Paraldehyde

| |
| Names | |
|---|---|
| IUPAC name
2,4,6-Trimethyl-1,3,5-trioxane
| |
| Systematic IUPAC name
2,4,6-Trimethyl-1,3,5-trioxane | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.219 |
| EC Number |
|
| KEGG | |
| MeSH | Paraldehyde |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H12O3 | |
| Molar mass | 132.159 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Sweet |
| Density | 0.996 g/cm3 |
| Melting point | 12 °C (54 °F; 285 K) |
| Boiling point | 124 °C (255 °F; 397 K)[1] |
| soluble 10% vv at 25 Deg. | |
| Vapor pressure | 13 hPa at 20 °C[1] |
| -86.2·10−6 cm3/mol | |
| Pharmacology | |
| N05CC05 (WHO) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Flammable |
| GHS labelling: | |

| |
| Warning | |
| H226 | |
| P210, P233, P303+P361+P353, P370+P378, P403+P235, P501 | |
| Flash point | 24°C - closed cup |
| Explosive limits | Upper limit: 17 %(V) Lower limit: 1.3 %(V) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
Oral - Rat - 1,530 mg/kg Dermal - Rabbit - 14,015 mg/kg |
| Safety data sheet (SDS) | [1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
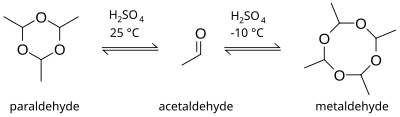
Paraldehyde is the cyclic trimer of acetaldehyde molecules.[2] Formally, it is a derivative of 1,3,5-trioxane, with a methyl group substituted for a hydrogen atom at each carbon. The corresponding tetramer is metaldehyde. A colourless liquid, it is sparingly soluble in water and highly soluble in ethanol. Paraldehyde slowly oxidizes in air, turning brown and producing an odour of acetic acid. It attacks most plastics and rubbers and should be kept in glass bottles.
Paraldehyde was first observed in 1835 by the German chemist Justus Liebig; its empirical formula was determined in 1838 by Liebig's student Hermann Fehling.[3][4] The German chemist Valentin Hermann Weidenbusch (1821–1893), another of Liebig's students, synthesized paraldehyde in 1848 by treating acetaldehyde with acid (either sulfuric or nitric acid) and cooling to 0°C. He found it quite remarkable that when paraldehyde was heated with a trace of the same acid, the reaction went the other way, recreating acetaldehyde.[5][6]
Paraldehyde has uses in industry and medicine.
Preparation
[edit]Paraldehyde can be produced by the direct reaction of acetaldehyde and sulfuric acid. The product of the reaction is dependent on the temperature. At room temperature and higher, the formation of trimer is preferred, but at lower temperatures, around −10 °C, the tetramer metaldehyde is more likely to be produced.[7]
The reaction of sulfuric acid and acetaldehyde is exothermic, with the heat of reaction being −113 kJ·mol−1.[8]
Stereochemistry
[edit]Paraldehyde is produced and used as a mixture of two diastereomers, known as cis- and trans-paraldehyde. For each diastereomer, two chair conformers are possible. The structures (1), (4) and (2), (3) are conformers of cis- and trans-paraldehyde, respectively. The structures (3) (a conformer of (2)) and (4) (a conformer of (1)) are high energy conformers on steric grounds (1,3-diaxial interactions are present) and do not exist to any appreciable extent in a sample of paraldehyde.[9][10]
Reactions
[edit]Heated with catalytic amounts of acid, it depolymerizes back to acetaldehyde:[11][12]
- C6H12O3 → 3CH3CHO
Since paraldehyde has better handling characteristics, it may be used indirectly or directly as a synthetic equivalent of anhydrous acetaldehyde (b.p. 20 °C). For example, it is used as-is in the synthesis of bromal (tribromoacetaldehyde):[13]
- C6H12O3 + 9 Br2 → 3 CBr3CHO + 9 HBr
Medical applications
[edit]Paraldehyde was introduced into clinical practice in the UK by the Italian physician Vincenzo Cervello (1854–1918) in 1882.[14][15][16]
It is a central nervous system depressant and was soon found to be an effective anticonvulsant, hypnotic and sedative. It was included in some cough medicines as an expectorant (though there is no known mechanism for this function beyond the placebo effect).
