The hypothesized pre-biotic environments are similar to hydrothermal vents, with additional components that help explain peculiarities of the LUCA.[243][168]
A phylogenomic and geochemical analysis of proteins plausibly traced to the LUCA shows that the ionic composition of its intracellular fluid is identical to that of hot springs. The LUCA likely was dependent upon synthesized organic matter for its growth.[243] Experiments show that RNA-like polymers can be synthesized in wet-dry cycling and UV light exposure. These polymers were encapsulated in vesicles after condensation.[238] Potential sources of organics at hot springs might have been transport by interplanetary dust particles, extraterrestrial projectiles, or atmospheric or geochemical synthesis. Hot springs could have been abundant in volcanic landmasses during the Hadean.[168]
Temperate surface bodies of water[edit]
A mesophilic start in surface bodies of waters hypothesis has evolved from Darwin's concept of a 'warm little pond' and the Oparin-Haldane hypothesis. Freshwater bodies under temperate climates can accumulate prebiotic materials while providing suitable environmental conditions conducive to simple life forms. The climate during the Archaean is still a highly debated topic, as there is uncertainty about what continents, oceans, and the atmosphere looked like then. Atmospheric reconstructions of the Archaean from geochemical proxies and models state that sufficient greenhouse gases were present to maintain surface temperatures between 0-40 °C. Under this assumption, there is a greater abundance of moderate temperature niches in which life could begin.[247]
Strong lines of evidence for mesophily from biomolecular studies include Galtier's G+C nucleotide thermometer. G+C are more abundant in thermophiles due to the added stability of an additional hydrogen bond not present between A+T nucleotides. rRNA sequencing on a diverse range of modern lifeforms show that LUCA's reconstructed G+C content was likely representative of moderate temperatures.[231]
Although most modern phylogenies are thermophilic or hyperthermophilic, it is possible that their widespread diversity today is a product of convergent evolution and horizontal gene transfer rather than an inherited trait from LUCA.[248] The reverse gyrase topoisomerase is found exclusively in thermophiles and hyperthermophiles as it allows for coiling of DNA.[249] The reverse gyrase enzyme requires ATP to function, both of which are complex biomolecules. If an origin of life is hypothesised to involve a simple organism that had not yet evolved a membrane, let alone ATP, this would make the existence of reverse gyrase improbable. Moreover, phylogenetic studies show that reverse gyrase had an archaeal origin, and that it was transferred to bacteria by horizontal gene transfer. This implies that reverse gyrase was not present in the LUCA.[250]
Icy surface bodies of water[edit]
Cold-start origin of life theories stem from the idea there may have been cold enough regions on the early Earth that large ice cover could be found. Stellar evolution models predict that the Sun's luminosity was ~25% weaker than it is today. Fuelner states that although this significant decrease in solar energy would have formed an icy planet, there is strong evidence for liquid water to be present, possibly driven by a greenhouse effect. This would create an early Earth with both liquid oceans and icy poles.[251]
Ice melts that form from ice sheets or glaciers melts create freshwater pools, another niche capable of experiencing wet-dry cycles. While these pools that exist on the surface would be exposed to intense UV radiation, bodies of water within and under ice are sufficiently shielded while remaining connected to UV exposed areas through ice cracks. Suggestions of impact melting of ice allow freshwater paired with meteoritic input, a popular vessel for prebiotic components.[252] Near-seawater levels of sodium chloride are found to destabilize fatty acid membrane self-assembly, making freshwater settings appealing for early membranous life.[253]
Icy environments would trade the faster reaction rates that occur in warm environments for increased stability and accumulation of larger polymers.[254] Experiments simulating Europa-like conditions of ~20 °C have synthesised amino acids and adenine, showing that Miller-Urey type syntheses can still occur at cold temperatures.[255] In an RNA world, the ribozyme would have had even more functions than in a later DNA-RNA-protein-world. For RNA to function, it must be able to fold, a process that is hindered by temperatures above 30 °C. While RNA folding in psychrophilic organisms is slower, the process is more successful as hydrolysis is also slower. Shorter nucleotides would not suffer from higher temperatures.[256][257]
Inside the continental crust[edit]
An alternative geological environment has been proposed by the geologist Ulrich Schreiber and the physical chemist Christian Mayer: the continental crust.[258] Tectonic fault zones could present a stable and well-protected environment for long-term prebiotic evolution. Inside these systems of cracks and cavities, water and carbon dioxide present the bulk solvents. Their phase state would depend on the local temperature and pressure conditions and could vary between liquid, gaseous and supercritical. When forming two separate phases (e.g., liquid water and supercritical carbon dioxide in depths of little more than 1 km), the system provides optimal conditions for phase transfer reactions. Concurrently, the contents of the tectonic fault zones are being supplied by a multitude of inorganic educts (e.g., carbon monoxide, hydrogen, ammonia, hydrogen cyanide, nitrogen, and even phosphate from dissolved apatite) and simple organic molecules formed by hydrothermal chemistry (e.g. amino acids, long-chain amines, fatty acids, long-chain aldehydes).[259][260] Finally, the abundant mineral surfaces provide a rich choice of catalytic activity.
An especially interesting section of the tectonic fault zones is located at a depth of approximately 1000 m. For the carbon dioxide part of the bulk solvent, it provides temperature and pressure conditions near the phase transition point between the supercritical and the gaseous state. This leads to a natural accumulation zone for lipophilic organic molecules that dissolve well in supercritical CO2, but not in its gaseous state, leading to their local precipitation.[261] Periodic pressure variations such as caused by geyser activity or tidal influences result in periodic phase transitions, keeping the local reaction environment in a constant non-equilibrium state. In presence of amphiphilic compounds (such as the long chain amines and fatty acids mentioned above), subsequent generations of vesicles are being formed[262] that are constantly and efficiently being selected for their stability.[263] The resulting structures could provide hydrothermal vents as well as hot springs with raw material for further development.
Homochirality[edit]

Homochirality is the geometric uniformity of materials composed of chiral (non-mirror-symmetric) units. Living organisms use molecules that have the same chirality (handedness): with almost no exceptions,[265] amino acids are left-handed while nucleotides and sugars are right-handed. Chiral molecules can be synthesized, but in the absence of a chiral source or a chiral catalyst, they are formed in a 50/50 (racemic) mixture of both forms. Known mechanisms for the production of non-racemic mixtures from racemic starting materials include: asymmetric physical laws, such as the electroweak interaction; asymmetric environments, such as those caused by circularly polarized light, quartz crystals, or the Earth's rotation, statistical fluctuations during racemic synthesis,[264] and spontaneous symmetry breaking.[266][267][268]
Once established, chirality would be selected for.[269] A small bias (enantiomeric excess) in the population can be amplified into a large one by asymmetric autocatalysis, such as in the Soai reaction.[270] In asymmetric autocatalysis, the catalyst is a chiral molecule, which means that a chiral molecule is catalyzing its own production. An initial enantiomeric excess, such as can be produced by polarized light, then allows the more abundant enantiomer to outcompete the other.[271]
Homochirality may have started in outer space, as on the Murchison meteorite the amino acid L-alanine (left-handed) is more than twice as frequent as its D (right-handed) form, and L-glutamic acid is more than three times as abundant as its D counterpart.[272][273] Amino acids from meteorites show a left-handed bias, whereas sugars show a predominantly right-handed bias: this is the same preference found in living organisms, suggesting an abiogenic origin of these compounds.[274]
In a 2010 experiment by Robert Root-Bernstein, "two D-RNA-oligonucleotides having inverse base sequences (D-CGUA and D-AUGC) and their corresponding L-RNA-oligonucleotides (L-CGUA and L-AUGC) were synthesized and their affinity determined for Gly and eleven pairs of L- and D-amino acids". The results suggest that homochirality, including codon directionality, might have "emerged as a function of the origin of the genetic code".[275]
See also[edit]
- Autopoiesis
- Formamide-based prebiotic chemistry – Scientific efforts aimed at reconstructing the beginnings of life
- Proto-metabolism
- GADV-protein world hypothesis – A hypothetical stage of abiogenesis
- Genetic recombination – Production of offspring with combinations of traits that differ from those found in either parent
- Shadow biosphere – Hypothetical biosphere of Earth
Notes[edit]
- ^ The reactions are:
- FeS + H2S → FeS2 + 2H+ + 2e−
- FeS + H2S + CO2 → FeS2 + HCOOH
- ^ The reactions are:
Reaction 1: Fayalite + water → magnetite + aqueous silica + hydrogen- 3Fe2SiO4 + 2H2O → 2Fe3O4 + 3SiO2 + 2H2
- 3Mg2SiO4 + SiO2 + 4H2O → 2Mg3Si2O5(OH)4
- 2Mg2SiO4 + 3H2O → Mg3Si2O5(OH)4 + Mg(OH)2
- 2 Ca2SiO4 + 4 H2O → 3 CaO · 2 SiO2 · 3 H2O + Ca(OH)2
References[edit]
- ^ a b Walker, Sara I.; Packard, N.; Cody, G. D. (13 November 2017). "Re-conceptualizing the origins of life". Philosophical Transactions of the Royal Society A. 375 (2109): 20160337. Bibcode:2017RSPTA.37560337W. doi:10.1098/rsta.2016.0337. PMC 5686397. PMID 29133439.
- ^ a b c "NASA Astrobiology Strategy" (PDF). NASA. 2015. Archived from the original (PDF) on 22 December 2016. Retrieved 24 September 2017.
- ^ Trifonov, Edward N. (17 March 2011). "Vocabulary of Definitions of Life Suggests a Definition". Journal of Biomolecular Structure and Dynamics. 29 (2): 259–266. doi:10.1080/073911011010524992. PMID 21875147. S2CID 38476092.
- ^ Voytek, Mary A. (6 March 2021). "About Life Detection". NASA. Archived from the original on 16 August 2021. Retrieved 8 March 2021.
- ^ a b Witzany, Guenther (2016). "Crucial steps to life: From chemical reactions to code using agents" (PDF). BioSystems. 140: 49–57. Bibcode:2016BiSys.140...49W. doi:10.1016/j.biosystems.2015.12.007. PMID 26723230. S2CID 30962295. Archived (PDF) from the original on 31 October 2018. Retrieved 30 October 2018.
- ^ a b Howell, Elizabeth (8 December 2014). "How Did Life Become Complex, And Could It Happen Beyond Earth?". Astrobiology Magazine. Archived from the original on 15 February 2018. Retrieved 14 April 2022.
{{cite web}}: CS1 maint: unfit URL (link) - ^ Oparin, Aleksandr Ivanovich (2003) [1938]. The Origin of Life. Translated by Morgulis, Sergius (2 ed.). Mineola, New York: Courier. ISBN 978-0-486-49522-4. Archived from the original on 2 April 2023. Retrieved 16 June 2018.
- ^ a b Peretó, Juli (2005). "Controversies on the origin of life" (PDF). International Microbiology. 8 (1): 23–31. PMID 15906258. Archived from the original (PDF) on 24 August 2015. Retrieved 1 June 2015.
- ^ Compare: Scharf, Caleb; et al. (18 December 2015). "A Strategy for Origins of Life Research". Astrobiology. 15 (12): 1031–1042. Bibcode:2015AsBio..15.1031S. doi:10.1089/ast.2015.1113. PMC 4683543. PMID 26684503.
What do we mean by the origins of life (OoL)? ... Since the early 20th century the phrase OoL has been used to refer to the events that occurred during the transition from non-living to living systems on Earth, i.e., the origin of terrestrial biology (Oparin, 1924; Haldane, 1929). The term has largely replaced earlier concepts such as abiogenesis (Kamminga, 1980; Fry, 2000).
- ^ a b c d Weiss, M. C.; Sousa, F. L.; Mrnjavac, N.; Neukirchen, S.; Roettger, M.; Nelson-Sathi, S.; Martin, W.F. (2016). "The physiology and habitat of the last universal common ancestor" (PDF). Nature Microbiology. 1 (9): 16116. doi:10.1038/NMICROBIOL.2016.116. PMID 27562259. S2CID 2997255. Archived (PDF) from the original on 29 January 2023. Retrieved 21 September 2022.
- ^ Tirard, Stephane (20 April 2015). "Abiogenesis". Encyclopedia of Astrobiology. p. 1. doi:10.1007/978-3-642-27833-4_2-4. ISBN 978-3-642-27833-4.
Thomas Huxley (1825–1895) used the term abiogenesis in an important text published in 1870. He strictly made the difference between spontaneous generation, which he did not accept, and the possibility of the evolution of matter from inert to living, without any influence of life. ... Since the end of the nineteenth century, evolutive abiogenesis means increasing complexity and evolution of matter from inert to living state in the abiotic context of evolution of primitive Earth.
