Hsp70
| Hsp70 protein | |||||||||
|---|---|---|---|---|---|---|---|---|---|
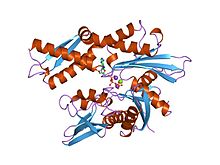 Structure of the ATPase fragment of a 70K heat-shock cognate protein.[1] | |||||||||
| Identifiers | |||||||||
| Symbol | HSP70 | ||||||||
| Pfam | PF00012 | ||||||||
| Pfam clan | CL0108 | ||||||||
| InterPro | IPR013126 | ||||||||
| PROSITE | PDOC00269 | ||||||||
| SCOP2 | 3hsc / SCOPe / SUPFAM | ||||||||
| |||||||||

The 70 kilodalton heat shock proteins (Hsp70s or DnaK) are a family of conserved ubiquitously expressed heat shock proteins. Proteins with similar structure exist in virtually all living organisms. Intracellularly localized Hsp70s are an important part of the cell's machinery for protein folding, performing chaperoning functions, and helping to protect cells from the adverse effects of physiological stresses.[2][3] Additionally, membrane-bound Hsp70s have been identified as a potential target for cancer therapies[4] and their extracellularly localized counterparts have been identified as having both membrane-bound and membrane-free structures.[5]
Discovery
[edit]Members of the Hsp70 family are very strongly upregulated by heat stress and toxic chemicals, particularly heavy metals such as arsenic, cadmium, copper, mercury, etc. Heat shock was originally discovered by Ferruccio Ritossa in the 1960s when a lab worker accidentally boosted the incubation temperature of Drosophila (fruit flies). When examining the chromosomes, Ritossa found a "puffing pattern" that indicated the elevated gene transcription of an unknown protein.[6][7] This was later described as the "Heat Shock Response" and the proteins were termed the "Heat Shock Proteins" (Hsps).
Structure
[edit]
The Hsp70 proteins have three major functional domains:
- N-terminal ATPase domain – binds ATP (Adenosine triphosphate) and hydrolyzes it to ADP (Adenosine diphosphate). The NBD (nucleotide binding domain) consists of two lobes with a deep cleft between them, at the bottom of which nucleotide (ATP and ADP) binds. The exchange of ATP and ADP leads to conformational changes in the other two domains.
- Substrate binding domain – is composed of a 15 kDa β sheet subdomain and a 10 kDa helical subdomain. The β sheet subdomain consists of stranded β sheets with upward protruding loops, as a typical β barrel, which enclose the peptide backbone of the substrate. SBD contains a groove with an affinity for neutral, hydrophobic amino acid residues. The groove is long enough to interact with peptides up to seven residues in length.
- C-terminal domain – rich in alpha helical structure acts as a 'lid' for the substrate binding domain. The helical subdomain consists of five helices, with two helices packed against two sides of the β sheet subdomain, stabilizing the inner structure. In addition, one of the helix forms a salt bridge and several hydrogen bonds to the outer Loops, thereby closing the substrate-binding pocket like a lid. Three helices in this domain form another hydrophobic core which may be stabilization of the "lid". When an Hsp70 protein is ATP bound, the lid is open and peptides bind and release relatively rapidly. When Hsp70 proteins are ADP bound, the lid is closed, and peptides are tightly bound to the substrate binding domain.[9]

Protein phosphorylation, a post-translational modification, helps to regulate protein function and involves the phosphorylation of amino acids with hydroxyl groups in their side chains (among eukaryotes). Serine, threonine, and tyrosine amino acids are common targets of phosphorylation. Phosphorylation of Hsp70 has become a point of greater exploration in scientific literature relatively recently. A 2020 publication suggests that phosphorylation of a serine residue between the NBD and substrate binding domain in yeast Hsp70s leads to a dramatic reduction of the normal Hsp70 heat shock response.[10] This deactivation via phosphorylation of a protein is a common motif in protein regulation, and demonstrates how relatively small changes to protein structure can have biologically significant effects on protein function.
Function
[edit]The Hsp70 system interacts with extended peptide segments of proteins as well as partially folded proteins to cause aggregation of proteins in key pathways to downregulate activity.[11][8] When not interacting with a substrate peptide, Hsp70 is usually in an ATP bound state. Hsp70 by itself is characterized by a very weak ATPase activity, such that spontaneous hydrolysis will not occur for many minutes. As newly synthesized proteins emerge from the ribosomes, the substrate binding domain of Hsp70 recognizes sequences of hydrophobic amino acid residues, and interacts with them. This spontaneous interaction is reversible, and in the ATP bound state Hsp70 may relatively freely bind and release peptides. However, the presence of a peptide in the binding domain stimulates the ATPase activity of Hsp70, increasing its normally slow rate of ATP hydrolysis. When ATP is hydrolyzed to ADP the binding pocket of Hsp70 closes, tightly binding the now-trapped peptide chain. Further speeding ATP hydrolysis are the so-called J-domain cochaperones: primarily Hsp40 in eukaryotes, and DnaJ in prokaryotes. These cochaperones dramatically increase the ATPase activity of Hsp70 in the presence of interacting peptides.

