Phenacemide: Difference between revisions
Appearance
Content deleted Content added
Entranced98 (talk | contribs) +sd |
consistent citation formatting |
||
| Line 54: | Line 54: | ||
}} |
}} |
||
'''Phenacemide''' ([[International Nonproprietary Name|INN]], [[British Approved Name|BAN]]) (brand name '''Phenurone'''), also known as '''phenylacetylurea''', is an [[anticonvulsant]] of the [[ureide]] (acetylurea) class.<ref name="GanellinTriggle1996">{{cite book| |
'''Phenacemide''' ([[International Nonproprietary Name|INN]], [[British Approved Name|BAN]]) (brand name '''Phenurone'''), also known as '''phenylacetylurea''', is an [[anticonvulsant]] of the [[ureide]] (acetylurea) class.<ref name="GanellinTriggle1996">{{cite book| vauthors = Ganellin CR, Triggle DJ |title=Dictionary of Pharmacological Agents|url=https://books.google.com/books?id=A0THacd46ZsC&pg=PA1578|date=21 November 1996|publisher=CRC Press|isbn=978-0-412-46630-4|pages=1578–}}</ref> It is a [[congener (chemistry)|congener]] and ring-opened [[structural analog|analogue]] of [[phenytoin]] (a [[hydantoin]]),<ref name="UniversitiesPress2010">{{cite book | vauthors = Prasad JP | chapter = Central Nervous System |title=Conceptual Pharmacology| chapter-url= https://books.google.com/books?id=s0e_FlM8LKYC&pg=PA236|year=2010|publisher=Universities Press|isbn=978-81-7371-679-9|pages=236–}}</ref><ref name="deStevensZingel2013">{{cite journal | vauthors = Saxena AK, Saxena M | title = Developments in anticonvulsants | journal = Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques | veditors = deStevens G, Zingel V, Leschke C, Hoeprich P, Schultz R, Mehrotra P, Batra S, Bhaduri A, Saxena A, Saxena M | display-editors = 6 | volume = 44 | issue = | pages = 185–291 | date = 1995 | pmid = 7644666 | doi = 10.1007/978-3-0348-7161-7_6 | publisher=Birkhäuser| location=Basel |isbn=978-3-0348-7161-7 }}</ref> and is structurally related to the [[barbiturate]]s and to other [[hydantoin]]s.<ref name="Kadam2007">{{cite book| vauthors = Kadam SS, Mahadik KR, Bothara KG | chapter = Central Nervous System Depresants |title = Principles of Medicinal Chemistry | volume = II |chapter-url=https://books.google.com/books?id=Z7Pb3lJuRksC&pg=PA147|date=1 July 2007|publisher=Pragati Books Pvt. Ltd.|isbn=978-81-85790-03-9|pages=147–}}</ref> Phenacemide was introduced in 1949 for the treatment of [[epilepsy]], but was eventually withdrawn due to [[toxicity]].<ref name="UniversitiesPress2010" /><ref name="deStevensZingel2013" /> |
||
==See also== |
== See also == |
||
* [[Pheneturide]] |
* [[Pheneturide]] |
||
==References== |
== References == |
||
{{Reflist|2}} |
{{Reflist|2}} |
||
== Further reading == |
|||
| ⚫ | |||
{{refbegin}} |
|||
| ⚫ | |||
| ⚫ | * {{cite journal | vauthors = Coker SB, Holmes EW, Egel RT | title = Phenacemide therapy of complex partial epilepsy in children: determination of plasma drug concentrations | journal = Neurology | volume = 37 | issue = 12 | pages = 1861–1866 | date = December 1987 | pmid = 3683877 | doi = 10.1212/wnl.37.12.1861 | s2cid = 219205208 }} |
||
{{refend}} |
|||
| ⚫ | |||
* {{DiseasesDB|34078}} |
* {{DiseasesDB|34078}} |
||
* {{MedlinePlusDrugInfo|uspdi|202454}} |
* {{MedlinePlusDrugInfo|uspdi|202454}} |
||
| ⚫ | |||
| ⚫ | * {{cite journal |vauthors=Coker |
||
{{Anticonvulsants}} |
{{Anticonvulsants}} |
||
Revision as of 09:10, 27 December 2023
 | |
| Clinical data | |
|---|---|
| Trade names | Phenurone |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | 22–25 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.519 |
| Chemical and physical data | |
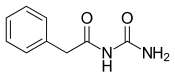
| Formula | C9H10N2O2 |
| Molar mass | 178.191 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Phenacemide (INN, BAN) (brand name Phenurone), also known as phenylacetylurea, is an anticonvulsant of the ureide (acetylurea) class.[1] It is a congener and ring-opened analogue of phenytoin (a hydantoin),[2][3] and is structurally related to the barbiturates and to other hydantoins.[4] Phenacemide was introduced in 1949 for the treatment of epilepsy, but was eventually withdrawn due to toxicity.[2][3]
See also
References
- ^ Ganellin CR, Triggle DJ (21 November 1996). Dictionary of Pharmacological Agents. CRC Press. pp. 1578–. ISBN 978-0-412-46630-4.
- ^ a b Prasad JP (2010). "Central Nervous System". Conceptual Pharmacology. Universities Press. pp. 236–. ISBN 978-81-7371-679-9.
- ^ a b Saxena AK, Saxena M (1995). deStevens G, Zingel V, Leschke C, Hoeprich P, Schultz R, Mehrotra P, et al. (eds.). "Developments in anticonvulsants". Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques. 44. Basel: Birkhäuser: 185–291. doi:10.1007/978-3-0348-7161-7_6. ISBN 978-3-0348-7161-7. PMID 7644666.
- ^ Kadam SS, Mahadik KR, Bothara KG (1 July 2007). "Central Nervous System Depresants". Principles of Medicinal Chemistry. Vol. II. Pragati Books Pvt. Ltd. pp. 147–. ISBN 978-81-85790-03-9.
Further reading
- Coker SB (1986). "The use of phenacemide for intractable partial complex epilepsy in children". Pediatric Neurology. 2 (4): 230–232. doi:10.1016/0887-8994(86)90053-6. PMID 3508693.
- Coker SB, Holmes EW, Egel RT (December 1987). "Phenacemide therapy of complex partial epilepsy in children: determination of plasma drug concentrations". Neurology. 37 (12): 1861–1866. doi:10.1212/wnl.37.12.1861. PMID 3683877. S2CID 219205208.