Paraldehyde was the last injection given to Edith Alice Morrell in 1950 by the suspected serial killer John Bodkin Adams. He was tried for her murder but acquitted.
As a hypnotic/sedative
[edit]It was commonly used to induce sleep in sufferers from delirium tremens but has been replaced by other drugs in this regard. It was considered to have been one of the safest hypnotics and was regularly given at bedtime in psychiatric hospitals and geriatric wards until the 1970s [citation needed], but after it was confirmed that acetaldehyde is a confirmed category-1 human carcinogen, it could no longer be considered appropriately safe to use. Up to 30% of the dose is excreted via the lungs (the rest via the liver). This contributes to a strong unpleasant odour on the breath.
As anti-seizure drug
[edit]Today, paraldehyde is sometimes used to treat status epilepticus. Unlike diazepam and other benzodiazepines, it does not suppress breathing at therapeutic doses and so is safer when no resuscitation facilities exist or when the patient's breathing is already compromised.[17] This makes it a useful emergency medication for parents and other caretakers of children with epilepsy. Since the dose margin between the anticonvulsant and hypnotic effect is small, paraldehyde treatment usually results in sleep.
Administration
[edit]This section needs additional citations for verification. (June 2015) |

Generic paraldehyde is available in 5 mL sealed glass ampoules. Production in the US has been discontinued, but it was previously marketed as Paral.
Paraldehyde has been given orally, rectally, intravenously and by intramuscular injection. It reacts with rubber and plastic which limits the time it may safely be kept in contact with some syringes or tubing before administration.
- Injection. Intramuscular injection can be very painful and lead to sterile abscesses, nerve damage, and tissue necrosis. Intravenous administration can lead to pulmonary edema, circulatory collapse and other complications.
- Oral. Paraldehyde has a hot burning taste and can upset the stomach. It is often mixed with milk or fruit juice in a glass cup and stirred with a metal spoon.
- Rectal. It may be mixed 1 part paraldehyde with 9 parts saline or, alternatively, with an equal mixture of peanut or olive oil.
Industrial applications
[edit]Paraldehyde is used in resin manufacture as an alternative to formaldehyde when making phenol formaldehyde resins. It has also found use as antimicrobial preservative, and rarely as a solvent. It has been used in the generation of aldehyde fuchsin.[18]
References
[edit]- ^ a b c Sigma-Aldrich Co., Paraldehyde.
- ^ Wankhede, N N; Wankhede, D S; Lande, M K; Arbad, B R (March 2006). "Densities and ultrasonic velocities of binary mixtures of 2,4,6-trimethyl-1,3,5-trioxane + n-alcohols at 298.15, 303.15 and 308.15 K" (PDF). Indian Journal of Chemical Technology. 13 (2): 149–155.
- ^ Liebig, Justus (1835) "Ueber die Producte der Oxydation des Alkohols" (On the products of the oxidation of ethanol), Annalen der Chemie, 14 : 133–167; see especially p. 141.
- ^ Fehling, H. (1838) "Ueber zwei dem Aldehyd isomere Verbindungen" (On two compounds that are isomeric to acetaldehyde), Annalen der Chemie, 27 : 319–322; see pp. 321–322.
- ^ Weidenbusch, H. (1848) "Ueber einige Producte der Einwirkung von Alkalien und Säuren auf den Aldehyd" (On some products of the reaction of alkalies and acids with acetaldehyde), Annalen der Chemie, 66 : 152-165; see pp. 155–158.