By binding tightly to partially synthesized peptide sequences (incomplete proteins), Hsp70 prevents them from aggregating and being rendered nonfunctional. Once the entire protein is synthesized, a nucleotide exchange factor (prokaryotic GrpE, eukaryotic BAG1 and HspBP1 are among those which have been identified) stimulates the release of ADP and binding of fresh ATP, opening the binding pocket. The protein is then free to fold on its own, or to be transferred to other chaperones for further processing.[12] HOP (the Hsp70/Hsp90 Organizing Protein) can bind to both Hsp70 and Hsp90 at the same time, and mediates the transfer of peptides from Hsp70 to Hsp90.[13]
Hsp70 also aids in transmembrane transport of proteins, by stabilizing them in a partially folded state. It is also known to be phosphorylated[14] which regulates several of its functions.[15][16][17]
Hsp70 proteins can act to protect cells from thermal or oxidative stress. These stresses normally act to damage proteins, causing partial unfolding and possible aggregation. By temporarily binding to hydrophobic residues exposed by stress, Hsp70 prevents these partially denatured proteins from aggregating, and inhibits them from refolding. Low ATP is characteristic of heat shock and sustained binding is seen as aggregation suppression, while recovery from heat shock involves substrate binding and nucleotide cycling. In a thermophile anaerobe (Thermotoga maritima) the Hsp70 demonstrates redox sensitive binding to model peptides, suggesting a second mode of binding regulation based on oxidative stress.
Hsp70 seems to be able to participate in disposal of damaged or defective proteins. Interaction with CHIP (Carboxyl-terminus of Hsp70 Interacting Protein)–an E3 ubiquitin ligase–allows Hsp70 to pass proteins to the cell's ubiquitination and proteolysis pathways.[18]
Finally, in addition to improving overall protein integrity, Hsp70 directly inhibits apoptosis.[19] One hallmark of apoptosis is the release of cytochrome c, which then recruits Apaf-1 and dATP/ATP into an apoptosome complex. This complex then cleaves procaspase-9, activating caspase-9 and eventually inducing apoptosis via caspase 3 activation. Hsp70 inhibits this process by blocking the recruitment of procaspase-9 to the Apaf-1/dATP/cytochrome c apoptosome complex. It does not bind directly to the procaspase-9 binding site, but likely induces a conformational change that renders procaspase-9 binding less favorable. Hsp70 is shown to interact with Endoplasmic reticulum stress sensor protein IRE1alpha thereby protecting the cells from ER stress - induced apoptosis. This interaction prolonged the splicing of XBP-1 mRNA thereby inducing transcriptional upregulation of targets of spliced XBP-1 like EDEM1, ERdj4 and P58IPK rescuing the cells from apoptosis.[20] Other studies suggest that Hsp70 may play an anti-apoptotic role at other steps, but is not involved in Fas-ligand-mediated apoptosis (although Hsp 27 is). Therefore, Hsp70 not only saves important components of the cell (the proteins) but also directly saves the cell as a whole. Considering that stress-response proteins (like Hsp70) evolved before apoptotic machinery, Hsp70's direct role in inhibiting apoptosis provides an interesting evolutionary picture of how more recent (apoptotic) machinery accommodated previous machinery (Hsps), thus aligning the improved integrity of a cell's proteins with the improved chances of that particular cell's survival.
In mice, exogenous recombinant human Hsp70 (eHsp70), delivered intranasally, increases lifespan. Although the maximum lifespan increased only moderately, the overall mortality rate in treated animals was much lower compared with the control group. Also this eHsp70-treatment improves learning and memory of mice in old age, increases their curiosity.[21]
Cancer
[edit]Hsp70 is overexpressed in malignant melanoma[22] and underexpressed in renal cell cancer.[23][24] In breast cancer cell line (MCF7) has been found that not only Hsp90 interacted with estrogen receptor alpha (ERα) but also Hsp70-1 and Hsc70 interacted with ERα too.[25]
Given the role of heat shock proteins as an ancient defense system for stabilizing cells and eliminating old and damaged cells, this system has been co-opted by cancer cells to promote their growth.[26] Increased Hsp70 in particular has been shown to inhibit apoptosis of cancer cells,[27] and increased Hsp70 has been shown to be associated with or directly induce endometrial,[28] lung,[29] colon,[30] prostate,[31] and breast[32] cancer, as well as leukemia.[33] Hsp70 in cancer cells may be responsible for tumorigenesis and tumor progression by providing resistance to chemotherapy. Inhibition of Hsp70 has been shown to reduce the size of tumors and can cause their complete regression.[34] Hsp70/Hsp90 is a particularly attractive target for therapeutics, because it is regulated by the inhibition of its ATPase activity, while other HSPs are regulated by nucleotides.[35] Several inhibitors have been designed for Hsp70 that are currently in clinical trials,[36] though as of now HSP90 inhibitors have been more successful.[37] In addition, Hsp70 has been shown to be a regulator of the immune system, activating the immune system as an antigen.[38] Thus, tumor-derived Hsp70 has been suggested as a potential vaccine [39] or avenue to target for immunotherapy.[40] Given the increased expression of Hsp70 in cancer, it has been suggested as a biomarker for cancer prognostics, with high levels portending poor prognosis.[41] An oncogenic mechanism illustrates how extracellular vesicles expressing HSP70 are produced by proliferative Acute Lymphoblastic Leukemia cells and can target and compromise a healthy hematopoiesis system during leukemia development.[42]
Expression in skin tissue