- ^ Luisi, Pier Luigi (2018). The Emergence of Life: From Chemical Origins to Synthetic Biology. Cambridge University Press. p. 416. ISBN 9781108735506.
However, the turning point of non-life to life has never been put into one experimental set up. There are, of course, several hypotheses, and this plethora of ideas means already that we do not have a convincing one.
- ^ Graham, Robert W. (February 1990). "Extraterrestrial Life in the Universe" (PDF). NASA (NASA Technical Memorandum 102363). Lewis Research Center, Cleveland, Ohio. Archived (PDF) from the original on 3 September 2014. Retrieved 2 June 2015.
- ^ Altermann 2009, p. xvii
- ^ Oparin 1953, p. vi
- ^ Warmflash, David; Warmflash, Benjamin (November 2005). "Did Life Come from Another World?". Scientific American. 293 (5): 64–71. Bibcode:2005SciAm.293e..64W. doi:10.1038/scientificamerican1105-64. PMID 16318028.
- ^ Yarus 2010, p. 47
- ^ Ward, Peter; Kirschvink, Joe (2015). A New History of Life: the radical discoveries about the origins and evolution of life on earth. Bloomsbury Press. pp. 39–40. ISBN 978-1-60819-910-5.
- ^ Sheldon 2005
- ^ Lennox 2001, pp. 229–258
- ^ a b Bernal 1967
- ^ Balme, D. M. (1962). "Development of Biology in Aristotle and Theophrastus: Theory of Spontaneous Generation". Phronesis. 7 (1–2): 91–104. doi:10.1163/156852862X00052.
- ^ Ross 1652
- ^ Dobell 1960
- ^ Bondeson 1999
- ^ Levine, R.; Evers, C. "The Slow Death of Spontaneous Generation (1668-1859)". Archived from the original on 26 April 2008. Retrieved 18 April 2013.
- ^ Oparin 1953, p. 196
- ^ Tyndall 1905, IV, XII (1876), XIII (1878)
- ^ Horneck, Gerda; Klaus, David M.; Mancinelli, Rocco L. (March 2010). "Space Microbiology". Microbiology and Molecular Biology Reviews. 74 (1): 121–156. Bibcode:2010MMBR...74..121H. doi:10.1128/MMBR.00016-09. PMC 2832349. PMID 20197502.
- ^ Wickramasinghe, Chandra (2011). "Bacterial morphologies supporting cometary panspermia: a reappraisal". International Journal of Astrobiology. 10 (1): 25–30. Bibcode:2011IJAsB..10...25W. CiteSeerX 10.1.1.368.4449. doi:10.1017/S1473550410000157. S2CID 7262449.
- ^ Rampelotto, P. H. (2010). "Panspermia: A promising field of research". In: Astrobiology Science Conference. Abs 5224.
- ^ Chang, Kenneth (12 September 2016). "Visions of Life on Mars in Earth's Depths". The New York Times. Archived from the original on 12 September 2016. Retrieved 12 September 2016.
- ^ "Letter no. 7471, Charles Darwin to Joseph Dalton Hooker, 1 February (1871)". Darwin Correspondence Project. Archived from the original on 7 July 2020. Retrieved 7 July 2020.
- ^ Priscu, John C. "Origin and Evolution of Life on a Frozen Earth". Arlington County, Virginia: National Science Foundation. Archived from the original on 18 December 2013. Retrieved 1 March 2014.
- ^ Marshall, Michael (11 November 2020). "Charles Darwin's hunch about early life was probably right". BBC News. Archived from the original on 11 November 2020. Retrieved 11 November 2020.
- ^ Bahadur, Krishna (1973). "Photochemical Formation of Self–sustaining Coacervates" (PDF). Proceedings of the Indian National Science Academy. 39 (4): 455–467. doi:10.1016/S0044-4057(75)80076-1. PMID 1242552. Archived from the original (PDF) on 19 October 2013.
- ^ Bahadur, Krishna (1975). "Photochemical Formation of Self-Sustaining Coacervates". Zentralblatt für Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene (Central Journal for Bacteriology, Parasitology, Infectious Diseases and Hygiene). 130 (3): 211–218. doi:10.1016/S0044-4057(75)80076-1. OCLC 641018092. PMID 1242552. Archived from the original on 13 December 2022. Retrieved 13 December 2022.
- ^ Bryson 2004, pp. 300–302
- ^ Bernal 1951
- ^ Martin, William F. (January 2003). "On the origins of cells: a hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells". Phil. Trans. R. Soc. Lond. A. 358 (1429): 59–83. doi:10.1098/rstb.2002.1183. PMC 1693102. PMID 12594918.
- ^ Bernal, John Desmond (September 1949). "The Physical Basis of Life". Proceedings of the Physical Society, Section A. 62 (9): 537–558. Bibcode:1949PPSA...62..537B. doi:10.1088/0370-1298/62/9/301. S2CID 83754271.
- ^ Miller, Stanley L. (15 May 1953). "A Production of Amino Acids Under Possible Primitive Earth Conditions". Science. 117 (3046): 528–529. Bibcode:1953Sci...117..528M. doi:10.1126/science.117.3046.528. PMID 13056598.
- ^ Parker, Eric T.; Cleaves, Henderson J.; Dworkin, Jason P.; et al. (5 April 2011). "Primordial synthesis of amines and amino acids in a 1958 Miller H2S-rich spark discharge experiment". PNAS. 108 (14): 5526–5531. Bibcode:2011PNAS..108.5526P. doi:10.1073/pnas.1019191108. PMC 3078417. PMID 21422282.
- ^ Bernal 1967, p. 143
- ^ a b Cleaves, H. James; Chalmers, John H.; Lazcano, Antonio; et al. (April 2008). "A Reassessment of Prebiotic Organic Synthesis in Neutral Planetary Atmospheres". Origins of Life and Evolution of Biospheres. 38 (2): 105–115. Bibcode:2008OLEB...38..105C. doi:10.1007/s11084-007-9120-3. PMID 18204914. S2CID 7731172.
- ^ Chyba, Christopher F. (13 May 2005). "Rethinking Earth's Early Atmosphere". Science. 308 (5724): 962–963. doi:10.1126/science.1113157. PMID 15890865. S2CID 93303848.
- ^ Barton et al. 2007, pp. 93–95
- ^ Bada & Lazcano 2009, pp. 56–57
- ^ Bada, Jeffrey L.; Lazcano, Antonio (2 May 2003). "Prebiotic Soup – Revisiting the Miller Experiment" (PDF). Science. 300 (5620): 745–746. doi:10.1126/science.1085145. PMID 12730584. S2CID 93020326. Archived (PDF) from the original on 4 March 2016. Retrieved 13 June 2015.
- ^ Madau, Piero; Dickinson, Mark (18 August 2014). "Cosmic Star-Formation History". Annual Review of Astronomy and Astrophysics. 52 (1): 415–486. arXiv:1403.0007. Bibcode:2014ARA&A..52..415M. doi:10.1146/annurev-astro-081811-125615. S2CID 658354. Archived from the original on 1 July 2022. Retrieved 8 December 2023.
- ^ Marigo, Paola; et al. (6 July 2020). "Carbon star formation as seen through the non-monotonic initial–final mass relation". Nature Astronomy. 152 (11): 1102–1110. arXiv:2007.04163. Bibcode:2020NatAs...4.1102M. doi:10.1038/s41550-020-1132-1. S2CID 220403402. Archived from the original on 16 February 2023. Retrieved 7 July 2020.
- ^ "WMAP- Life in the Universe". Archived from the original on 29 January 2023. Retrieved 27 September 2019.
- ^ "Formation of Solar Systems: Solar Nebular Theory". University of Massachusetts Amherst. Archived from the original on 27 September 2019. Retrieved 27 September 2019.
- ^ "Age of the Earth". United States Geological Survey. 9 July 2007. Archived from the original on 23 December 2005. Retrieved 10 January 2006.
- ^ Dalrymple 2001, pp. 205–221
- ^ Fesenkov 1959, p. 9
- ^ Bottke, W. F.; Vokrouhlický, D.; Marchi, S.; Swindle, T.; Scott, E. R. D.; Weirich, J. R.; Levison, H. (17 April 2015). "Dating the Moon-forming impact event with asteroidal meteorites". Science. 348 (6232): 321–323. Bibcode:2015Sci...348..321B. doi:10.1126/science.aaa0602. PMID 25883354. S2CID 206632612.
- ^ Kasting, James F. (12 February 1993). "Earth's Early Atmosphere" (PDF). Science. 259 (5097): 920–926. Bibcode:1993Sci...259..920K. doi:10.1126/science.11536547. PMID 11536547. S2CID 21134564. Archived from the original (PDF) on 10 October 2015. Retrieved 28 July 2015.
- ^ a b c d e f Follmann, Hartmut; Brownson, Carol (November 2009). "Darwin's warm little pond revisited: from molecules to the origin of life". Naturwissenschaften. 96 (11): 1265–1292. Bibcode:2009NW.....96.1265F. doi:10.1007/s00114-009-0602-1. PMID 19760276. S2CID 23259886.
- ^ Morse, John (September 1998). "Hadean Ocean Carbonate Geochemistry". Aquatic Geochemistry. 4 (3/4): 301–319. Bibcode:1998MinM...62.1027M. doi:10.1023/A:1009632230875. S2CID 129616933.
- ^ Sleep, Norman H.; Zahnle, Kevin J.; Lupu, Roxana E. (13 September 2014). "Terrestrial aftermath of the Moon-forming impact". Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 372 (2024): 20130172. Bibcode:2014RSPTA.37230172S. doi:10.1098/rsta.2013.0172. PMID 25114303. S2CID 6902632.
- ^ Morse, John W.; Mackenzie, Fred T. (1998). "[No title found]". Aquatic Geochemistry. 4 (3/4): 301–319. doi:10.1023/A:1009632230875. S2CID 129616933. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Crowley, James L.; Myers, John S.; Sylvester, Paul J; Cox, Richard A. (May 2005). "Detrital Zircon from the Jack Hills and Mount Narryer, Western Australia: Evidence for Diverse >4.0 Ga Source Rocks". The Journal of Geology. 113 (3): 239–263. Bibcode:2005JG....113..239C. doi:10.1086/428804. S2CID 140715676. Archived from the original on 16 December 2023. Retrieved 8 December 2023.
- ^ Wilde, Simon A.; Valley, John W.; Peck, William H.; Graham, Colin M. (11 January 2001). "Evidence from detrital zircons for the existence of continental crust and oceans on the Earth 4.4 Gyr ago" (PDF). Nature. 409 (6817): 175–178. Bibcode:2001Natur.409..175W. doi:10.1038/35051550. PMID 11196637. S2CID 4319774. Archived (PDF) from the original on 5 June 2015. Retrieved 3 June 2015.
- ^ Korenaga, Jun (December 2008). "Plate tectonics, flood basalts and the evolution of Earth's oceans". Terra Nova. 20 (6): 419–439. Bibcode:2008TeNov..20..419K. doi:10.1111/j.1365-3121.2008.00843.x. S2CID 36766331.
- ^ Rosing, Minik T.; Bird, Dennis K.; Sleep, Norman H.; et al. (22 March 2006). "The rise of continents – An essay on the geologic consequences of photosynthesis". Palaeogeography, Palaeoclimatology, Palaeoecology. 232 (2–4): 99–113. Bibcode:2006PPP...232...99R. doi:10.1016/j.palaeo.2006.01.007. Archived (PDF) from the original on 14 July 2015. Retrieved 8 June 2015.
- ^ Tera, Fouad; Papanastassiou, D.A.; Wasserburg, G.J. (April 1974). "Isotopic evidence for a terminal lunar cataclysm". Earth and Planetary Science Letters. 22 (1): 1–21. Bibcode:1974E&PSL..22....1T. doi:10.1016/0012-821x(74)90059-4. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Stoffler, D. (1 January 2006). "Cratering History and Lunar Chronology". Reviews in Mineralogy and Geochemistry. 60 (1): 519–596. Bibcode:2006RvMG...60..519S. doi:10.2138/rmg.2006.60.05. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Sleep, Norman H.; Zahnle, Kevin J.; Kasting, James F.; Morowitz, Harold J. (December 1989). "Annihilation of ecosystems by large asteroid impacts on the early Earth". Nature. 342 (6246): 139–142. Bibcode:1989Natur.342..139S. doi:10.1038/342139a0. PMID 11536616. S2CID 1137852. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Fassett, Caleb I.; Minton, David A. (23 June 2013). "Impact bombardment of the terrestrial planets and the early history of the Solar System". Nature Geoscience. 6 (7): 520–524. Bibcode:2013NatGe...6..520F. doi:10.1038/ngeo1841. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Abramov, Oleg; Mojzsis, Stephen J. (May 2009). "Microbial habitability of the Hadean Earth during the late heavy bombardment". Nature. 459 (7245): 419–422. Bibcode:2009Natur.459..419A. doi:10.1038/nature08015. PMID 19458721. S2CID 3304147. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Boehnke, Patrick; Harrison, T. Mark (12 September 2016). "Illusory Late Heavy Bombardments". Proceedings of the National Academy of Sciences. 113 (39): 10802–10806. Bibcode:2016PNAS..11310802B. doi:10.1073/pnas.1611535113. PMC 5047187. PMID 27621460.