- ^ Paraldehyde was first synthesized by Weidenbusch in 1848:
- (Editorial staff) (April 15, 1885) "The action of paraldehyde," The Therapeutic Gazette, 9 : 247-250; see p. 247.
- See also: Henry Watts, Matthew Moncrieff Pattison Muir, and Henry Forster Morley, Watts' Dictionary of Chemistry, rev'd, vol. 1 (London, England: Longmans, Green, and Co., 1905), p. 106.
- Neill Busse, Der Meister und seine Schüler: Das Netzwerk Justus Liebigs und seiner Studenten [The Master and His Disciples: The network of Justus Liebig and his students] (Hildesheim, Germany: Georg Olms Verlag, 2015); for Weidenbusch's dates, see p. 274.
- See also: Joseph S. Fruton (March 1988) "The Liebig research group: A reappraisal," Proceedings of the American Philosophical Society, 132 (1) : 1–66; see p. 59.
- See also: Deutsche Biographische Enzyklopädie Archived 2014-08-11 at the Wayback Machine (German Biographical Encyclopedia), p. 1154.
- ^ Latscha, Hans Peter; Kazmaier, Uli; Klein, Helmut A. (2005). Chemie für Biologen mit 71 Tabellen (in German). Berlin. p. 515. ISBN 978-3-540-21161-7. OCLC 76495748.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Eckert, Marc; Fleischmann, Gerald; Jira, Reinhard; Bolt, Hermann M.; Golka, Klaus (2006-12-15), "Acetaldehyde", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, doi:10.1002/14356007.a01_031.pub2, ISBN 3527306730
- ^ Kewley, R. (1970). "Microwave spectrum of paraldehyde". Canadian Journal of Chemistry. 48 (5): 852–855. doi:10.1139/v70-136.
- ^ Carpenter, D. C.; Brockway, L. O. (1936). "The Electron Diffraction Study of Paraldehyde". Journal of the American Chemical Society. 58 (7): 1270–1273. Bibcode:1936JAChS..58.1270C. doi:10.1021/ja01298a053.
- ^ Kendall, E. C.; McKenzie, B. F. (1941). "dl-Alanine". Organic Syntheses; Collected Volumes, vol. 1, p. 21.
- ^ Nathan L. Drake & Giles B. Cooke (1943). "Methyl isopropyl carbinol". Organic Syntheses; Collected Volumes, vol. 2, p. 406.
- ^ F. A. Long & J. W. Howard. "Bromal". Organic Syntheses; Collected Volumes, vol. 2, p. 87.
- ^ López-Muñoz F, Ucha-Udabe R, Alamo C (December 2005). "The history of barbiturates a century after their clinical introduction". Neuropsychiatric Disease and Treatment. 1 (4): 329–43. PMC 2424120. PMID 18568113.
- ^ See:
- Cervello, Vincenzo (1883) "Sull'azione fisiologica della paraldeide e contributo allo studio del cloralio idrato" (On the physiological action of paraldehyde and contribution to the study of chloral hydrate), Archivio per le Scienze Mediche, 6 (12) : 177–214.
- Cervello, Vincenzo (1884) "Recherches cliniques et physiologiques sur la paraldehyde" (Clinical and physiological investigations into paraldehyde), Archives italiennes de biologie, 6 : 113–134.
- ^ For biographical information about Vencenzo Cervello, see: Dizionario Biografico (in Italian)
- ^ Norris E, Marzouk O, Nunn A, McIntyre J, Choonara I (1999). "Respiratory depression in children receiving diazepam for acute seizures: a prospective study". Dev Med Child Neurol. 41 (5): 340–3. doi:10.1017/S0012162299000742 (inactive 9 December 2024). PMID 10378761.
{{cite journal}}: CS1 maint: DOI inactive as of December 2024 (link) - ^ Nettleton GS (February 1982). "The role of paraldehyde in the rapid preparation of aldehyde fuchsin". Journal of Histochemistry and Cytochemistry. 30 (2): 175–8. doi:10.1177/30.2.6174561. PMID 6174561.