[edit]Both Hsp70 and HSP47 were shown to be expressed in dermis and epidermis following laser irradiation, and the spatial and temporal changes in HSP expression patterns define the laser-induced thermal damage zone and the process of healing in tissues. Hsp70 may define biochemically the thermal damage zone in which cells are targeted for destruction, and HSP47 may illustrate the process of recovery from thermally induced damage.[43] HSP70 helps in protecting skin against the increased melanin and wrinkled formation induced due to UV exposure.[44]
Neurodegeneration
[edit]Inhibition of Hsp90 leads to Hsp70 and Hsp40 upregulation, which can channel misfolded protein for proteasome degradation, which can potentially inhibit the progression of neurodegenerative diseases.[45] For example, Hsp70 overexpression in human neuroglioma cells transfected with mutant alpha-synuclein led to 50% less oligomeric alpha-synuclein species,[46] pointing towards the possibility that increasing its expression could diminish the spread of Parkinson's disease. Similarly, Hsp70 overexpression suppressed poly-Q dependent aggregation and neurodegeneration in cell cultures, yeast,[47] fly,[48] and mouse [49] models, and deletion of hsp70 increased the size of polyQ inclusion bodies,[50] suggesting that increasing its expression could help to prevent Huntington's disease. Similarly, reductions in Hsp70 have been shown in transgenic mouse models of ALS and patients with sporadic ALS.[51] Lastly, increased expression or activity of Hsp70 has been proposed as a method to prevent the progression of Alzheimer's disease, because knock down of Hsp70 promoted A-beta toxicity,[52] and Hsp70 was shown to promote tau stability, while Hsp70 levels are decreased in tauopathies like Alzheimer's disease.[53] Given the complex interplay between the different chaperone proteins, therapeutic development in this field is aimed at investigating how the chaperone network as a whole can be manipulated and the effect of this manipulation on the progression of neurodegenerative disease, but the balance of Hsp70 and Hsp90 levels appears to be central in this pathophysiology.
In diabetes
[edit]The fluctuations in the levels of chaperone HSP70 affect the homeostasis. Diabetes leads to several microvasculature and microvasculature diseases like retinopathy, Toll like receptors are integral part of innate immune system and eHSP70 binds to toll like receptors and activates the MyD88 pathway, further stimulating NF-kB, cytokines like TNFα and IL1 β, increased production of reactive oxygen species contributing to insulin resistance and diabetes. Whereas there is decrease in the levels of iHSP70.[54]
In cardiovascular diseases
[edit]HSP70 is a chaperone with ubiquitous presence.[55] It is crucial in the cardiovascular system. HSP70 normally aids in protein folding and aggregation; when present in the cell, functioning as an anti-inflammatory molecule; however, under stress conditions, it is localized to the extracellular milieu, where it is involved in inducing inflammatory pathways and contributes to disease pathogenesis.[56] It is well established that intracellular HSP70 (iHSP70) levels play a protective role, whereas extracellular HSP70 (eHSP70) levels in circulating blood are linked to pathophysiology in micro and microvasculature, which results in a variety of cardiovascular illnesses. HSP70 homologues identified in human cytosol includes HSPA1A, HSPA1B, HSPA1L, HSPA12B, HSPA13, HSPA14 whereas HSPA9 in mitochondria. The HSP70 acts as DAMP and activates innate immune response which as involved in cardiovascular disease progression.[57]
The chaperone protein acts as auto antigen in atherosclerosis. Increased oxidative stress causes the formation of high-density oxidized LDL, the first event in the formation of plaque. This activates HSP70 and its promoter in the endothelial and smooth muscle cells, which contributes to atherosclerosis by inducing JAK/STAT pathway expression.[58][59]
HSP70 is also linked to high blood pressure, a worldwide concern and risk factor for a variety of cardiovascular diseases. Hypertension causes endothelial dysfunction and vascular wall damage, both of which contribute to arterial stiffness and atherosclerosis.[60] HSPA1A, HSPA1B, and HSPA1L are three genes in humans that encode HSP70, and their polymorphism is linked to the onset of high blood pressure and cardiovascular disease.[61] Angiotensin II, endothelin-1, or phenylepinephrine cause HSP70 overexpression, which activates several molecular pathways, resulting in increased production of ROS, CRP, IL-10, TNF-alpha, and IL-6 [62][61] These inflammatory signals interfere with the antioxidant machinery and results in rapid disease progression.
HSP70 expression increases after the coronary bypass surgery. Exercise has a positive and protective impact on cardiovascular disorders and stimulates the increased production of chaperone protein together known to be cardioprotective.
Family members
[edit]Prokaryotes express three Hsp70 proteins: DnaK, HscA (Hsc66), and HscC (Hsc62).[63]
Eukaryotic organisms express several slightly different Hsp70 proteins. All share the common domain structure, but each has a unique pattern of expression or subcellular localization. These are, among others:
- Hsc70 (Hsp73/HSPA8) is a constitutively expressed chaperone protein. It typically makes up one to three percent of total cellular protein.
- Hsp70 (encoded by three very closely related paralogs: HSPA1A, HSPA1B, and HSPA1L) is a stress-induced protein. High levels can be produced by cells in response to hyperthermia, oxidative stress, and changes in pH.
- Binding immunoglobulin protein (BiP or Grp78) is a protein localized to the endoplasmic reticulum. It is involved in protein folding there, and can be upregulated in response to stress or starvation.
- mtHsp70 or Grp75 is the mitochondrial Hsp70.
