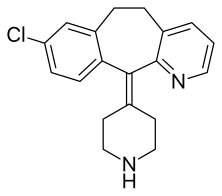
Desloratadine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Clarinex, Aerius, Allex, others[1] |
| Other names | descarboethoxyloratadine[2] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602002 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Rapidly absorbed |
| Protein binding | 83 to 87% |
| Metabolism | UGT2B10, CYP2C8 |
| Metabolites | 3-Hydroxydesloratadine |
| Onset of action | within 1 hour[6] |
| Elimination half-life | 27 hours,[6] 33.7 hours in elderly patients[3] |
| Duration of action | up to 24 hours[6] |
| Excretion | 40% as conjugated metabolites into urine Similar amount into the feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.166.554 |
| Chemical and physical data | |
| Formula | C19H19ClN2 |
| Molar mass | 310.83 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Desloratadine sold under the brand name Clarinex among others, is a tricyclic H1 inverse agonist that is used to treat allergies. It is an active metabolite of loratadine.[6]
It was patented in 1984 and came into medical use in 2001.[7] It was brought to the market in the US by Schering Corporation, later named Schering-Plough.[3]
Medical uses
[edit]Desloratadine is used to treat allergic rhinitis, nasal congestion and chronic idiopathic urticaria (hives).[8] It is the major metabolite of loratadine and the two drugs are similar in safety and effectiveness.[8] Desloratadine is available in many dosage forms and under many brand names worldwide.[9]
An emerging indication for desloratadine is in the treatment of acne, as an inexpensive adjuvant to isotretinoin and possibly as maintenance therapy or monotherapy.[10][11]
Side effects
[edit]The most common side-effects are fatigue (1.2%[12]), dry mouth (3%[12]), and headache (0.6%[12]).[8]
Interactions
[edit]Co-administration with erythromycin, ketoconazole, azithromycin, fluoxetine, or cimetidine resulted in elevated blood plasma concentrations of desloratadine and its metabolite 3-hydroxydesloratadine in studies. However, no clinically relevant changes were observed.[3][13]
Pharmacology
[edit]Pharmacodynamics
[edit]Desloratadine is a selective H1-antihistamine which functions as an inverse agonist at the histamine H1 receptor.[14]
At very high doses, is also an antagonist at various subtypes of the muscarinic acetylcholine receptors. This effect is not relevant for the drug's action at therapeutic doses.[15]
Pharmacokinetics
[edit]Desloratadine is well absorbed from the gut and reaches highest blood plasma concentrations after about three hours. In the bloodstream, 83 to 87% of the substance are bound to plasma proteins.[13]
Desloratadine is metabolized to 3-hydroxydesloratadine in a three-step sequence in normal metabolizers. First, N-glucuronidation of desloratadine by UGT2B10; then, 3-hydroxylation of desloratadine N-glucuronide by CYP2C8; and finally, a non-enzymatic deconjugation of 3-hydroxydesloratadine N-glucuronide.[16][17] Both desloratadine and 3-hydroxydesloratadine are eliminated via urine and feces with a half-life of 27 hours in normal metabolizers.[13][18]

It exhibits only peripheral activity since it does not readily cross the blood–brain barrier; hence, it does not normally cause drowsiness because it does not readily enter the central nervous system.[19]
Desloratadine does not have a strong effect on a number of tested enzymes in the cytochrome P450 system. It was found to weakly inhibit CYP2B6, CYP2D6, and CYP3A4/CYP3A5, and not to inhibit CYP1A2, CYP2C8, CYP2C9, or CYP2C19. Desloratadine was found to be a potent and relatively selective inhibitor of UGT2B10, a weak to moderate inhibitor of UGT2B17, UGT1A10, and UGT2B4, and not to inhibit UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, UGT2B15, UGT1A7, and UGT1A8.[17]
Pharmacogenomics
[edit]2% of Caucasian people and 18% of people from African descent are desloratadine poor metabolizers. In these people, the drug reaches threefold higher plasma concentrations at seven hours after intake, and it has a half-life of 89 hours (compared to a 27-hour half-life in normal metabolizers). Adverse effects were reported at similar rates in poor metabolizers, suggesting that it is not clinically relevant.[13][18]
References
[edit]- ^ Murdoch D, Goa KL, Keam SJ (7 April 2003). "Desloratadine: an update of its efficacy in the management of allergic disorders". Drugs. 63 (19): 2051–2077. doi:10.2165/00003495-200363190-00010. PMID 12962522. S2CID 195689362.
