FUBIMINA
This article may be too technical for most readers to understand. (June 2015) |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
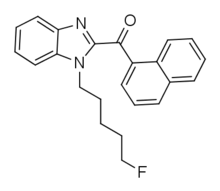
| Formula | C23H21FN2O |
| Molar mass | 360.432 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
FUBIMINA (also known as BIM-2201, BZ-2201 and FTHJ) is a synthetic cannabinoid that is the benzimidazole analog of AM-2201[1] and has been used as an active ingredient in synthetic cannabis products.[2] It was first identified in Japan in 2013, alongside MEPIRAPIM.[3]
FUBIMINA acts as a reasonably potent agonist for the CB2 receptor (Ki = 23.45 nM), with 12x selectivity over CB1 (Ki = 296.1 nM), and does not fully substitute for Δ9-THC in rat discrimination studies.[4]
Related benzimidazole derivatives have been reported to be highly selective agonists for the CB2 receptor.[5]
See also[edit]
- AM-694
- AM-1235
- AM-2232
- AM-2233
- BIM-018
- JWH-018
- List of AM cannabinoids
- List of JWH cannabinoids
- THJ-2201
References[edit]
- ^ "FUBIMINA". Cayman Chemical. Retrieved 22 June 2015.
- ^ Diao X, Scheidweiler KB, Wohlfarth A, Zhu M, Pang S, Huestis MA (2016). "Strategies to distinguish new synthetic cannabinoid FUBIMINA (BIM-2201) intake from its isomer THJ-2201: metabolism of FUBIMINA in human hepatocytes". Forensic Toxicology. 34 (2): 256–267. doi:10.1007/s11419-016-0312-2. PMC 4971051. PMID 27547265.
- ^ Uchiyama N, Shimokawa Y, Matsuda S, Kawamura M, Kikura-Hanajiri R, Goda Y (2014). "Two new synthetic cannabinoids, AM-2201 benzimidazole analog (FUBIMINA) and (4-methylpiperazin-1-yl)(1-pentyl-1H-indol-3-yl)methanone (MEPIRAPIM), and three phenethylamine derivatives, 25H-NBOMe 3,4,5-trimethoxybenzyl analog, 25B-NBOMe, and 2C-N-NBOMe, identified in illegal products". Forensic Toxicology. 32 (1): 105–115. doi:10.1007/s11419-013-0217-2. S2CID 32599561.
- ^ Wiley JL, Marusich JA, Lefever TW, Antonazzo KR, Wallgren MT, Cortes RA, et al. (September 2015). "AB-CHMINACA, AB-PINACA, and FUBIMINA: Affinity and Potency of Novel Synthetic Cannabinoids in Producing Δ9-Tetrahydrocannabinol-Like Effects in Mice". The Journal of Pharmacology and Experimental Therapeutics. 354 (3): 328–339. doi:10.1124/jpet.115.225326. PMC 4538877. PMID 26105953.
- ^ Pagé D, Balaux E, Boisvert L, Liu Z, Milburn C, Tremblay M, et al. (July 2008). "Novel benzimidazole derivatives as selective CB2 agonists". Bioorganic & Medicinal Chemistry Letters. 18 (13): 3695–3700. doi:10.1016/j.bmcl.2008.05.073. PMID 18522867.