The following is a list of human Hsp70 genes and their corresponding proteins:[2]
| gene | protein | synonyms | subcellular location |
|---|---|---|---|
| HSPA1A | Hsp70 | HSP70-1, Hsp72 | Nuc/Cyto |
| HSPA1B | Hsp70 | HSP70-2 | Nuc/Cyto |
| HSPA1L | Hsp70 | ? | |
| HSPA2 | Hsp70-2 | ? | |
| HSPA5 | Hsp70-5 | BiP/Grp78 | ER |
| HSPA6 | Hsp70-6 | ? | |
| HSPA7 | Hsp70-7 | ? | |
| HSPA8 | Hsp70-8 | Hsc70 | Nuc/Cyto |
| HSPA9 | Hsp70-9 | Grp75/mtHsp70 | Mito |
| HSPA12A | Hsp70-12a | ? | |
| HSPA14 | Hsp70-14 | ? |
HSP70s are found in many plants including Arabidopsis, soybean (Glycine max), barley (Hordeum vulgare) and wheat (Triticum aestivum).[64]
Hsps 90 and 110
[edit]
Hsp90s are essential for protein remodeling, similar to Hsp70 proteins, and play an especially vital role in eukaryotes, where it has been suggested that Hsp90 interacts with the DnaK system (composed of DnaK, GrpE, and either DnaJ or CbpA) to facilitate the process of protein remodeling.[65] In E. coli, Hsp90s works collaboratively with Hsp70s to facilitate protein remodeling and activation. Hsp90Ec and DnaK are chaperones of Hsp90 and Hsp70, respectively. DnaK initially binds and stabilizes the misfolded protein before working collaboratively with Hsp90Ec to refold this substrate and cause its activation. Given conditions of excess DnaK, this chaperone has been found to inhibit remodeling of proteins. However, the presence of Hsp90Ec can mitigate this effect and enable protein remodeling despite conditions of excess DnaK.[66]
The Hsp70 superfamily also includes a family of Hsp110/Grp170 (Sse) proteins, which are larger proteins related to Hsp70.[67] The Hsp110 family of proteins have divergent functions: yeast Sse1p has little ATPase activity but is a chaperone on its own as well as a nucleotide exchange factor for Hsp70, while the closely related Sse2p has little unfoldase activity.[12]
The following is a list of currently named human HSP110 genes. HSPH2-4 are proposed names and the current name is linked:[67]
| gene | synonyms | subcellular location |
|---|---|---|
| HSPH1 | HSP105 | Cyto |
| HSPH2 | HSPA4; APG-2; HSP110 | Cyto |
| HSPH3 | HSPA4L; APG-1 | Nuc |
| HSPH4 | HYOU1/Grp170; ORP150; HSP12A | ER |
See also
[edit]References
[edit]- ^ Flaherty KM, DeLuca-Flaherty C, McKay DB (August 1990). "Three-dimensional structure of the ATPase fragment of a 70K heat-shock cognate protein". Nature. 346 (6285): 623–8. Bibcode:1990Natur.346..623F. doi:10.1038/346623a0. PMID 2143562. S2CID 4338916.
- ^ a b Tavaria M, Gabriele T, Kola I, Anderson RL (April 1996). "A hitchhiker's guide to the human Hsp70 family". Cell Stress & Chaperones. 1 (1): 23–8. doi:10.1379/1466-1268(1996)001<0023:ahsgtt>2.3.co;2 (inactive 2024-06-22). PMC 313013. PMID 9222585.
{{cite journal}}: CS1 maint: DOI inactive as of June 2024 (link) - ^ Morano KA (October 2007). "New tricks for an old dog: the evolving world of Hsp70". Annals of the New York Academy of Sciences. 1113 (1): 1–14. Bibcode:2007NYASA1113....1M. doi:10.1196/annals.1391.018. PMID 17513460. S2CID 20917046.
- ^ Giri B, Sethi V, Modi S, Garg B, Banerjee S, Saluja A, Dudeja V (July 2017). ""Heat shock protein 70 in pancreatic diseases: Friend or foe"". Journal of Surgical Oncology. 116 (1): 114–122. doi:10.1002/jso.24653. PMC 5714583. PMID 28543919.
- ^ De Maio A (May 2014). "Extracellular Hsp70: export and function". Current Protein & Peptide Science. 15 (3): 225–31. doi:10.2174/1389203715666140331113057. PMID 24694368.
- ^ Ritossa F (1962). "A new puffing pattern induced by temperature shock and DNP in drosophila". Cellular and Molecular Life Sciences. 18 (12): 571–573. doi:10.1007/BF02172188. S2CID 32525462.
- ^ Ritossa F (June 1996). "Discovery of the heat shock response". Cell Stress & Chaperones. 1 (2): 97–8. doi:10.1379/1466-1268(1996)001<0097:dothsr>2.3.co;2 (inactive 2024-04-26). PMC 248460. PMID 9222594.
{{cite journal}}: CS1 maint: DOI inactive as of April 2024 (link) - ^ a b c Vostakolaei MA, Hatami-Baroogh L, Babaei G, Molavi O, Kordi S, Abdolalizadeh J (May 2021). "Hsp70 in cancer: A double agent in the battle between survival and death". Journal of Cellular Physiology. 236 (5): 3420–3444. doi:10.1002/jcp.30132. PMID 33169384. S2CID 226295557.
- ^ Mayer MP (August 2010). "Gymnastics of molecular chaperones". Molecular Cell. 39 (3): 321–31. doi:10.1016/j.molcel.2010.07.012. PMID 20705236.