- ^ Zellner, Nicolle E. B. (3 May 2017). "Cataclysm No More: New Views on the Timing and Delivery of Lunar Impactors". Origins of Life and Evolution of Biospheres. 47 (3): 261–280. arXiv:1704.06694. Bibcode:2017OLEB...47..261Z. doi:10.1007/s11084-017-9536-3. PMC 5602003. PMID 28470374. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Lowe, Donald R.; Byerly, Gary R. (1 April 2018). "The terrestrial record of Late Heavy Bombardment". New Astronomy Reviews. 81: 39–61. Bibcode:2018NewAR..81...39L. doi:10.1016/j.newar.2018.03.002.
- ^ Davies 1999, p. 155
- ^ Bock & Goode 1996
- ^ a b Dodd, Matthew S.; Papineau, Dominic; Grenne, Tor; et al. (1 March 2017). "Evidence for early life in Earth's oldest hydrothermal vent precipitates". Nature. 543 (7643): 60–64. Bibcode:2017Natur.543...60D. doi:10.1038/nature21377. PMID 28252057. Archived from the original on 8 September 2017. Retrieved 2 March 2017.
- ^ Ohtomo, Yoko; Kakegawa, Takeshi; Ishida, Akizumi; et al. (January 2014). "Evidence for biogenic graphite in early Archaean Isua metasedimentary rocks". Nature Geoscience. 7 (1): 25–28. Bibcode:2014NatGe...7...25O. doi:10.1038/ngeo2025.
- ^ Noffke, Nora; Christian, Daniel; Wacey, David; Hazen, Robert M. (16 November 2013). "Microbially Induced Sedimentary Structures Recording an Ancient Ecosystem in the ca. 3.48 Gyo Dresser Formation, Pilbara, Western Australia". Astrobiology. 13 (12): 1103–1124. Bibcode:2013AsBio..13.1103N. doi:10.1089/ast.2013.1030. PMC 3870916. PMID 24205812.
- ^ Davies 1999
- ^ Hassenkam, T.; Andersson, M. P.; Dalby, K. N.; Mackenzie, D. M. A.; Rosing, M.T. (2017). "Elements of Eoarchean life trapped in mineral inclusions". Nature. 548 (7665): 78–81. Bibcode:2017Natur.548...78H. doi:10.1038/nature23261. PMID 28738409. S2CID 205257931.
- ^ O'Donoghue, James (21 August 2011). "Oldest reliable fossils show early life was a beach". New Scientist. 211: 13. doi:10.1016/S0262-4079(11)62064-2. Archived from the original on 30 June 2015.
- ^ Wacey, David; Kilburn, Matt R.; Saunders, Martin; et al. (October 2011). "Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia". Nature Geoscience. 4 (10): 698–702. Bibcode:2011NatGe...4..698W. doi:10.1038/ngeo1238.
- ^ Bell, Elizabeth A.; Boehnke, Patrick; Harrison, T. Mark; Mao, Wendy L. (24 November 2015). "Potentially biogenic carbon preserved in a 4.1 billion-year-old zircon". Proceedings of the National Academy of Sciences. 112 (47): 14518–14521. Bibcode:2015PNAS..11214518B. doi:10.1073/pnas.1517557112. PMC 4664351. PMID 26483481.
- ^ Baumgartner, Rafael; Van Kranendonk, Martin; Wacey, David; et al. (2019). "Nano−porous pyrite and organic matter in 3.5-billion-year-old stromatolites record primordial life" (PDF). Geology. 47 (11): 1039–1043. Bibcode:2019Geo....47.1039B. doi:10.1130/G46365.1. S2CID 204258554. Archived (PDF) from the original on 5 December 2020. Retrieved 10 January 2021.
- ^ Djokic, Tara; Van Kranendonk, Martin J.; Campbell, Kathleen A.; Walter, Malcolm R.; Ward, Colin R. (9 May 2017). "Earliest signs of life on land preserved in ca. 3.5 Gao hot spring deposits". Nature Communications. 8: 15263. Bibcode:2017NatCo...815263D. doi:10.1038/ncomms15263. PMC 5436104. PMID 28486437.
- ^ Betts, Holly C.; Puttick, Mark N.; Clark, James W.; Williams, Tom A.; Donoghue, Philip C. J.; Pisani, Davide (20 August 2018). "Integrated genomic and fossil evidence illuminates life's early evolution and eukaryote origin". Nature Ecology & Evolution. 2 (10): 1556–1562. Bibcode:2018NatEE...2.1556B. doi:10.1038/s41559-018-0644-x. PMC 6152910. PMID 30127539.
- ^ Landau, Elizabeth (12 October 2016). "Building Blocks of Life's Building Blocks Come From Starlight". NASA. Archived from the original on 13 October 2016. Retrieved 13 October 2016.
- ^ a b Ehrenfreund, Pascale; Cami, Jan (December 2010). "Cosmic carbon chemistry: from the interstellar medium to the early Earth". Cold Spring Harbor Perspectives in Biology. 2 (12): a002097. doi:10.1101/cshperspect.a002097. PMC 2982172. PMID 20554702.
- ^ Geballe, Thomas R.; Najarro, Francisco; Figer, Donald F.; et al. (10 November 2011). "Infrared diffuse interstellar bands in the Galactic Centre region". Nature. 479 (7372): 200–202. arXiv:1111.0613. Bibcode:2011Natur.479..200G. doi:10.1038/nature10527. PMID 22048316. S2CID 17223339.
- ^ Klyce 2001
- ^ a b c d Hoover, Rachel (21 February 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". Ames Research Center. NASA. Archived from the original on 6 September 2015. Retrieved 22 June 2015.
- ^ Goncharuk, Vladislav V.; Zui, O. V. (February 2015). "Water and carbon dioxide as the main precursors of organic matter on Earth and in space". Journal of Water Chemistry and Technology. 37 (1): 2–3. Bibcode:2015JWCT...37....2G. doi:10.3103/S1063455X15010026. S2CID 97965067.
- ^ Abou Mrad, Ninette; Vinogradoff, Vassilissa; Duvernay, Fabrice; et al. (2015). "Laboratory experimental simulations: Chemical evolution of the organic matter from interstellar and cometary ice analogs". Bulletin de la Société Royale des Sciences de Liège. 84: 21–32. Bibcode:2015BSRSL..84...21A. Archived from the original on 13 April 2015. Retrieved 6 April 2015.
- ^ Oba, Yasuhiro; et al. (26 April 2022). "Identifying the wide diversity of extraterrestrial purine and pyrimidine nucleobases in carbonaceous meteorites". Nature Communications. 13 (2008): 2008. Bibcode:2022NatCo..13.2008O. doi:10.1038/s41467-022-29612-x. PMC 9042847. PMID 35473908. S2CID 248402205.
- ^ "'Life chemical' detected in comet". BBC News. London. 18 August 2009. Archived from the original on 25 May 2015. Retrieved 23 June 2015.
- ^ Thompson, William Reid; Murray, B. G.; Khare, Bishun Narain; Sagan, Carl (30 December 1987). "Coloration and darkening of methane clathrate and other ices by charged particle irradiation: Applications to the outer solar system". Journal of Geophysical Research. 92 (A13): 14933–14947. Bibcode:1987JGR....9214933T. doi:10.1029/JA092iA13p14933. PMID 11542127.
- ^ Goldman, Nir; Tamblyn, Isaac (20 June 2013). "Prebiotic Chemistry within a Simple Impacting Icy Mixture". Journal of Physical Chemistry A. 117 (24): 5124–5131. Bibcode:2013JPCA..117.5124G. doi:10.1021/jp402976n. PMID 23639050. S2CID 5144843. Archived from the original on 21 July 2018. Retrieved 29 August 2019.
- ^ "NASA Ames PAH IR Spectroscopic Database". NASA. Archived from the original on 29 June 2015. Retrieved 17 June 2015.
- ^ a b c Hudgins, Douglas M.; Bauschlicher, Charles W. Jr.; Allamandola, Louis J. (10 October 2005). "Variations in the Peak Position of the 6.2 μm Interstellar Emission Feature: A Tracer of N in the Interstellar Polycyclic Aromatic Hydrocarbon Population". The Astrophysical Journal. 632 (1): 316–332. Bibcode:2005ApJ...632..316H. CiteSeerX 10.1.1.218.8786. doi:10.1086/432495. S2CID 7808613.
- ^ a b c Des Marais, David J.; Allamandola, Louis J.; Sandford, Scott; et al. (2009). "Cosmic Distribution of Chemical Complexity". Ames Research Center. Mountain View, California: NASA. Archived from the original on 27 February 2014. Retrieved 24 June 2015.
- ^ a b Carey, Bjorn (18 October 2005). "Life's Building Blocks 'Abundant in Space'". Space.com. Watsonville, California: Imaginova. Archived from the original on 26 June 2015. Retrieved 23 June 2015.
- ^ García-Hernández, Domingo. A.; Manchado, Arturo; García-Lario, Pedro; et al. (20 November 2010). "Formation of Fullerenes in H-Containing Planetary Nebulae". The Astrophysical Journal Letters. 724 (1): L39–L43. arXiv:1009.4357. Bibcode:2010ApJ...724L..39G. doi:10.1088/2041-8205/724/1/L39. S2CID 119121764.
- ^ d'Ischia, Marco; Manini, Paola; Moracci, Marco; et al. (21 August 2019). "Astrochemistry and Astrobiology: Materials Science in Wonderland?". International Journal of Molecular Sciences. 20 (17): 4079. doi:10.3390/ijms20174079. PMC 6747172. PMID 31438518.
- ^ Gudipati, Murthy S.; Yang, Rui (1 September 2012). "In-situ Probing of Radiation-induced Processing of Organics in Astrophysical Ice Analogs – Novel Laser Desorption Laser Ionization Time-of-flight Mass Spectroscopic Studies". The Astrophysical Journal Letters. 756 (1): L24. Bibcode:2012ApJ...756L..24G. doi:10.1088/2041-8205/756/1/L24. S2CID 5541727.
- ^ a b Gallori, Enzo (June 2011). "Astrochemistry and the origin of genetic material". Rendiconti Lincei. 22 (2): 113–118. doi:10.1007/s12210-011-0118-4. S2CID 96659714. "Paper presented at the Symposium 'Astrochemistry: molecules in space and time' (Rome, 4–5 November 2010), sponsored by Fondazione 'Guido Donegani', Accademia Nazionale dei Lincei."
- ^ Martins, Zita (February 2011). "Organic Chemistry of Carbonaceous Meteorites". Elements. 7 (1): 35–40. Bibcode:2011Eleme...7...35M. doi:10.2113/gselements.7.1.35.
- ^ Martins, Zita; Botta, Oliver; Fogel, Marilyn L.; et al. (15 June 2008). "Extraterrestrial nucleobases in the Murchison meteorite". Earth and Planetary Science Letters. 270 (1–2): 130–136. arXiv:0806.2286. Bibcode:2008E&PSL.270..130M. doi:10.1016/j.epsl.2008.03.026. S2CID 14309508.
- ^ Callahan, Michael P.; Smith, Karen E.; Cleaves, H. James II; et al. (23 August 2011). "Carbonaceous meteorites contain a wide range of extraterrestrial nucleobases". PNAS. 108 (34): 13995–13998. Bibcode:2011PNAS..10813995C. doi:10.1073/pnas.1106493108. PMC 3161613. PMID 21836052.
- ^ Steigerwald, John (8 August 2011). "NASA Researchers: DNA Building Blocks Can Be Made in Space". Goddard Space Flight Center. NASA. Archived from the original on 23 June 2015. Retrieved 23 June 2015.
