Mestanolone
 | |
| Clinical data | |
|---|---|
| Trade names | Androstalone, Ermalone, others |
| Other names | RU-143; Methylandrostanolone; Methyldihydrotestosterone; Methyl-DHT; 17α-Methyl-4,5α-dihydrotestosterone; 17α-Methyl-DHT; 17α-Methyl-5α-androstan-17β-ol-3-one; |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| Drug class | Androgen; Anabolic steroid |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Liver |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.549 |
| Chemical and physical data | |
| Formula | C20H32O2 |
| Molar mass | 304.474 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mestanolone, also known as methylandrostanolone and sold under the brand names Androstalone and Ermalone among others, is an androgen and anabolic steroid (AAS) medication which is mostly no longer used.[1][2][3][4] It is still available for use in Japan however.[2][3] It is taken by mouth.[4]
Side effects of mestanolone include symptoms of masculinization like acne, increased hair growth, voice changes, and increased sexual desire.[4] It can also cause liver damage.[4] The drug is a synthetic androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT).[4][5] It has strong androgenic effects and weak anabolic effects, which make it useful for producing masculine psychological and behavioral effects.[4] The drug has no estrogenic effects.[4]
Mestanolone was discovered in 1935 and was introduced for medical use in the 1950s.[6][7][8][4] In addition to its medical use, mestanolone has been used to improve physique and performance.[4] It was used in East Germany in Olympic athletes as part of a state-sponsored doping program in the 1970s and 1980s.[4] The drug is a controlled substance in many countries and so non-medical use is generally illicit.[4]
Medical uses
[edit]Available forms
[edit]Mestanolone was available in the form of 25 mg sublingual tablets (brand name Ermalone).[9]
Pharmacology
[edit]Pharmacodynamics
[edit]Mestanolone is an AAS, with both androgenic and anabolic effects.[4] It is very similar in its effects to androstanolone (dihydrotestosterone; DHT), and can be thought of as an orally active version of this AAS.[4] Due to inactivation by 3α-hydroxysteroid dehydrogenase (3α-HSD) in skeletal muscle, mestanolone is described as a very poor anabolic agent, similarly to androstanolone and mesterolone.[4] As mestanolone is 5α-reduced, it cannot be aromatized and hence has no propensity for estrogenic side effects such as gynecomastia.[4] The drug also has no progestogenic activity.[4] Like other 17α-alkylated AAS, mestanolone is hepatotoxic.[4]
Pharmacokinetics
[edit]Due to its C17α methyl group, unlike androstanolone, mestanolone is orally active.[4]
Chemistry
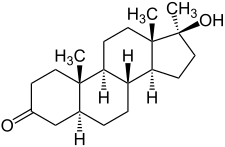
[edit]Mestanolone, also known as 17α-methyl-4,5α-dihydrotestosterone (17α-methyl-DHT) or as 17α-methyl-5α-androstan-17β-ol-3-one, is a synthetic androstane steroid and a 17α-alkylated derivative of dihydrotestosterone (DHT).[1][4] It differs from DHT only by the presence of the methyl group at the C17α position.[1][4] Close synthetic relatives of mestanolone include oxandrolone (2-oxa-17α-methyl-DHT), oxymetholone (2-hydroxymethylene-17α-methyl-DHT), and stanozolol (a derivative of 17α-methyl-DHT (mestanolone) with a pyrazole ring fused to the A ring).[1][4]
Side effects
[edit]Side effects of mestanolone include virilization and hepatotoxicity among others.[4]
History
[edit]Mestanolone was first synthesized in 1935 along with methyltestosterone and methandriol.[6][7] It was developed by Roussel in the 1950s and was introduced for medical use, under the brand names Androstalone and Ermalone, by at least 1960.[4][10][8] It was marketed in Germany.[4] The drug was originally thought to be a potent anabolic agent, but subsequent research showed that it actually has relatively weak anabolic effects and is mostly an androgen.[4] Mestanolone was used as a doping agent in athletes competing in the Olympics from East Germany due to a state-sponsored doping program in the 1970s and 1980s.[4] Its value is said to have been less as a muscle-builder and more as an androgen in the central nervous system and neuromuscular interaction, improving speed, strength, aggression, focus, endurance, and stress resilience.[4] Today, mestanolone has mostly been discontinued in medicine, though it is still available in Japan.[2][3][4]
Society and culture
[edit]Generic names
[edit]Mestanolone is the generic name of the drug and its INN, BAN, and JAN.[1][11]
Brand names
[edit]Mestanolone was marketed under the brand names Andoron, Androstalone, Ermalone, Mesanolon, and Notandron among many others.[4][2][12][3]
Availability
[edit]Mestanolone has mostly been discontinued but remains available in Japan.[2][3][4]
References
[edit]- ^ a b c d e Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 775–. ISBN 978-1-4757-2085-3.
- ^ a b c d e "Mestanolone". Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 655–. ISBN 978-3-88763-075-1.
- ^ a b c d e "S1. Anabolic Agents". Drugs in Sport: Anti-Doping Prohibited List – via Drugs.com.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad Llewellyn W (2009). Anabolics. Molecular Nutrition Llc. p. 241. ISBN 978-0967930473.
- ^ Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ^ a b Schänzer W (July 1996). "Metabolism of anabolic androgenic steroids". Clinical Chemistry. 42 (7): 1001–1020. doi:10.1093/clinchem/42.7.1001. PMID 8674183.
- ^ a b Ruzicka L, Goldberg MW, Rosenberg HR (1935). "Sexualhormone X. Herstellung des 17-Methyl-testosterons und anderer Androsten- und Androstanderivate. Zusammenhänge zwischen chemischer Konstitution und männlicher Hormonwirkung". Helvetica Chimica Acta. 18 (1): 1487–1498. doi:10.1002/hlca.193501801203. ISSN 0018-019X.
- ^ a b Arnold A, Potts GO, Beyler AL (December 1963). "The Ratio of Anabolic to Androgenic Activity of 7: 17-Dimethyltestosterone, Oxymesterone, Mestanolone and Fluoxymesterone". The Journal of Endocrinology. 28: 87–92. doi:10.1677/joe.0.0280087. PMID 14086172.
- ^ Krüskemper HL (22 October 2013). Anabolic Steroids. Elsevier. pp. 196–. ISBN 978-1-4832-6504-9.
- ^ Bishop PM (January 1960). "Male sex hormones". British Medical Journal. 1 (5167): 184–186. doi:10.1136/bmj.1.5167.184. PMC 1966335. PMID 13800998.
- ^ "Mestanolone [INN:BAN:JAN]". ChemIDplus. U.S. National Library of Medicine.
- ^ Negwer M (1987). Organic-chemical Drugs and Their Synonyms: (an International Survey). VCH Publishers. ISBN 978-0-89573-552-2.
Anavormol, Andoron, Androne, Androstalone, Antalon "Kobayashi K.", Assimil, Ermalone, Etnabolate, Hermalone-Glosset, Macrobin (Tabl. -- Syrup), Mesanolon, Mestalone, Mestanolone", Methyantalon, Methybol, 172-Methylandrostanolone, Preroide,. 1045.