- ^ Kao CH, Ryu SW, Kim MJ, Wen X, Wimalarathne O, Paull TT (May 2020). "Growth-Regulated Hsp70 Phosphorylation Regulates Stress Responses and Prion Maintenance". Molecular and Cellular Biology. 40 (12). doi:10.1128/MCB.00628-19. PMC 7261718. PMID 32205407.
- ^ Mashaghi A, Bezrukavnikov S, Minde DP, Wentink AS, Kityk R, Zachmann-Brand B, et al. (November 2016). "Alternative modes of client binding enable functional plasticity of Hsp70". Nature. 539 (7629): 448–451. Bibcode:2016Natur.539..448M. doi:10.1038/nature20137. PMID 27783598. S2CID 4401991.
- ^ a b Bracher A, Verghese J (2015). "GrpE, Hsp110/Grp170, HspBP1/Sil1 and BAG Domain Proteins: Nucleotide Exchange Factors for Hsp70 Molecular Chaperones". The Networking of Chaperones by Co-chaperones. Subcellular Biochemistry. Vol. 78. pp. 1–33. doi:10.1007/978-3-319-11731-7_1. ISBN 978-3-319-11730-0. PMID 25487014.
- ^ Wegele H, Müller L, Buchner J (2004). "Hsp70 and Hsp90—a relay team for protein folding". Hsp70 and Hsp90 – a relay team for protein folding. Reviews of Physiology, Biochemistry and Pharmacology. Vol. 151. pp. 1–44. doi:10.1007/s10254-003-0021-1. ISBN 978-3-540-22096-1. PMID 14740253.
{{cite book}}:|journal=ignored (help) - ^ Cvoro A, Dundjerski J, Trajković D, Matić G (1999-04-01). "The level and phosphorylation of Hsp70 in the rat liver cytosol after adrenalectomy and hyperthermia". Cell Biology International. 23 (4): 313–20. doi:10.1006/cbir.1998.0247. PMID 10600240. S2CID 9113738.
- ^ Gao T, Newton AC (August 2002). "The turn motif is a phosphorylation switch that regulates the binding of Hsp70 to protein kinase C". The Journal of Biological Chemistry. 277 (35): 31585–92. doi:10.1074/jbc.M204335200. PMID 12080070.
- ^ Truman AW, Kristjansdottir K, Wolfgeher D, Hasin N, Polier S, Zhang H, et al. (December 2012). "CDK-dependent Hsp70 Phosphorylation controls G1 cyclin abundance and cell-cycle progression". Cell. 151 (6): 1308–18. doi:10.1016/j.cell.2012.10.051. PMC 3778871. PMID 23217712.
- ^ Muller P, Ruckova E, Halada P, Coates PJ, Hrstka R, Lane DP, Vojtesek B (June 2013). "C-terminal phosphorylation of Hsp70 and Hsp90 regulates alternate binding to co-chaperones CHIP and HOP to determine cellular protein folding/degradation balances". Oncogene. 32 (25): 3101–10. doi:10.1038/onc.2012.314. PMID 22824801. S2CID 12604151.
- ^ Lüders J, Demand J, Höhfeld J (February 2000). "The ubiquitin-related BAG-1 provides a link between the molecular chaperones Hsc70/Hsp70 and the proteasome". The Journal of Biological Chemistry. 275 (7): 4613–7. doi:10.1074/jbc.275.7.4613. PMID 10671488.
- ^ Beere HM, Wolf BB, Cain K, Mosser DD, Mahboubi A, Kuwana T, et al. (August 2000). "Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome". Nature Cell Biology. 2 (8): 469–75. doi:10.1038/35019501. PMID 10934466. S2CID 1507966.
- ^ Gupta S, Deepti A, Deegan S, Lisbona F, Hetz C, Samali A (July 2010). Kelly JW (ed.). "HSP72 protects cells from ER stress-induced apoptosis via enhancement of IRE1alpha-XBP1 signaling through a physical interaction". PLOS Biology. 8 (7): e1000410. doi:10.1371/journal.pbio.1000410. PMC 2897763. PMID 20625543.
- ^ Bobkova NV, Evgen'ev M, Garbuz DG, Kulikov AM, Morozov A, Samokhin A, et al. (December 2015). "Exogenous Hsp70 delays senescence and improves cognitive function in aging mice". Proceedings of the National Academy of Sciences of the United States of America. 112 (52): 16006–16011. Bibcode:2015PNAS..11216006B. doi:10.1073/pnas.1516131112. PMC 4702952. PMID 26668376.
- ^ Ricaniadis N, Kataki A, Agnantis N, Androulakis G, Karakousis CP (February 2001). "Long-term prognostic significance of HSP-70, c-myc and HLA-DR expression in patients with malignant melanoma". European Journal of Surgical Oncology. 27 (1): 88–93. doi:10.1053/ejso.1999.1018. PMID 11237497.
- ^ Ramp U, Mahotka C, Heikaus S, Shibata T, Grimm MO, Willers R, Gabbert HE (October 2007). "Expression of heat shock protein 70 in renal cell carcinoma and its relation to tumor progression and prognosis". Histology and Histopathology. 22 (10): 1099–107. doi:10.14670/HH-22.1099. PMID 17616937.
- ^ Sherman M, Multhoff G (October 2007). "Heat shock proteins in cancer". Annals of the New York Academy of Sciences. 1113 (1): 192–201. Bibcode:2007NYASA1113..192S. doi:10.1196/annals.1391.030. PMID 17978282. S2CID 39372827.
