MCHB-1
Appearance
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
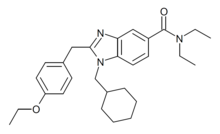
| Formula | C28H37N3O2 |
| Molar mass | 447.623 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
MCHB-1 is a benzimidazole derived drug which was researched as an analgesic but never developed for medical use. It acts as a potent agonist of the CB2 receptor, with an EC50 of 0.52nM at CB2, and ~30x selectivity over CB1 (Ki of 110nM at CB1 vs 3.7nM at CB2).[1][2] It has been sold online as a designer drug, first being identified in Germany in December 2013.[3]
See also
References
- ^ Pagé D, Balaux E, Boisvert L, Liu Z, Milburn C, Tremblay M, et al. (July 2008). "Novel benzimidazole derivatives as selective CB2 agonists". Bioorganic & Medicinal Chemistry Letters. 18 (13): 3695–700. doi:10.1016/j.bmcl.2008.05.073. PMID 18522867.
- ^ Yu XH, Cao CQ, Martino G, Puma C, Morinville A, St-Onge S, et al. (November 2010). "A peripherally restricted cannabinoid receptor agonist produces robust anti-nociceptive effects in rodent models of inflammatory and neuropathic pain". Pain. 151 (2): 337–44. doi:10.1016/j.pain.2010.07.019. PMID 20696525. S2CID 27731273.
- ^ Westphal F, Sönnichsen FD, Knecht S, Auwärter V, Huppertz L (April 2015). "Two thiazolylindoles and a benzimidazole: novel compounds on the designer drug market with potential cannabinoid receptor activity". Forensic Science International. 249: 133–47. doi:10.1016/j.forsciint.2015.01.014. PMID 25698514.