- ^ Kwok, Sun; Zhang, Yong (3 November 2011). "Mixed aromatic–aliphatic organic nanoparticles as carriers of unidentified infrared emission features". Nature. 479 (7371): 80–83. Bibcode:2011Natur.479...80K. doi:10.1038/nature10542. PMID 22031328. S2CID 4419859.
- ^ Jørgensen, Jes K.; Favre, Cécile; Bisschop, Suzanne E.; et al. (2012). "Detection of the simplest sugar, glycolaldehyde, in a solar-type protostar with ALMA" (PDF). The Astrophysical Journal Letters. 757 (1): L4. arXiv:1208.5498. Bibcode:2012ApJ...757L...4J. doi:10.1088/2041-8205/757/1/L4. S2CID 14205612. Archived (PDF) from the original on 24 September 2015. Retrieved 23 June 2015.
- ^ Furukawa, Yoshihiro; Chikaraishi, Yoshito; Ohkouchi, Naohiko; et al. (13 November 2019). "Extraterrestrial ribose and other sugars in primitive meteorites". PNAS. 116 (49): 24440–24445. Bibcode:2019PNAS..11624440F. doi:10.1073/pnas.1907169116. PMC 6900709. PMID 31740594.
- ^ Oró, Joan; Kimball, Aubrey P. (February 1962). "Synthesis of purines under possible primitive earth conditions: II. Purine intermediates from hydrogen cyanide". Archives of Biochemistry and Biophysics. 96 (2): 293–313. doi:10.1016/0003-9861(62)90412-5. PMID 14482339.
- ^ Cleaves II, Henderson (2010). "The origin of the biologically coded amino acids". Journal of Theoretical Biology. 263 (4): 490–498. Bibcode:2010JThBi.263..490C. doi:10.1016/j.jtbi.2009.12.014. PMID 20034500.
- ^ Green, N. J., Russell, D. A., Tanner, S. H., Sutherland, J. D. (2023). "Prebiotic Synthesis of N-Formylaminonitriles and Derivatives in Formamide". Journal of the American Chemical Society. 145 (19): 10533–10541. doi:10.1021/jacs.2c13306. PMC 10197134. PMID 37146260.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Breslow, R. (1959). "On the Mechanism of the Formose Reaction". Tetrahedron Letters. 1 (21): 22–26. doi:10.1016/S0040-4039(01)99487-0.
- ^ Oró, Joan (16 September 1961). "Mechanism of Synthesis of Adenine from Hydrogen Cyanide under Possible Primitive Earth Conditions". Nature. 191 (4794): 1193–1194. Bibcode:1961Natur.191.1193O. doi:10.1038/1911193a0. PMID 13731264. S2CID 4276712.
- ^ a b Saladino, Raffaele; Crestini, Claudia; Pino, Samanta; et al. (March 2012). "Formamide and the origin of life" (PDF). Physics of Life Reviews. 9 (1): 84–104. Bibcode:2012PhLRv...9...84S. doi:10.1016/j.plrev.2011.12.002. hdl:2108/85168. PMID 22196896. Archived (PDF) from the original on 27 January 2023. Retrieved 29 August 2019.
- ^ a b Saladino, Raffaele; Botta, Giorgia; Pino, Samanta; et al. (July 2012). "From the one-carbon amide formamide to RNA all the steps are prebiotically possible". Biochimie. 94 (7): 1451–1456. doi:10.1016/j.biochi.2012.02.018. hdl:11573/515604. PMID 22738728.
- ^ Marlaire, Ruth, ed. (3 March 2015). "NASA Ames Reproduces the Building Blocks of Life in Laboratory". Ames Research Center. NASA. Archived from the original on 5 March 2015. Retrieved 5 March 2015.
- ^ Ferus, Martin; Nesvorný, David; Šponer, Jiří; et al. (2015). "High-energy chemistry of formamide: A unified mechanism of nucleobase formation". PNAS. 112 (3): 657–662. Bibcode:2015PNAS..112..657F. doi:10.1073/pnas.1412072111. PMC 4311869. PMID 25489115.
- ^ Basile, Brenda; Lazcano, Antonio; Oró, Joan (1984). "Prebiotic syntheses of purines and pyrimidines". Advances in Space Research. 4 (12): 125–131. Bibcode:1984AdSpR...4l.125B. doi:10.1016/0273-1177(84)90554-4. PMID 11537766.
- ^ Orgel, Leslie E. (August 2004). "Prebiotic Adenine Revisited: Eutectics and Photochemistry". Origins of Life and Evolution of Biospheres. 34 (4): 361–369. Bibcode:2004OLEB...34..361O. doi:10.1023/B:ORIG.0000029882.52156.c2. PMID 15279171. S2CID 4998122.
- ^ Robertson, Michael P.; Miller, Stanley L. (29 June 1995). "An efficient prebiotic synthesis of cytosine and uracil". Nature. 375 (6534): 772–774. Bibcode:1995Natur.375..772R. doi:10.1038/375772a0. PMID 7596408. S2CID 4351012.
- ^ Fox, Douglas (1 February 2008). "Did Life Evolve in Ice?". Discover. Archived from the original on 30 June 2008. Retrieved 3 July 2008.
- ^ Levy, Matthew; Miller, Stanley L.; Brinton, Karen; Bada, Jeffrey L. (June 2000). "Prebiotic Synthesis of Adenine and Amino Acids Under Europa-like Conditions". Icarus. 145 (2): 609–613. Bibcode:2000Icar..145..609L. doi:10.1006/icar.2000.6365. PMID 11543508.
- ^ Menor-Salván, César; Ruiz-Bermejo, Marta; Guzmán, Marcelo I.; et al. (20 April 2009). "Synthesis of Pyrimidines and Triazines in Ice: Implications for the Prebiotic Chemistry of Nucleobases". Chemistry: A European Journal. 15 (17): 4411–4418. doi:10.1002/chem.200802656. PMID 19288488.
- ^ Roy, Debjani; Najafian, Katayoun; von Ragué Schleyer, Paul (30 October 2007). "Chemical evolution: The mechanism of the formation of adenine under prebiotic conditions". PNAS. 104 (44): 17272–17277. Bibcode:2007PNAS..10417272R. doi:10.1073/pnas.0708434104. PMC 2077245. PMID 17951429.
- ^ a b Frenkel-Pinter, Moran; Samanta, Mousumi; Ashkenasy, Gonen; Leman, Luke J. (10 June 2020). "Prebiotic Peptides: Molecular Hubs in the Origin of Life". Chemical Reviews. 120 (11): 4707–4765. doi:10.1021/acs.chemrev.9b00664. ISSN 0009-2665. PMID 32101414.
- ^ Campbell, Thomas D.; Febrian, Rio; McCarthy, Jack T.; Kleinschmidt, Holly E.; Forsythe, Jay G.; Bracher, Paul J. (2019). "Prebiotic condensation through wet–dry cycling regulated by deliquescence". Nature Communications. 10 (1): 4508. Bibcode:2019NatCo..10.4508C. doi:10.1038/s41467-019-11834-1. PMC 6778215. PMID 31586058.
- ^ Marshall-Bowman, Karina; Ohara, Shohei; Sverjensky, Dimitri A.; Hazen, Robert M.; Cleaves, H. James (October 2010). "Catalytic peptide hydrolysis by mineral surface: Implications for prebiotic chemistry". Geochimica et Cosmochimica Acta. 74 (20): 5852–5861. Bibcode:2010GeCoA..74.5852M. doi:10.1016/j.gca.2010.07.009. ISSN 0016-7037.
- ^ Matreux, Thomas; Aikkila, Paula; Scheu, Bettina; Braun, Dieter; Mast, Christof B. (April 2024). "Heat flows enrich prebiotic building blocks and enhance their reactivity". Nature. 628 (8006): 110–116. Bibcode:2024Natur.628..110M. doi:10.1038/s41586-024-07193-7. ISSN 1476-4687. PMC 10990939. PMID 38570715.
- ^ Paecht-Horowitz, Mella (1 January 1974). "The possible role of clays in prebiotic peptide synthesis". Origins of Life. 5 (1): 173–187. Bibcode:1974OrLi....5..173P. doi:10.1007/BF00927022. ISSN 1573-0875. PMID 4842069.
- ^ Lancet, Doron (30 December 2014). "Systems Prebiology-Studies of the origin of Life". The Lancet Lab. Rehovot, Israel: Department of Molecular Genetics; Weizmann Institute of Science. Archived from the original on 26 June 2015. Retrieved 26 June 2015.
- ^ Segré, Daniel; Ben-Eli, Dafna; Deamer, David W.; Lancet, Doron (February 2001). "The Lipid World" (PDF). Origins of Life and Evolution of Biospheres. 31 (1–2): 119–145. Bibcode:2001OLEB...31..119S. doi:10.1023/A:1006746807104. PMID 11296516. S2CID 10959497. Archived (PDF) from the original on 26 June 2015.
- ^ a b c Chen, Irene A.; Walde, Peter (July 2010). "From Self-Assembled Vesicles to Protocells". Cold Spring Harbor Perspectives in Biology. 2 (7): a002170. doi:10.1101/cshperspect.a002170. PMC 2890201. PMID 20519344.
- ^ Eigen, Manfred; Schuster, Peter (November 1977). "The Hypercycle. A Principle of Natural Self-Organization. Part A: Emergence of the Hypercycle" (PDF). Naturwissenschaften. 64 (11): 541–65. Bibcode:1977NW.....64..541E. doi:10.1007/bf00450633. PMID 593400. S2CID 42131267. Archived from the original (PDF) on 3 March 2016.
- Eigen, Manfred; Schuster, Peter (1978). "The Hypercycle. A Principle of Natural Self-Organization. Part B: The Abstract Hypercycle" (PDF). Naturwissenschaften. 65 (1): 7–41. Bibcode:1978NW.....65....7E. doi:10.1007/bf00420631. S2CID 1812273. Archived from the original (PDF) on 3 March 2016.
- Eigen, Manfred; Schuster, Peter (July 1978). "The Hypercycle. A Principle of Natural Self-Organization. Part C: The Realistic Hypercycle" (PDF). Naturwissenschaften. 65 (7): 341–369. Bibcode:1978NW.....65..341E. doi:10.1007/bf00439699. S2CID 13825356. Archived from the original (PDF) on 16 June 2016.
- ^ Markovitch, Omer; Lancet, Doron (Summer 2012). "Excess Mutual Catalysis Is Required for Effective Evolvability". Artificial Life. 18 (3): 243–266. doi:10.1162/artl_a_00064. PMID 22662913. S2CID 5236043.
- ^ Tessera, Marc (2011). "Origin of Evolution versus Origin of Life: A Shift of Paradigm". International Journal of Molecular Sciences. 12 (6): 3445–3458. doi:10.3390/ijms12063445. PMC 3131571. PMID 21747687. Special Issue: "Origin of Life 2011"
- ^ "Protocells". Exploring Life's Origins: A Virtual Exhibit. Arlington County, Virginia: National Science Foundation. Archived from the original on 28 February 2014. Retrieved 13 September 2023.
- ^ a b c Chen, Irene A. (8 December 2006). "The Emergence of Cells During the Origin of Life". Science. 314 (5805): 1558–1559. doi:10.1126/science.1137541. PMID 17158315.
- ^ Zimmer, Carl (26 June 2004). "What Came Before DNA?". Discover. Archived from the original on 19 March 2014.
- ^ Onsager, Lars (1931). "Reciprocal Relations in Irreversible Processes I". Physical Review. 37 (4): 405. Bibcode:1931PhRv...37..405O. doi:10.1103/PhysRev.37.405.
- ^ Onsager, Lars (1931). "Reciprocal Relations in Irreversible Processes II". Physical Review. 38 (12): 2265. Bibcode:1931PhRv...38.2265O. doi:10.1103/PhysRev.38.2265.
- ^ Prigogine, Ilya (1967). An Introduction to the Thermodynamics of Irreversible Processes. New York: Wiley.
- ^ Shapiro, Robert (June 2007). "A Simpler Origin for Life". Scientific American. 296 (6): 46–53. Bibcode:2007SciAm.296f..46S. doi:10.1038/scientificamerican0607-46. PMID 17663224. Archived from the original on 14 June 2015.
- ^ Chang 2007
- ^ a b c d Lane, Nick (2015). The Vital Question: Why Is Life The Way It Is?. Profile Books. pp. 129–140. ISBN 978-1-78125-036-5.
- ^ Jordan, Sean F.; Nee, Eloise; Lane, Nick (6 December 2019). "Isoprenoids enhance the stability of fatty acid membranes at the emergence of life potentially leading to an early lipid divide". Interface Focus. 9 (6): 20190067. doi:10.1098/rsfs.2019.0067. PMC 6802135. PMID 31641436.