- ^ Dhamad AE, Zhou Z, Zhou J, Du Y (2016). "Systematic Proteomic Identification of the Heat Shock Proteins (Hsp) that Interact with Estrogen Receptor Alpha (ERα) and Biochemical Characterization of the ERα-Hsp70 Interaction". PLOS ONE. 11 (8): e0160312. Bibcode:2016PLoSO..1160312D. doi:10.1371/journal.pone.0160312. PMC 4970746. PMID 27483141.
- ^ Martinková V, Trčka F, Vojtěšek B, Müller P (2018). "The Role of HSP70 in Cancer and its Exploitation as a Therapeutic Target". Klinicka Onkologie. 31 (Suppl 2): 46–54. doi:10.14735/amko20182S46. PMID 31023024.
- ^ Moradi-Marjaneh R, Paseban M, Moradi Marjaneh M (December 2019). "Hsp70 inhibitors: Implications for the treatment of colorectal cancer". IUBMB Life. 71 (12): 1834–1845. doi:10.1002/iub.2157. PMID 31441584. S2CID 201619393.
- ^ Du XL, Jiang T, Wen ZQ, Gao R, Cui M, Wang F (April 2009). "Silencing of heat shock protein 70 expression enhances radiotherapy efficacy and inhibits cell invasion in endometrial cancer cell line". Croatian Medical Journal. 50 (2): 143–50. doi:10.3325/cmj.2009.50.143. PMC 2681060. PMID 19399947.
- ^ Gunther S, Ostheimer C, Stangl S, Specht HM, Mozes P, Jesinghaus M, et al. (2015). "Correlation of Hsp70 Serum Levels with Gross Tumor Volume and Composition of Lymphocyte Subpopulations in Patients with Squamous Cell and Adeno Non-Small Cell Lung Cancer". Frontiers in Immunology. 6: 556. doi:10.3389/fimmu.2015.00556. PMC 4629690. PMID 26579130.
- ^ Black JD, Rezvani K (2016). "Heat Shock Protein 70s as Potential Molecular Targets for Colon Cancer Therapeutics". Current Medicinal Chemistry. 23 (28): 3171–3188. doi:10.2174/0929867323666160627105033. PMID 27356538.
- ^ Moses MA, Kim YS, Rivera-Marquez GM, Oshima N, Watson MJ, Beebe KE, et al. (July 2018). "Targeting the Hsp40/Hsp70 Chaperone Axis as a Novel Strategy to Treat Castration-Resistant Prostate Cancer". Cancer Research. 78 (14): 4022–4035. doi:10.1158/0008-5472.CAN-17-3728. PMC 6050126. PMID 29764864.
- ^ Barnes JA, Dix DJ, Collins BW, Luft C, Allen JW (October 2001). "Expression of inducible Hsp70 enhances the proliferation of MCF-7 breast cancer cells and protects against the cytotoxic effects of hyperthermia". Cell Stress & Chaperones. 6 (4): 316–25. doi:10.1379/1466-1268(2001)006<0316:eoihet>2.0.co;2 (inactive 2024-06-22). PMC 434414. PMID 11795468.
{{cite journal}}: CS1 maint: DOI inactive as of June 2024 (link) - ^ Guo D, Zhang A, Huang J, Suo M, Zhong Y, Liang Y (November 2019). "Suppression of HSP70 inhibits the development of acute lymphoblastic leukemia via TAK1/Egr-1". Biomedicine & Pharmacotherapy. 119: 109399. doi:10.1016/j.biopha.2019.109399. PMID 31521893. S2CID 202582093.
- ^ Kumar S, Stokes J, Singh UP, Scissum Gunn K, Acharya A, Manne U, Mishra M (April 2016). "Targeting Hsp70: A possible therapy for cancer". Cancer Letters. 374 (1): 156–166. doi:10.1016/j.canlet.2016.01.056. PMC 5553548. PMID 26898980.
- ^ Powers MV, Jones K, Barillari C, Westwood I, van Montfort RL, Workman P (April 2010). "Targeting HSP70: the second potentially druggable heat shock protein and molecular chaperone?". Cell Cycle. 9 (8): 1542–50. doi:10.4161/cc.9.8.11204. PMID 20372081. S2CID 37329279.
- ^ Albakova Z, Armeev GA, Kanevskiy LM, Kovalenko EI, Sapozhnikov AM (March 2020). "HSP70 Multi-Functionality in Cancer". Cells. 9 (3): 587. doi:10.3390/cells9030587. PMC 7140411. PMID 32121660.
- ^ Mellatyar H, Talaei S, Pilehvar-Soltanahmadi Y, Barzegar A, Akbarzadeh A, Shahabi A, et al. (June 2018). "Targeted cancer therapy through 17-DMAG as an Hsp90 inhibitor: Overview and current state of the art". Biomedicine & Pharmacotherapy. 102: 608–617. doi:10.1016/j.biopha.2018.03.102. PMID 29602128. S2CID 4505299.
- ^ Kumar S, Deepak P, Kumar S, Kishore D, Acharya A (2009). "Autologous Hsp70 induces antigen specific Th1 immune responses in a murine T-cell lymphoma". Immunological Investigations. 38 (6): 449–65. doi:10.1080/08820130902802673. PMID 19811405. S2CID 24747281.