- ^ Schering Corporation (2000). "CLARITIN brand of Loratadine - Full Prescribing Information (US FDA)" (PDF). US FDA. Retrieved 17 May 2024.
loratadine is metabolized to descarboethoxyloratadine predominantly by cytochrome P450 3A4 (CYP3A4) and, to a lesser extent, by cytochrome P450 2D6 (CYP2D6).
- ^ a b c d "Clarinex- desloratadine tablet, film coated". DailyMed. 14 November 2022. Retrieved 18 May 2024.
- ^ "Clarinex-D 12 HOUR- desloratadine and pseudoephedrine sulfate tablet, extended release". DailyMed. 14 November 2022. Retrieved 18 May 2024.
- ^ "Allex EPAR". European Medicines Agency (EMA). 19 May 2004.
- ^ a b c d Lieberman P, Hernandez-Trujillo V, Lieberman J, Frew AJ (2008). "Antihistamines". Clinical Immunology. Elsevier. pp. 1317–1329. doi:10.1016/b978-0-323-04404-2.10089-2. ISBN 9780323044042.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 549. ISBN 9783527607495.
- ^ a b c See S (November 2003). "Desloratadine for allergic rhinitis". American Family Physician. 68 (10): 2015–2016. PMID 14655812. Archived from the original on 24 July 2005. Retrieved 1 August 2005.
- ^ "Desloratadine". Drugs.com. Retrieved 4 May 2015.
- ^ Lee HE, Chang IK, Lee Y, Kim CD, Seo YJ, Lee JH, et al. (December 2014). "Effect of antihistamine as an adjuvant treatment of isotretinoin in acne: a randomized, controlled comparative study". Journal of the European Academy of Dermatology and Venereology. 28 (12): 1654–1660. doi:10.1111/jdv.12403. PMID 25081735. S2CID 3406128.
- ^ Layton AM (April 2016). "Top Ten List of Clinical Pearls in the Treatment of Acne Vulgaris". Dermatologic Clinics. 34 (2): 147–157. doi:10.1016/j.det.2015.11.008. PMID 27015774.
- ^ a b c González-Núñez V, Valero A, Mullol J (May 2013). "Safety evaluation of desloratadine in allergic rhinitis". Expert Opinion on Drug Safety. 12 (3). Informa Healthcare: 445–453. doi:10.1517/14740338.2013.788148. PMID 23574541. S2CID 40472187.
- ^ a b c d "Aerius: EPAR – Product Information" (PDF). European Medicines Agency. Archived from the original (PDF) on 5 July 2019. Retrieved 21 January 2022.
- ^ Canonica GW, Blaiss M (February 2011). "Antihistaminic, anti-inflammatory, and antiallergic properties of the nonsedating second-generation antihistamine desloratadine: a review of the evidence". The World Allergy Organization Journal. 4 (2): 47–53. doi:10.1097/WOX.0b013e3182093e19. PMC 3500039. PMID 23268457.
- ^ "Aerius: EPAR – Scientific Discussion" (PDF). European Medicines Agency. 3 April 2006. Archived from the original (PDF) on 16 March 2018. Retrieved 13 October 2017.
- ^ Kazmi F, Barbara JE, Yerino P, Parkinson A (April 2015). "A long-standing mystery solved: the formation of 3-hydroxydesloratadine is catalyzed by CYP2C8 but prior glucuronidation of desloratadine by UDP-glucuronosyltransferase 2B10 is an obligatory requirement". Drug Metabolism and Disposition. 43 (4): 523–533. doi:10.1124/dmd.114.062620. PMID 25595597.
- ^ a b Kazmi F, Yerino P, Barbara JE, Parkinson A (September 2015). "Further Characterization of the Metabolism of Desloratadine and Its Cytochrome P450 and UDP-glucuronosyltransferase Inhibition Potential: Identification of Desloratadine as a Relatively Selective UGT2B10 Inhibitor". Drug Metabolism and Disposition. 43 (9): 1294–1302. doi:10.1124/dmd.115.065011. PMID 26135009.
- ^ a b Drugs.com: Desloratadine Monograph.
- ^ Mann RD, Pearce GL, Dunn N, Shakir S (April 2000). "Sedation with "non-sedating" antihistamines: four prescription-event monitoring studies in general practice". BMJ. 320 (7243): 1184–1186. doi:10.1136/bmj.320.7243.1184. PMC 27362. PMID 10784544.