- ^ Mayer, Christian; Schreiber, Ulrich; Dávila, María J.; Schmitz, Oliver J.; Bronja, Amela; Meyer, Martin; Klein, Julia; Meckelmann, Sven W. (2018). "Molecular Evolution in a Peptide-Vesicle System". Life. 8 (2): 16. Bibcode:2018Life....8...16M. doi:10.3390/life8020016. ISSN 2075-1729. PMC 6027363. PMID 29795023.
- ^ Mayer, Christian; Schreiber, Ulrich; Dávila, María J. (1 June 2015). "Periodic Vesicle Formation in Tectonic Fault Zones—an Ideal Scenario for Molecular Evolution". Origins of Life and Evolution of Biospheres. 45 (1): 139–148. Bibcode:2015OLEB...45..139M. doi:10.1007/s11084-015-9411-z. ISSN 1573-0875. PMC 4457167. PMID 25716918.
- ^ Dávila, María J.; Mayer, Christian (2022). "Membrane Structure Obtained in an Experimental Evolution Process". Life. 12 (2): 145. Bibcode:2022Life...12..145D. doi:10.3390/life12020145. ISSN 2075-1729. PMC 8875328. PMID 35207433.
- ^ Mayer, Christian (2022). "Spontaneous Formation of Functional Structures in Messy Environments". Life. 12 (5): 720. Bibcode:2022Life...12..720M. doi:10.3390/life12050720. ISSN 2075-1729. PMC 9148140. PMID 35629387.
- ^ Dávila, María J.; Mayer, Christian (2023). "Structural Phenomena in a Vesicle Membrane Obtained through an Evolution Experiment: A Study Based on MD Simulations". Life. 13 (8): 1735. Bibcode:2023Life...13.1735D. doi:10.3390/life13081735. ISSN 2075-1729. PMC 10455627. PMID 37629592.
- ^ Mayer, Christian (18 January 2020). "Life in The Context of Order and Complexity". Life. 10 (1): 5. Bibcode:2020Life...10....5M. doi:10.3390/life10010005. ISSN 2075-1729. PMC 7175320. PMID 31963637.
- ^ Sharov, Alexei A.; Gordon, Richard (2018). "Life Before Earth". Habitability of the Universe Before Earth: Life Before Earth. Astrobiology Exploring Life on Earth and Beyond. Academic Press. pp. 265–296. doi:10.1016/B978-0-12-811940-2.00011-3. ISBN 978-0-12-811940-2. S2CID 117048600. Archived from the original on 30 April 2022. Retrieved 30 April 2022.
- ^ Ladyman, J.; Lambert, J.; Weisner, K. B. (2013). "What is a Complex System?". European Journal of the Philosophy of Science. 3: 33–67. doi:10.1007/s13194-012-0056-8. S2CID 18787276.
- ^ Esposito, M.; Lindenberg, Katja; Van den Broeck, C. (2010). "Entropy production as correlation between system and reservoir". New Journal of Physics. 12 (1): 013013. arXiv:0908.1125. Bibcode:2010NJPh...12a3013E. doi:10.1088/1367-2630/12/1/013013. S2CID 26657293.
- ^ Bar-Nun, A.; Bar-Nun, N.; Bauer, S. H.; Sagan, Carl (24 April 1970). "Shock Synthesis of Amino Acids in Simulated Primitive Environments". Science. 168 (3930): 470–473. Bibcode:1970Sci...168..470B. doi:10.1126/science.168.3930.470. PMID 5436082. S2CID 42467812.
- ^ Anbar, Michael (27 September 1968). "Cavitation during Impact of Liquid Water on Water: Geochemical Implications". Science. 161 (3848): 1343–1344. Bibcode:1968Sci...161.1343A. doi:10.1126/science.161.3848.1343. PMID 17831346.
- ^ Dharmarathne, Leena; Grieser, Franz (7 January 2016). "Formation of Amino Acids on the Sonolysis of Aqueous Solutions Containing Acetic Acid, Methane, or Carbon Dioxide, in the Presence of Nitrogen Gas". The Journal of Physical Chemistry A. 120 (2): 191–199. Bibcode:2016JPCA..120..191D. doi:10.1021/acs.jpca.5b11858. PMID 26695890.
- ^ Patehebieke, Yeersen; Zhao, Ze-Run; Wang, Su; Xu, Hao-Xing; Chen, Qian-Qian; Wang, Xiao (2021). "Cavitation as a plausible driving force for the prebiotic formation of N9 purine nucleosides". Cell Reports Physical Science. 2 (3): 100375. Bibcode:2021CRPS....200375P. doi:10.1016/j.xcrp.2021.100375. S2CID 233662126.
- ^ Kalson, Natan-Haim; Furman, David; Zeiri, Yehuda (11 September 2017). "Cavitation-Induced Synthesis of Biogenic Molecules on Primordial Earth". ACS Central Science. 3 (9): 1041–1049. doi:10.1021/acscentsci.7b00325. PMC 5620973. PMID 28979946. S2CID 21409351.
- ^ Muller, Anthonie W. J. (1995). "Were the first organisms heat engines? A new model for biogenesis and the early evolution of biological energy conversion". Progress in Biophysics and Molecular Biology. 63 (2): 193–231. doi:10.1016/0079-6107(95)00004-7. PMID 7542789.
- ^ Muller, Anthonie W. J.; Schulze-Makuch, Dirk (2006). "Thermal energy and the origin of life". Origins of Life and Evolution of Biospheres. 36 (2): 77–189. Bibcode:2006OLEB...36..177M. doi:10.1007/s11084-005-9003-4. PMID 16642267. S2CID 22179552.
- ^ Junge, Wolfgang; Nelson, Nathan (2 June 2015). "ATP Synthase". Annual Review of Biochemistry. 84 (1): 631–657. doi:10.1146/annurev-biochem-060614-034124. PMID 25839341.
- ^ a b c Damer, Bruce; Deamer, David (1 April 2020). "The Hot Spring Hypothesis for an Origin of Life". Astrobiology. 20 (4): 429–452. Bibcode:2020AsBio..20..429D. doi:10.1089/ast.2019.2045. PMC 7133448. PMID 31841362.
- ^ Benner, S. A.; Bell, E. A.; Biondi, E.; Brasser, R.; Carell, T.; Kim, H.-J.; Mojzsis, S. J.; Omran, A.; Pasek, M. A.; Trail, D. (2020). "When Did Life Likely Emerge on Earth in an RNA-First Process?". ChemSystemsChem. 2 (2). arXiv:1908.11327. doi:10.1002/syst.201900035.
- ^ * Copley, Shelley D.; Smith, Eric; Morowitz, Harold J. (December 2007). "The origin of the RNA world: Co-evolution of genes and metabolism" (PDF). Bioorganic Chemistry. 35 (6): 430–443. doi:10.1016/j.bioorg.2007.08.001. PMID 17897696. Archived (PDF) from the original on 5 September 2013. Retrieved 8 June 2015.
The proposal that life on Earth arose from an RNA world is widely accepted.
- Orgel, Leslie E. (April 2003). "Some consequences of the RNA world hypothesis". Origins of Life and Evolution of Biospheres. 33 (2): 211–218. Bibcode:2003OLEB...33..211O. doi:10.1023/A:1024616317965. PMID 12967268. S2CID 32779859.
It now seems very likely that our familiar DNA/RNA/protein world was preceded by an RNA world...
- Robertson & Joyce 2012: "There is now strong evidence indicating that an RNA World did indeed exist before DNA- and protein-based life."
- Neveu, Kim & Benner 2013: "[The RNA world's existence] has broad support within the community today."
- Orgel, Leslie E. (April 2003). "Some consequences of the RNA world hypothesis". Origins of Life and Evolution of Biospheres. 33 (2): 211–218. Bibcode:2003OLEB...33..211O. doi:10.1023/A:1024616317965. PMID 12967268. S2CID 32779859.
- ^ a b c Robertson, Michael P.; Joyce, Gerald F. (May 2012). "The origins of the RNA world". Cold Spring Harbor Perspectives in Biology. 4 (5): a003608. doi:10.1101/cshperspect.a003608. PMC 3331698. PMID 20739415.
- ^ a b c Cech, Thomas R. (July 2012). "The RNA Worlds in Context". Cold Spring Harbor Perspectives in Biology. 4 (7): a006742. doi:10.1101/cshperspect.a006742. PMC 3385955. PMID 21441585.
- ^ Pearce, Ben K. D.; Pudritz, Ralph E.; Semenov, Dmitry A.; Henning, Thomas K. (24 October 2017). "Origin of the RNA world: The fate of nucleobases in warm little ponds". PNAS. 114 (43): 11327–11332. arXiv:1710.00434. Bibcode:2017PNAS..11411327P. doi:10.1073/pnas.1710339114. PMC 5664528. PMID 28973920.
- ^ Yarus, Michael (April 2011). "Getting Past the RNA World: The Initial Darwinian Ancestor". Cold Spring Harbor Perspectives in Biology. 3 (4): a003590. doi:10.1101/cshperspect.a003590. PMC 3062219. PMID 20719875.
- ^ Voet & Voet 2004, p. 29
- ^ Fox, George.E. (9 June 2010). "Origin and evolution of the ribosome". Cold Spring Harbor Perspectives in Biology. 2 (9(a003483)): a003483. doi:10.1101/cshperspect.a003483. PMC 2926754. PMID 20534711.
- ^ Neveu, Marc; Kim, Hyo-Joong; Benner, Steven A. (22 April 2013). "The 'Strong' RNA World Hypothesis: Fifty Years Old". Astrobiology. 13 (4): 391–403. Bibcode:2013AsBio..13..391N. doi:10.1089/ast.2012.0868. PMID 23551238.
- ^ Gilbert, Walter (20 February 1986). "Origin of life: The RNA world". Nature. 319 (6055): 618. Bibcode:1986Natur.319..618G. doi:10.1038/319618a0. S2CID 8026658.
- ^ Orgel, Leslie E. (October 1994). "The origin of life on Earth". Scientific American. 271 (4): 76–83. Bibcode:1994SciAm.271d..76O. doi:10.1038/scientificamerican1094-76. PMID 7524147.
- ^ Lincoln, Tracey A.; Joyce, Gerald F. (27 February 2009). "Self-Sustained Replication of an RNA Enzyme". Science. 323 (5918): 1229–1232. Bibcode:2009Sci...323.1229L. doi:10.1126/science.1167856. PMC 2652413. PMID 19131595.
- ^ Joyce, Gerald F. (2009). "Evolution in an RNA world". Cold Spring Harbor Perspectives in Biology. 74 (Evolution: The Molecular Landscape): 17–23. doi:10.1101/sqb.2009.74.004. PMC 2891321. PMID 19667013.
- ^ Szostak, Jack W. (5 February 2015). "The Origins of Function in Biological Nucleic Acids, Proteins, and Membranes". Chevy Chase, Maryland: Howard Hughes Medical Institute. Archived from the original on 14 July 2015. Retrieved 16 June 2015.
- ^ Bernstein, Harris; Byerly, Henry C.; Hopf, Frederick A.; et al. (June 1983). "The Darwinian Dynamic". The Quarterly Review of Biology. 58 (2): 185–207. doi:10.1086/413216. JSTOR 2828805. S2CID 83956410.
- ^ Michod 1999
- ^ Palasek, Stan (23 May 2013). "Primordial RNA Replication and Applications in PCR Technology". arXiv:1305.5581v1 [q-bio.BM].
- ^ Vlassov, Alexander V.; Kazakov, Sergei A.; Johnston, Brian H.; et al. (August 2005). "The RNA World on Ice: A New Scenario for the Emergence of RNA Information". Journal of Molecular Evolution. 61 (2): 264–273. Bibcode:2005JMolE..61..264V. doi:10.1007/s00239-004-0362-7. PMID 16044244. S2CID 21096886.
- ^ Nussinov, Mark D.; Otroshchenko, Vladimir A.; Santoli, Salvatore (1997). "The emergence of the non-cellular phase of life on the fine-grained clayish particles of the early Earth's regolith". BioSystems. 42 (2–3): 111–118. Bibcode:1997BiSys..42..111N. doi:10.1016/S0303-2647(96)01699-1. PMID 9184757.
- ^ Kühnlein, Alexandra; Lanzmich, Simon A.; Brun, Dieter (2 March 2021). "tRNA sequences can assemble into a replicator". eLife. 10. doi:10.7554/eLife.63431. PMC 7924937. PMID 33648631.