- ^ Guzhova IV, Margulis BA (October 2016). "HSP70-based anti-cancer immunotherapy". Human Vaccines & Immunotherapeutics. 12 (10): 2529–2535. doi:10.1080/21645515.2016.1190057. PMC 5084976. PMID 27294301.
- ^ Kottke T, Sanchez-Perez L, Diaz RM, Thompson J, Chong H, Harrington K, et al. (December 2007). "Induction of hsp70-mediated Th17 autoimmunity can be exploited as immunotherapy for metastatic prostate cancer". Cancer Research. 67 (24): 11970–9. doi:10.1158/0008-5472.CAN-07-2259. PMID 18089828.
- ^ Chanteloup G, Cordonnier M, Isambert N, Bertaut A, Hervieu A, Hennequin A, et al. (2020). "Monitoring HSP70 exosomes in cancer patients' follow up: a clinical prospective pilot study". Journal of Extracellular Vesicles. 9 (1): 1766192. doi:10.1080/20013078.2020.1766192. PMC 7301715. PMID 32595915.
- ^ Georgievski A, Michel A, Thomas C, Mlamla Z, Pais de Barros JP, Lemaire-Ewing S, et al. (2022). "Acute lymphoblastic leukemia-derived extracellular vesicles affect quiescence of hematopoietic stem and progenitor cells". Cell Death Dis. 12 (4): 337. doi:10.1038/s41419-022-04761-5. PMC 9005650. PMID 35414137. S2CID 248127986.
- ^ Sajjadi AY, Mitra K, Grace M (October 2013). "Expression of heat shock proteins 70 and 47 in tissues following short-pulse laser irradiation: assessment of thermal damage and healing" (PDF). Medical Engineering & Physics. 35 (10): 1406–14. doi:10.1016/j.medengphy.2013.03.011. PMID 23587755.
- ^ Matsuda M, Hoshino T, Yamakawa N, Tahara K, Adachi H, Sobue G, et al. (April 2013). "Suppression of UV-induced wrinkle formation by induction of HSP70 expression in mice". The Journal of Investigative Dermatology. 133 (4): 919–928. doi:10.1038/jid.2012.383. PMID 23096703.
- ^ Lackie RE, Maciejewski A, Ostapchenko VG, Marques-Lopes J, Choy WY, Duennwald ML, et al. (2017). "The Hsp70/Hsp90 Chaperone Machinery in Neurodegenerative Diseases". Frontiers in Neuroscience. 11: 254. doi:10.3389/fnins.2017.00254. PMC 5433227. PMID 28559789.
- ^ Outeiro TF, Putcha P, Tetzlaff JE, Spoelgen R, Koker M, Carvalho F, et al. (April 2008). "Formation of toxic oligomeric alpha-synuclein species in living cells". PLOS ONE. 3 (4): e1867. Bibcode:2008PLoSO...3.1867O. doi:10.1371/journal.pone.0001867. PMC 2270899. PMID 18382657.
- ^ Carmichael J, Chatellier J, Woolfson A, Milstein C, Fersht AR, Rubinsztein DC (August 2000). "Bacterial and yeast chaperones reduce both aggregate formation and cell death in mammalian cell models of Huntington's disease". Proceedings of the National Academy of Sciences of the United States of America. 97 (17): 9701–5. Bibcode:2000PNAS...97.9701C. doi:10.1073/pnas.170280697. PMC 16928. PMID 10920207.
- ^ Warrick JM, Chan HY, Gray-Board GL, Chai Y, Paulson HL, Bonini NM (December 1999). "Suppression of polyglutamine-mediated neurodegeneration in Drosophila by the molecular chaperone HSP70". Nature Genetics. 23 (4): 425–8. doi:10.1038/70532. PMID 10581028. S2CID 24632055.
- ^ Wacker JL, Zareie MH, Fong H, Sarikaya M, Muchowski PJ (December 2004). "Hsp70 and Hsp40 attenuate formation of spherical and annular polyglutamine oligomers by partitioning monomer". Nature Structural & Molecular Biology. 11 (12): 1215–22. doi:10.1038/nsmb860. PMID 15543156. S2CID 43035.
- ^ Wacker JL, Huang SY, Steele AD, Aron R, Lotz GP, Nguyen Q, et al. (July 2009). "Loss of Hsp70 exacerbates pathogenesis but not levels of fibrillar aggregates in a mouse model of Huntington's disease". The Journal of Neuroscience. 29 (28): 9104–14. doi:10.1523/JNEUROSCI.2250-09.2009. PMC 2739279. PMID 19605647.
- ^ Chen HJ, Mitchell JC, Novoselov S, Miller J, Nishimura AL, Scotter EL, et al. (May 2016). "The heat shock response plays an important role in TDP-43 clearance: evidence for dysfunction in amyotrophic lateral sclerosis". Brain. 139 (Pt 5): 1417–32. doi:10.1093/brain/aww028. PMC 4845254. PMID 26936937.
- ^ Brehme M, Voisine C, Rolland T, Wachi S, Soper JH, Zhu Y, et al. (November 2014). "A chaperome subnetwork safeguards proteostasis in aging and neurodegenerative disease". Cell Reports. 9 (3): 1135–50. doi:10.1016/j.celrep.2014.09.042. PMC 4255334. PMID 25437566.