- ^ Noller, Harry F. (April 2012). "Evolution of protein synthesis from an RNA world". Cold Spring Harbor Perspectives in Biology. 4 (4): a003681. doi:10.1101/cshperspect.a003681. PMC 3312679. PMID 20610545.
- ^ Koonin, Eugene V. (31 May 2007). "The cosmological model of eternal inflation and the transition from chance to biological evolution in the history of life". Biology Direct. 2: 15. doi:10.1186/1745-6150-2-15. PMC 1892545. PMID 17540027.
- ^ a b Tamura, K.; Alexander, R. W. (1 May 2004). "Peptide synthesis through evolution". Cellular and Molecular Life Sciences. 61 (11): 1317–1330. doi:10.1007/s00018-004-3449-9. ISSN 1420-9071. PMID 15170510.
- ^ Tamura, Koji; Schimmel, Paul (22 July 2003). "Peptide synthesis with a template-like RNA guide and aminoacyl phosphate adaptors". Proceedings of the National Academy of Sciences. 100 (15): 8666–8669. Bibcode:2003PNAS..100.8666T. doi:10.1073/pnas.1432909100. ISSN 0027-8424. PMC 166369. PMID 12857953.
- ^ a b Pressman, Abe D.; Liu, Ziwei; Janzen, Evan; Blanco, Celia; Müller, Ulrich F.; Joyce, Gerald F.; Pascal, Robert; Chen, Irene A. (17 April 2019). "Mapping a Systematic Ribozyme Fitness Landscape Reveals a Frustrated Evolutionary Network for Self-Aminoacylating RNA". Journal of the American Chemical Society. 141 (15): 6213–6223. doi:10.1021/jacs.8b13298. ISSN 0002-7863. PMC 6548421. PMID 30912655.
- ^ Tyagi, Sanjay; Ponnamperuma, Cyril (1 May 1990). "Nonrandomness in prebiotic peptide synthesis". Journal of Molecular Evolution. 30 (5): 391–399. Bibcode:1990JMolE..30..391T. doi:10.1007/BF02101111. ISSN 1432-1432. PMID 2111852.
- ^ Keefe, Anthony D.; Szostak, Jack W. (2001). "Functional proteins from a random-sequence library". Nature. 410 (6829): 715–718. Bibcode:2001Natur.410..715K. doi:10.1038/35070613. PMC 4476321. PMID 11287961.
- ^ Tong, Cher Ling; Lee, Kun-Hwa; Seelig, Burckhard (June 2021). "De novo proteins from random sequences through in vitro evolution". Current Opinion in Structural Biology. 68: 129–134. doi:10.1016/j.sbi.2020.12.014. PMC 8222087. PMID 33517151.
- ^ Boone, David R.; Castenholz, Richard W.; Garrity, George M., eds. (2001). The Archaea and the Deeply Branching and Phototrophic Bacteria. Bergey's Manual of Systematic Bacteriology. Springer. ISBN 978-0-387-21609-6. Archived from the original on 25 December 2014.[page needed]
- ^ Woese, C. R.; Fox, G. E. (1977). "Phylogenetic structure of the prokaryotic domain: the primary kingdoms". PNAS. 7 (11): 5088–5090. Bibcode:1977PNAS...74.5088W. doi:10.1073/pnas.74.11.5088. PMC 432104. PMID 270744.
- ^ Valas, R. E.; Bourne, P. E. (2011). "The origin of a derived superkingdom: how a gram-positive bacterium crossed the desert to become an archaeon". Biology Direct. 6: 16. doi:10.1186/1745-6150-6-16. PMC 3056875. PMID 21356104.
- ^ Cavalier-Smith, Thomas (2006). "Rooting the tree of life by transition analyses". Biology Direct. 1: 19. doi:10.1186/1745-6150-1-19. PMC 1586193. PMID 16834776.
- ^ "Early life liked it hot". Nature. 535 (7613): 468. 2016. doi:10.1038/535468b. S2CID 49905802.
- ^ Gogarten, Johann Peter; Deamer, David (25 November 2016). "Is LUCA a thermophilic progenote?". Nature Microbiology. 1 (12): 16229. doi:10.1038/nmicrobiol.2016.229. PMID 27886195. S2CID 205428194. Archived from the original on 3 April 2020. Retrieved 21 September 2022.
- ^ Catchpole, Ryan; Forterre, Patrick (2019). "The evolution of Reverse Gyrase suggests a non-hyperthermophilic Last Universal Common Ancestor". Molecular Biology and Evolution. 36 (12): 2737–2747. doi:10.1093/molbev/msz180. PMC 6878951. PMID 31504731. Archived from the original on 27 January 2023. Retrieved 18 September 2022.
- ^ Berkemer, Sarah J.; McGlynn, Shawn E (8 August 2020). "A New Analysis of Archaea–Bacteria Domain Separation: Variable Phylogenetic Distance and the Tempo of Early Evolution". Molecular Biology and Evolution. 37 (8): 2332–2340. doi:10.1093/molbev/msaa089. PMC 7403611. PMID 32316034. Archived from the original on 27 January 2023. Retrieved 21 September 2022.
- ^ Hoffmann, Geoffrey W. (25 June 1974). "On the origin of the genetic code and the stability of the translation apparatus". Journal of Molecular Biology. 86 (2): 349–362. doi:10.1016/0022-2836(74)90024-2. PMID 4414916.
- ^ Orgel, Leslie E. (April 1963). "The Maintenance of the Accuracy of Protein Synthesis and its Relevance to Ageing". PNAS. 49 (4): 517–521. Bibcode:1963PNAS...49..517O. doi:10.1073/pnas.49.4.517. PMC 299893. PMID 13940312.
- ^ Hoffmann, Geoffrey W. (October 1975). "The Stochastic Theory of the Origin of the Genetic Code". Annual Review of Physical Chemistry. 26: 123–144. Bibcode:1975ARPC...26..123H. doi:10.1146/annurev.pc.26.100175.001011.
- ^ Harrison, Stuart A.; Palmeira, Raquel Nunes; Halpern, Aaron; Lane, Nick (1 November 2022). "A biophysical basis for the emergence of the genetic code in protocells". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1863 (8): 148597. doi:10.1016/j.bbabio.2022.148597. PMID 35868450. S2CID 250707510.
- ^ Harrison, Stuart A.; Lane, Nick (12 December 2018). "Life as a guide to prebiotic nucleotide synthesis". Nature Communications. 9 (1): 5176. Bibcode:2018NatCo...9.5176H. doi:10.1038/s41467-018-07220-y. PMC 6289992. PMID 30538225.
- ^ Cantine, Marjorie D.; Fournier, Gregory P. (1 March 2018). "Environmental Adaptation from the Origin of Life to the Last Universal Common Ancestor". Origins of Life and Evolution of Biospheres. 48 (1): 35–54. Bibcode:2018OLEB...48...35C. doi:10.1007/s11084-017-9542-5. hdl:1721.1/114219. PMID 28685374. S2CID 254888920. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Mat, Wai-Kin (1 May 2008). "The genomics of LUCA" (PDF). Frontiers in Bioscience. 13 (14): 5605–5613. doi:10.2741/3103. PMID 18508609. Archived (PDF) from the original on 16 June 2022. Retrieved 8 December 2023.
- ^ Brasier, M. D. (2012). Secret Chambers: The Inside Story of Cells and Complex Life. Oxford University Press. p. 298.
- ^ Ward, Peter & Kirschvink, Joe, op cit, p. 42
- ^ a b Colín-García, M.; Heredia, A.; Cordero, G.; et al. (2016). "Hydrothermal vents and prebiotic chemistry: a review". Boletín de la Sociedad Geológica Mexicana. 68 (3): 599–620. doi:10.18268/BSGM2016v68n3a13. Archived from the original on 18 August 2017.
- ^ Schirber, Michael (24 June 2014). "Hydrothermal Vents Could Explain Chemical Precursors to Life". NASA Astrobiology: Life in the Universe. NASA. Archived from the original on 29 November 2014. Retrieved 19 June 2015.
- ^ a b Martin, William; Russell, Michael J. (29 January 2003). "On the origins of cells: a hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells". Philosophical Transactions of the Royal Society B. 358 (1429): 59–83, discussion 83–85. doi:10.1098/rstb.2002.1183. PMC 1693102. PMID 12594918.
- ^ Lane 2009
- ^ Usher, Oli (27 April 2015). "Chemistry of seabed's hot vents could explain emergence of life" (Press release). University College London. Archived from the original on 20 June 2015. Retrieved 19 June 2015.
- ^ Roldan, Alberto; Hollingsworth, Nathan; Roffey, Anna; et al. (May 2015). "Bio-inspired CO2 conversion by iron sulfide catalysts under sustainable conditions". Chemical Communications. 51 (35): 7501–7504. doi:10.1039/C5CC02078F. PMID 25835242. Archived from the original on 20 June 2015. Retrieved 19 June 2015.
- ^ Baross, J. A.; Hoffman, S. E. (1985). "Submarine hydrothermal vents and associated gradient environments as sites for the origin and evolution of life". Origins of Life and Evolution of Biospheres. 15 (4): 327–345. Bibcode:1985OrLi...15..327B. doi:10.1007/bf01808177. S2CID 4613918.
- ^ Russell, M. J.; Hall, A. J. (1997). "The emergence of life from iron monosulphide bubbles at a submarine hydrothermal redox and pH front". Journal of the Geological Society. 154 (3): 377–402. Bibcode:1997JGSoc.154..377R. doi:10.1144/gsjgs.154.3.0377. PMID 11541234. S2CID 24792282.
- ^ Amend, J. P.; LaRowe, D. E.; McCollom, T. M.; Shock, E. L. (2013). "The energetics of organic synthesis inside and outside the cell". Philosophical Transactions of the Royal Society B. 368 (1622): 20120255. doi:10.1098/rstb.2012.0255. PMC 3685458. PMID 23754809.
- ^ Shock, E. L.; Boyd, E. S. (2015). "Geomicrobiology and microbial geochemistry:principles of geobiochemistry". Elements. 11: 389–394. doi:10.2113/gselements.11.6.395.
- ^ Martin, W.; Russell, M. J. (2007). "On the origin of biochemistry at an alkaline hydrothermal vent". Philosophical Transactions of the Royal Society B. 362 (1486): 1887–1925. doi:10.1098/rstb.2006.1881. PMC 2442388. PMID 17255002.
- ^ Lane, Nick; Martin, William F. (21 December 2012). "The Origin of Membrane Bioenergetics". Cell. 151 (7): 1406–1416. doi:10.1016/j.cell.2012.11.050. PMID 23260134. S2CID 15028935.
- ^ Baaske, Philipp; Weinert, Franz M.; Duhr, Stefan; Lemke, Kono H.; Russell, Michael J.; Braun, Dieter (29 May 2007). "Extreme accumulation of nucleotides in simulated hydrothermal pore systems". Proceedings of the National Academy of Sciences. 104 (22): 9346–9351. doi:10.1073/pnas.0609592104. PMC 1890497. PMID 17494767.
- ^ Nunes Palmeira, Raquel; Colnaghi, Marco; Harrison, Stuart A.; Pomiankowski, Andrew; Lane, Nick (9 November 2022). "The limits of metabolic heredity in protocells". Proceedings of the Royal Society B: Biological Sciences. 289 (1986). doi:10.1098/rspb.2022.1469. PMC 9653231. PMID 36350219.
- ^ Woese, Carl R. (1987). "Bacterial evolution". Microbiological Reviews. 51.2 (1987) (2): 221–271. doi:10.1128/mr.51.2.221-271.1987. PMC 373105. PMID 2439888. S2CID 734579.
- ^ Russell, Michael J.; Hall, Allan J. (2006), "The onset and early evolution of life", Evolution of Early Earth's Atmosphere, Hydrosphere, and Biosphere - Constraints from Ore Deposits, Geological Society of America, doi:10.1130/2006.1198(01), ISBN 9780813711980, archived from the original on 31 January 2024, retrieved 30 November 2023
- ^ Boussau, Bastien; Blanquart, Samuel; Necsulea, Anamaria; Lartillot, Nicolas; Gouy, Manolo (December 2008). "Parallel adaptations to high temperatures in the Archaean eon". Nature. 456 (7224): 942–945. Bibcode:2008Natur.456..942B. doi:10.1038/nature07393. PMID 19037246. S2CID 4348746. Archived from the original on 16 December 2023. Retrieved 8 December 2023.