- ^ Dou F, Netzer WJ, Tanemura K, Li F, Hartl FU, Takashima A, et al. (January 2003). "Chaperones increase association of tau protein with microtubules". Proceedings of the National Academy of Sciences of the United States of America. 100 (2): 721–6. Bibcode:2003PNAS..100..721D. doi:10.1073/pnas.242720499. PMC 141063. PMID 12522269.
- ^ Mulyani WR, Sanjiwani MI, Prabawa IP, Lestari AA, Wihandani DM, Suastika K, et al. (February 2020). "Chaperone-Based Therapeutic Target Innovation: Heat Shock Protein 70 (HSP70) for Type 2 Diabetes Mellitus". Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 13: 559–568. doi:10.2147/dmso.s232133. PMC 7051252. PMID 32161482.
- ^ Havalová H, Ondrovičová G, Keresztesová B, Bauer JA, Pevala V, Kutejová E, Kunová N (July 2021). "Mitochondrial HSP70 Chaperone System-The Influence of Post-Translational Modifications and Involvement in Human Diseases". International Journal of Molecular Sciences. 22 (15): 8077. doi:10.3390/ijms22158077. PMC 8347752. PMID 34360841.
- ^ Linder M, Pogge von Strandmann E (September 2021). "The Role of Extracellular HSP70 in the Function of Tumor-Associated Immune Cells". Cancers. 13 (18): 4721. doi:10.3390/cancers13184721. PMC 8466959. PMID 34572948.
- ^ Cai WF, Zhang XW, Yan HM, Ma YG, Wang XX, Yan J, et al. (October 2010). "Intracellular or extracellular heat shock protein 70 differentially regulates cardiac remodelling in pressure overload mice". Cardiovascular Research. 88 (1): 140–149. doi:10.1093/cvr/cvq182. PMID 20542874.
- ^ Wick C (2019). "Heat Shock Protein 60: A Mediator of Atherosclerosis and Its Potential Therapeutic Role". Chaperokine Activity of Heat Shock Proteins. Vol. 16. Cham: Springer International Publishing. pp. 81–103. doi:10.1007/978-3-030-02254-9_4. ISBN 978-3-030-02253-2. S2CID 104325416.
- ^ Leng X, Wang X, Pang W, Zhan R, Zhang Z, Wang L, et al. (July 2013). "Evidence of a role for both anti-Hsp70 antibody and endothelial surface membrane Hsp70 in atherosclerosis". Cell Stress & Chaperones. 18 (4): 483–493. doi:10.1007/s12192-013-0404-4. PMC 3682019. PMID 23334859.
- ^ Gallo G, Volpe M, Savoia C (2022-01-20). "Endothelial Dysfunction in Hypertension: Current Concepts and Clinical Implications". Frontiers in Medicine. 8: 798958. doi:10.3389/fmed.2021.798958. PMC 8811286. PMID 35127755.
- ^ a b Chen Y, Zhao G, Li N, Luo H, Wang X, Gu J (January 2023). "[Corrigendum] Role of 4‑aminobutyrate aminotransferase (ABAT) and the lncRNA co‑expression network in the development of myelodysplastic syndrome". Oncology Reports. 49 (1): 1223–1229. doi:10.1161/01.cir.92.5.1223. PMC 9685366. PMID 36416343.
- ^ Srivastava K, Narang R, Bhatia J, Saluja D (2016-03-18). "Expression of Heat Shock Protein 70 Gene and Its Correlation with Inflammatory Markers in Essential Hypertension". PLOS ONE. 11 (3): e0151060. Bibcode:2016PLoSO..1151060S. doi:10.1371/journal.pone.0151060. PMC 4798713. PMID 26989902.
- ^ Yoshimune K, Yoshimura T, Nakayama T, Nishino T, Esaki N (May 2002). "Hsc62, Hsc56, and GrpE, the third Hsp70 chaperone system of Escherichia coli". Biochemical and Biophysical Research Communications. 293 (5): 1389–95. doi:10.1016/S0006-291X(02)00403-5. PMID 12054669.
- ^ Berka M, Kopecká R, Berková V, Brzobohatý B, Černý M (April 2022). "Regulation of heat shock proteins 70 and their role in plant immunity". Review Paper. Journal of Experimental Botany. 73 (7). Oxford University Press (OUP): 1894–1909. doi:10.1093/jxb/erab549. PMC 8982422. PMID 35022724. Society for Experimental Biology (SEB).
- ^ Genest O, Hoskins JR, Camberg JL, Doyle SM, Wickner S (May 2011). "Heat shock protein 90 from Escherichia coli collaborates with the DnaK chaperone system in client protein remodeling". Proceedings of the National Academy of Sciences of the United States of America. 108 (20): 8206–11. Bibcode:2011PNAS..108.8206G. doi:10.1073/pnas.1104703108. PMC 3100916. PMID 21525416.
- ^ Genest O, Wickner S, Doyle SM (February 2019). "Hsp90 and Hsp70 chaperones: Collaborators in protein remodeling". The Journal of Biological Chemistry. 294 (6): 2109–2120. doi:10.1074/jbc.REV118.002806. PMC 6369297. PMID 30401745.
- ^ a b Kampinga HH, Hageman J, Vos MJ, Kubota H, Tanguay RM, Bruford EA, et al. (January 2009). "Guidelines for the nomenclature of the human heat shock proteins". Cell Stress & Chaperones. 14 (1): 105–11. doi:10.1007/s12192-008-0068-7. PMC 2673902. PMID 18663603.
External links
- HSP70+Heat-Shock+Proteins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