- ^ a b Galtier, Nicolas; Tourasse, Nicolas; Gouy, Manolo (8 January 1999). "A Nonhyperthermophilic Common Ancestor to Extant Life Forms". Science. 283 (5399): 220–221. doi:10.1126/science.283.5399.220. PMID 9880254. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Kelley, Deborah S.; Karson, Jeffrey A.; Blackman, Donna K.; Früh-Green, Gretchen L.; Butterfield, David A.; Lilley, Marvin D.; Olson, Eric J.; Schrenk, Matthew O.; Roe, Kevin K.; Lebon, Geoff T.; Rivizzigno, Pete (July 2001). "An off-axis hydrothermal vent field near the Mid-Atlantic Ridge at 30° N". Nature. 412 (6843): 145–149. Bibcode:2001Natur.412..145K. doi:10.1038/35084000. PMID 11449263. S2CID 4407013. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ a b Ehrenfreund, P.; Irvine, W.; Becker, L.; Blank, J.; Brucato, J. R.; et al. (August 2002). "Astrophysical and astrochemical insights into the origin of life". Reports on Progress in Physics. 65 (10): 1427. Bibcode:2002RPPh...65.1427E. doi:10.1088/0034-4885/65/10/202. S2CID 250904448.
- ^ Chyba, C.F.; Chyba, C.F.; Hand, K.P. (2006). "Comets and Prebiotic Organic Molecules on Early Earth". Comets and the Origin and Evolution of Life. Advances in Astrobiology and Biogeophysics. Springer Berlin Heidelberg. pp. 169–206. doi:10.1007/3-540-33088-7_6. ISBN 978-3-540-33086-8. Archived from the original on 31 January 2024. Retrieved 30 November 2023.
- ^ Chatterjee, Sankar (2023), Chatterjee, Sankar (ed.), "The Cradle of Life", From Stardust to First Cells: The Origin and Evolution of Early Life, Cham: Springer International Publishing, pp. 43–66, doi:10.1007/978-3-031-23397-5_6, ISBN 978-3-031-23397-5, archived from the original on 31 January 2024, retrieved 5 December 2023
- ^ Deamer, David W. (7 February 2019). "Prospects for Life on Other Planets". Assembling Life. Oxford University Press. doi:10.1093/oso/9780190646387.003.0017. ISBN 978-0-19-064638-7. Archived from the original on 31 January 2024. Retrieved 6 December 2023.
- ^ Pearce, Ben K. D.; Pudritz, Ralph E.; Semenov, Dmitry A.; Henning, Thomas K. (2 October 2017). "Origin of the RNA world: The fate of nucleobases in warm little ponds". Proceedings of the National Academy of Sciences. 114 (43): 11327–11332. arXiv:1710.00434. Bibcode:2017PNAS..11411327P. doi:10.1073/pnas.1710339114. PMC 5664528. PMID 28973920.
- ^ a b Deamer, David (10 February 2021). "Where Did Life Begin? Testing Ideas in Prebiotic Analogue Conditions". Life. 11 (2): 134. Bibcode:2021Life...11..134D. doi:10.3390/life11020134. PMC 7916457. PMID 33578711.
- ^ Korenaga, Jun (November 2021). "Was There Land on the Early Earth?". Life. 11 (11): 1142. Bibcode:2021Life...11.1142K. doi:10.3390/life11111142. PMC 8623345. PMID 34833018.
- ^ Powner, Matthew W.; Gerland, Béatrice; Sutherland, John D. (May 2009). "Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions". Nature. 459 (7244): 239–242. Bibcode:2009Natur.459..239P. doi:10.1038/nature08013. PMID 19444213. S2CID 4412117. Archived from the original on 12 November 2023. Retrieved 8 December 2023.
- ^ Zahnle, Kevin; Arndt, Nick; Cockell, Charles; Halliday, Alex; Nisbet, Euan; Selsis, Franck; Sleep, Norman H. (1 March 2007). "Emergence of a Habitable Planet". Space Science Reviews. 129 (1): 35–78. Bibcode:2007SSRv..129...35Z. doi:10.1007/s11214-007-9225-z. S2CID 12006144. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Woese, C R (June 1987). "Bacterial evolution". Microbiological Reviews. 51 (2): 221–271. doi:10.1128/mr.51.2.221-271.1987. PMC 373105. PMID 2439888.
- ^ a b c Mulkidjanian, Armen Y.; Bychkov, Andrew Yu.; Dibrova, Daria V.; Galperin, Michael Y.; Koonin, Eugene V. (3 April 2012). "Origin of first cells at terrestrial, anoxic geothermal fields". Proceedings of the National Academy of Sciences. 109 (14): E821-30. Bibcode:2012PNAS..109E.821M. doi:10.1073/pnas.1117774109. PMC 3325685. PMID 22331915.
- ^ Chandru, Kuhan; Guttenberg, Nicholas; Giri, Chaitanya; et al. (31 May 2018). "Simple prebiotic synthesis of high diversity dynamic combinatorial polyester libraries". Communications Chemistry. 1 (1). doi:10.1038/s42004-018-0031-1.
- ^ Forsythe, Jay G.; Yu, Sheng-Sheng; Mamajanov, Irena; et al. (17 August 2015). "Ester-Mediated Amide Bond Formation Driven by Wet–Dry Cycles: A Possible Path to Polypeptides on the Prebiotic Earth". Angewandte Chemie International Edition in English. 54 (34): 9871–9875. doi:10.1002/anie.201503792. PMC 4678426. PMID 26201989.
- ^ Patel, Bhavesh H.; Percivalle, Claudia; Ritson, Dougal J.; Duffy, Colm. D.; Sutherland, John D. (16 March 2015). "Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism". Nature Chemistry. 7 (4): 301–307. Bibcode:2015NatCh...7..301P. doi:10.1038/nchem.2202. PMC 4568310. PMID 25803468.
- ^ Catling, David C.; Zahnle, Kevin J. (28 February 2020). "The Archean atmosphere". Science Advances. 6 (9): eaax1420. Bibcode:2020SciA....6.1420C. doi:10.1126/sciadv.aax1420. PMC 7043912. PMID 32133393.
- ^ Miller, Stanley L.; Lazcano, Antonio (December 1995). "The origin of life?did it occur at high temperatures?". Journal of Molecular Evolution. 41 (6): 689–692. Bibcode:1995JMolE..41..689M. doi:10.1007/bf00173146. hdl:2060/19980211388. PMID 11539558. S2CID 25141419. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Forterre, Patrick; Bergerat, Agnes; Lopex-Garcia, Purificacion (May 1996). "The unique DNA topology and DNA topoisomerases of hyperthermophilic archaea". FEMS Microbiology Reviews. 18 (2–3): 237–248. doi:10.1111/j.1574-6976.1996.tb00240.x. PMID 8639331. S2CID 6001830.
- ^ Brochier-Armanet, Céline; Forterre, Patrick (May 2006). "Widespread distribution of archaeal reverse gyrase in thermophilic bacteria suggests a complex history of vertical inheritance and lateral gene transfers". Archaea. 2 (2): 83–93. doi:10.1155/2006/582916. PMC 2686386. PMID 17350929.
- ^ Feulner, Georg (June 2012). "The faint young Sun problem". Reviews of Geophysics. 50 (2). arXiv:1204.4449. Bibcode:2012RvGeo..50.2006F. doi:10.1029/2011RG000375. S2CID 119248267. Archived from the original on 8 December 2023. Retrieved 8 December 2023.
- ^ Bada, J. L.; Bigham, C.; Miller, S. L. (15 February 1994). "Impact melting of frozen oceans on the early Earth: Implications for the origin of life". Proceedings of the National Academy of Sciences. 91 (4): 1248–1250. Bibcode:1994PNAS...91.1248B. doi:10.1073/pnas.91.4.1248. PMC 43134. PMID 11539550.
- ^ Monnard, Pierre-Alain; Apel, Charles L.; Kanavarioti, Anastassia; Deamer, David W. (June 2002). "Influence of Ionic Inorganic Solutes on Self-Assembly and Polymerization Processes Related to Early Forms of Life: Implications for a Prebiotic Aqueous Medium". Astrobiology. 2 (2): 139–152. Bibcode:2002AsBio...2..139M. doi:10.1089/15311070260192237. PMID 12469365. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Attwater, James; Wochner, Aniela; Holliger, Philipp (December 2013). "In-ice evolution of RNA polymerase ribozyme activity". Nature Chemistry. 5 (12): 1011–1018. Bibcode:2013NatCh...5.1011A. doi:10.1038/nchem.1781. PMC 3920166. PMID 24256864.
- ^ Levy, Matthew; Miller, Stanley L.; Brinton, Karen; Bada, Jeffrey L. (1 June 2000). "Prebiotic Synthesis of Adenine and Amino Acids Under Europa-like Conditions". Icarus. 145 (2): 609–613. Bibcode:2000Icar..145..609L. doi:10.1006/icar.2000.6365. PMID 11543508.
- ^ Moulton, Vincent; Gardner, Paul P.; Pointon, Robert F.; Creamer, Lawrence K.; Jameson, Geoffrey B.; Penny, David (1 October 2000). "RNA Folding Argues Against a Hot-Start Origin of Life". Journal of Molecular Evolution. 51 (4): 416–421. Bibcode:2000JMolE..51..416M. doi:10.1007/s002390010104. PMID 11040293. S2CID 20787323. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Zemora, Georgeta; Waldsich, Christina (November 2010). "RNA folding in living cells". RNA Biology. 7 (6): 634–641. doi:10.4161/rna.7.6.13554. PMC 3073324. PMID 21045541. Archived from the original on 31 January 2024. Retrieved 8 December 2023.
- ^ Schreiber, Ulrich; Locker-Grütjen, Oliver; Mayer, Christian (2012). "Hypothesis: Origin of Life in Deep-Reaching Tectonic Faults". Origins of Life and Evolution of Biospheres. 42 (1): 47–54. Bibcode:2012OLEB...42...47S. doi:10.1007/s11084-012-9267-4. ISSN 0169-6149. PMID 22373604.
- ^ Schreiber, Ulrich; Mayer, Christian; Schmitz, Oliver J.; Rosendahl, Pia; Bronja, Amela; Greule, Markus; Keppler, Frank; Mulder, Ines; Sattler, Tobias; Schöler, Heinz F. (14 June 2017). Stüeken, Eva Elisabeth (ed.). "Organic compounds in fluid inclusions of Archean quartz—Analogues of prebiotic chemistry on early Earth". PLOS ONE. 12 (6): e0177570. Bibcode:2017PLoSO..1277570S. doi:10.1371/journal.pone.0177570. ISSN 1932-6203. PMC 5470662. PMID 28614348.
- ^ Großmann, Yildiz; Schreiber, Ulrich; Mayer, Christian; Schmitz, Oliver J. (21 June 2022). "Aliphatic Aldehydes in the Earth's Crust—Remains of Prebiotic Chemistry?". Life. 12 (7): 925. Bibcode:2022Life...12..925G. doi:10.3390/life12070925. ISSN 2075-1729. PMC 9319801. PMID 35888015.
- ^ Mayer, Christian; Schreiber, Ulrich; Dávila, María (7 January 2017). "Selection of Prebiotic Molecules in Amphiphilic Environments". Life. 7 (1): 3. Bibcode:2017Life....7....3M. doi:10.3390/life7010003. ISSN 2075-1729. PMC 5370403. PMID 28067845.
- ^ Mayer, Christian; Schreiber, Ulrich; Dávila, María J. (2015). "Periodic Vesicle Formation in Tectonic Fault Zones—an Ideal Scenario for Molecular Evolution". Origins of Life and Evolution of Biospheres. 45 (1–2): 139–148. Bibcode:2015OLEB...45..139M. doi:10.1007/s11084-015-9411-z. ISSN 0169-6149. PMC 4457167. PMID 25716918.
- ^ Mayer, Christian; Schreiber, Ulrich; Dávila, María; Schmitz, Oliver; Bronja, Amela; Meyer, Martin; Klein, Julia; Meckelmann, Sven (24 May 2018). "Molecular Evolution in a Peptide-Vesicle System". Life. 8 (2): 16. Bibcode:2018Life....8...16M. doi:10.3390/life8020016. ISSN 2075-1729. PMC 6027363. PMID 29795023.
- ^ a b Plasson, Raphaël; Kondepudi, Dilip K.; Bersini, Hugues; et al. (August 2007). "Emergence of homochirality in far-from-equilibrium systems: Mechanisms and role in prebiotic chemistry". Chirality. 19 (8): 589–600. doi:10.1002/chir.20440. PMID 17559107. "Special Issue: Proceedings from the Eighteenth International Symposium on Chirality (ISCD-18), Busan, Korea, 2006"
- ^ Chaichian, Rojas & Tureanu 2014, pp. 353–364
- ^ Jafarpour, Farshid; Biancalani, Tommaso; Goldenfeld, Nigel (2017). "Noise-induced symmetry breaking far from equilibrium and the emergence of biological homochirality" (PDF). Physical Review E. 95 (3): 032407. Bibcode:2017PhRvE..95c2407J. doi:10.1103/PhysRevE.95.032407. PMID 28415353. Archived from the original on 2 April 2023. Retrieved 29 August 2019.
- ^ Jafarpour, Farshid; Biancalani, Tommaso; Goldenfeld, Nigel (2015). "Noise-induced mechanism for biological homochirality of early life self-replicators". Physical Review Letters. 115 (15): 158101. arXiv:1507.00044. Bibcode:2015PhRvL.115o8101J. doi:10.1103/PhysRevLett.115.158101. PMID 26550754. S2CID 9775791.
- ^ Frank, F. C. (1953). "On spontaneous asymmetric synthesis". Biochimica et Biophysica Acta. 11 (4): 459–463. doi:10.1016/0006-3002(53)90082-1. PMID 13105666.
- ^ Clark, Stuart (July–August 1999). "Polarized Starlight and the Handedness of Life". American Scientist. 87 (4): 336. Bibcode:1999AmSci..87..336C. doi:10.1511/1999.4.336. S2CID 221585816.
- ^ Shibata, Takanori; Morioka, Hiroshi; Hayase, Tadakatsu; et al. (17 January 1996). "Highly Enantioselective Catalytic Asymmetric Automultiplication of Chiral Pyrimidyl Alcohol". Journal of the American Chemical Society. 118 (2): 471–472. doi:10.1021/ja953066g.
- ^ Soai, Kenso; Sato, Itaru; Shibata, Takanori (2001). "Asymmetric autocatalysis and the origin of chiral homogeneity in organic compounds". The Chemical Record. 1 (4): 321–332. doi:10.1002/tcr.1017. PMID 11893072.
- ^ Hazen 2005, p. 184
- ^ Meierhenrich, Uwe (2008). Amino acids and the asymmetry of life caught in the act of formation. Berlin: Springer. pp. 76–79. ISBN 978-3-540-76886-9.
- ^ Mullen, Leslie (5 September 2005). "Building Life from Star-Stuff". Astrobiology Magazine. Archived from the original on 14 July 2015.
{{cite journal}}: CS1 maint: unfit URL (link) - ^ Root-Bernstein, Robert (23 June 2010). "Experimental Test of L- and D-Amino Acid Binding to L- and D-Codons Suggests that Homochirality and Codon Directionality Emerged with the Genetic Code". Symmetry. 2 (2): 1180–1200. Bibcode:2010Symm....2.1180R. doi:10.3390/sym2021180.
Sources[edit]
- Altermann, Wladyslaw (2009). "Introduction: A Roadmap to Fata Morgana?". In Seckbach, Joseph; Walsh, Maud (eds.). From Fossils to Astrobiology: Records of Life on Earth and the Search for Extraterrestrial Biosignatures. Cellular Origin, Life in Extreme Habitats and Astrobiology. Vol. 12. Dordrecht, the Netherlands; London: Springer. ISBN 978-1-4020-8836-0.
- Bada, Jeffrey L.; Lazcano, Antonio (2009). "The Origin of Life". In Ruse, Michael; Travis, Joseph (eds.). Evolution: The First Four Billion Years. Foreword by Edward O. Wilson. Cambridge: Belknap Press of Harvard University Press. ISBN 978-0-674-03175-3. OCLC 225874308.
- Barton, Nicholas H.; Briggs, Derek E.G.; Eisen, Jonathan A.; et al. (2007). Evolution. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press. ISBN 978-0-87969-684-9. OCLC 86090399.
- Bastian, H. Charlton (1871). The Modes of Origin of Lowest Organisms. London; New York: Macmillan and Company. OCLC 42959303. Retrieved 6 June 2015.
- Bernal, J. D. (1951). The Physical Basis of Life. London: Routledge & Kegan Paul.
- Bernal, J. D. (1960). "The Problem of Stages in Biopoesis". In Florkin, M. (ed.). Aspects of the Origin of Life. International Series of Monographs on Pure and Applied Biology. Oxford, UK; New York: Pergamon Press. ISBN 978-1-4831-3587-8.
- Bernal, J. D. (1967) [Reprinted work by A.I. Oparin originally published 1924; Moscow: The Moscow Worker]. The Origin of Life. The Weidenfeld and Nicolson Natural History. Translation of Oparin by Ann Synge. London: Weidenfeld & Nicolson.
- Bock, Gregory R.; Goode, Jamie A., eds. (1996). Evolution of Hydrothermal Ecosystems on Earth (and Mars?). Ciba Foundation Symposium. Vol. 202. Chichester, UK; New York: John Wiley & Sons. ISBN 978-0-471-96509-1.
- Bondeson, Jan (1999). The Feejee Mermaid and Other Essays in Natural and Unnatural History. Ithaca, NY: Cornell University Press. ISBN 978-0-8014-3609-3.
- Bryson, Bill (2004). A Short History of Nearly Everything. London: Black Swan. ISBN 978-0-552-99704-1. OCLC 55589795.
- Calvin, Melvin (1969). Chemical Evolution: Molecular Evolution Towards the Origin of Living Systems on the Earth and Elsewhere. Oxford, UK: Clarendon Press. ISBN 978-0-19-855342-7. OCLC 25220.
- Chaichian, Masud; Rojas, Hugo Perez; Tureanu, Anca (2014). "Physics and Life". Basic Concepts in Physics. Undergraduate Lecture Notes in Physics. Berlin; Heidelberg: Springer Berlin Heidelberg. pp. 353–364. doi:10.1007/978-3-642-19598-3_12. ISBN 978-3-642-19597-6. OCLC 900189038. S2CID 115247432.
- Chang, Thomas Ming Swi (2007). Artificial Cells: Biotechnology, Nanomedicine, Regenerative Medicine, Blood Substitutes, Bioencapsulation, and Cell/Stem Cell Therapy. Regenerative Medicine, Artificial Cells and Nanomedicine. Vol. 1. Hackensack, New Jersey: World Scientific. ISBN 978-981-270-576-1. OCLC 173522612.
- Dalrymple, G. Brent (2001). "The age of the Earth in the twentieth century: a problem (mostly) solved". In Lewis, C.L.E.; Knell, S.J. (eds.). The Age of the Earth: from 4004 BC to AD 2002. Geological Society Special Publication. Vol. 190. London: Geological Society of London. pp. 205–221. Bibcode:2001GSLSP.190..205D. doi:10.1144/gsl.sp.2001.190.01.14. ISBN 978-1-86239-093-5. OCLC 48570033. S2CID 130092094.
- Darwin, Charles (1887). Darwin, Francis (ed.). The Life and Letters of Charles Darwin, Including an Autobiographical Chapter. Vol. 3 (3rd ed.). London: John Murray. OCLC 834491774.
- Davies, Paul (1999). The Fifth Miracle: The Search for the Origin of Life. London: Penguin Books. ISBN 978-0-14-028226-9.
- Dobell, Clifford (1960) [Originally published 1932; New York: Harcourt, Brace & Company]. Antony van Leeuwenhoek and His 'Little Animals'. New York: Dover Publications.
- Dyson, Freeman (1999). Origins of Life (Revised ed.). Cambridge, UK; New York: Cambridge University Press. ISBN 978-0-521-62668-2.
- Fesenkov, V. G. (1959). "Some Considerations about the Primaeval State of the Earth". In Oparin, A. I.; et al. (eds.). The Origin of Life on the Earth. I.U.B. Symposium Series. Vol. 1. Edited for the International Union of Biochemistry by Frank Clark and R.L.M. Synge (English-French-German ed.). London; New York: Pergamon Press. ISBN 978-1-4832-2240-0. International Symposium on the Origin of Life on the Earth (held at Moscow, 19–24 August 1957)
- Hazen, Robert M. (2005). Genesis: The Scientific Quest for Life's Origin. Washington, DC: Joseph Henry Press. ISBN 978-0-309-09432-0. OCLC 60321860.
- Huxley, Thomas Henry (1968) [1897]. "VIII Biogenesis and Abiogenesis [1870]". Discourses, Biological and Geological. Collected Essays. Vol. VIII (Reprint ed.). New York: Greenwood Press. OCLC 476737627. Archived from the original on 7 May 2015. Retrieved 19 May 2014.
- Klyce, Brig (22 January 2001). Kingsley, Stuart A.; Bhathal, Ragbir (eds.). Panspermia Asks New Questions. The Search for Extraterrestrial Intelligence (SETI) in the Optical Spectrum III. Vol. 4273. Bellingham, WA: SPIE. doi:10.1117/12.435366. ISBN 0-8194-3951-7. Archived from the original on 3 September 2013. Retrieved 9 June 2015. Proceedings of the SPIE held at San Jose, California, 22–24 January 2001
- Lane, Nick (2009). Life Ascending: The 10 Great Inventions of Evolution (1st American ed.). New York: W.W. Norton & Company. ISBN 978-0-393-06596-1. OCLC 286488326.
- Lennox, James G. (2001). Aristotle's Philosophy of Biology: Studies in the Origins of Life Science. Cambridge Studies in Philosophy and Biology. Cambridge, UK; New York: Cambridge University Press. ISBN 978-0-521-65976-5.
- Michod, Richard E. (1999). Darwinian Dynamics: Evolutionary Transitions in Fitness and Individuality. Princeton, New Jersey: Princeton University Press. ISBN 978-0-691-02699-2. OCLC 38948118.
- Oparin, A.I. (1953) [Originally published 1938; New York: The Macmillan Company]. The Origin of Life. Translation and new introduction by Sergius Morgulis (2nd ed.). Mineola, NY: Dover Publications. ISBN 978-0-486-49522-4.
- Ross, Alexander (1652). Arcana Microcosmi. Vol. II. London. OCLC 614453394. Archived from the original on 24 February 2024. Retrieved 14 July 2015.
- Sheldon, Robert B. (22 September 2005). "Historical Development of the Distinction between Bio- and Abiogenesis" (PDF). In Hoover, Richard B.; Levin, Gilbert V.; Rozanov, Alexei Y.; Gladstone, G. Randall (eds.). Astrobiology and Planetary Missions. Astrobiology and Planetary Missions. Vol. 5906. Bellingham, WA: SPIE. pp. 59061I. doi:10.1117/12.663480. ISBN 978-0-8194-5911-4. Archived (PDF) from the original on 13 April 2015. Retrieved 13 April 2015. Proceedings of the SPIE held at San Diego, California, 31 July–2 August 2005
- Tyndall, John (1905) [Originally published 1871; London; New York: Longmans, Green & Co.; D. Appleton and Company]. Fragments of Science. Vol. 2 (6th ed.). New York: P.F. Collier & Sons. OCLC 726998155.
- Voet, Donald; Voet, Judith G. (2004). Biochemistry. Vol. 1 (3rd ed.). New York: John Wiley & Sons. ISBN 978-0-471-19350-0.
- Yarus, Michael (2010). Life from an RNA World: The Ancestor Within. Cambridge, Massachusetts: Harvard University Press. p. 287. ISBN 978-0-674-05075-4.
External links[edit]
- Making headway with the mysteries of life's origins – Adam Mann (PNAS; 14 April 2021)
- Exploring Life's Origins Archived 8 April 2023 at the Wayback Machine a virtual exhibit at the Museum of Science (Boston)
- How life began on Earth – Marcia Malory (Earth Facts; 2015)
- The Origins of Life – Richard Dawkins et al. (BBC Radio; 2004)
- Life in the Universe – Essay by Stephen Hawking (1996)



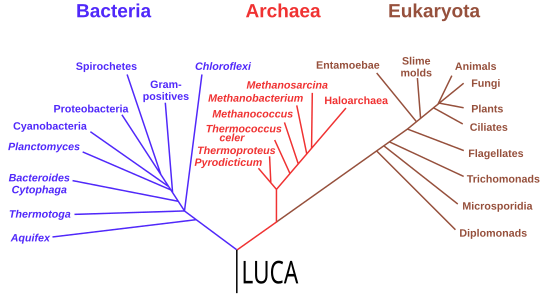
![LUCA systems and environment included the Wood–Ljungdahl pathway.[10]](http://upload.wikimedia.org/wikipedia/commons/thumb/e/ed/LUCA_systems_and_environment.svg/622px-LUCA_systems_and_environment.svg.png)
