Interleukin 16
Interleukin 16 is a pro-inflammatory pleiotropic cytokine. Its precursor, pro-interleukin-16 is a protein that in humans is encoded by the IL16 gene.[5][6] This gene was discovered in 1982 at Boston University by Dr. David Center and Dr. William Cruikshank.[7]
Function
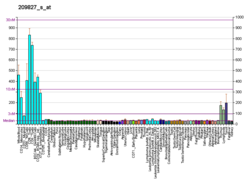
[edit]The cytokine encoded by this gene is a pleiotropic cytokine that functions as a chemoattractant, a modulator of T cell activation, and an inhibitor of HIV replication. The signaling process of this cytokine is mediated by CD4. The product of this gene undergoes proteolytic processing, which is found to yield two functional proteins. The cytokine function is exclusively attributed to the secreted C-terminal peptide, while the N-terminal product may play a role in cell cycle control. Caspase 3 is reported to be involved in the proteolytic processing of this protein. Two alternatively spliced transcript variants encoding distinct isoforms have been reported.[6] Interleukin 16 (IL-16) is released by a variety of cells (including lymphocytes and some epithelial cells) that has been characterized as a chemoattractant for certain immune cells expressing the cell surface molecule CD4. IL-16 was originally described as a factor that could attract activated T cells in humans, it was previously called lymphocyte chemoattractant factor (LCF).[7] Since then, this interleukin has been shown to recruit and activate many other cells expressing the CD4 molecule, including monocytes, eosinophils, and dendritic cells.[8]
The structure of IL-16 was determined following its cloning in 1994.[9] This cytokine is produced as a precursor peptide (pro-IL-16) that requires processing by an enzyme called caspase-3 to become active. CD4 is the cell signaling receptor for mature IL-16.
Interactions
[edit]Interleukin 16 has been shown to interact with:
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000172349 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000001741 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Baier M, Bannert N, Werner A, Lang K, Kurth R (May 1997). "Molecular cloning, sequence, expression, and processing of the interleukin 16 precursor". Proceedings of the National Academy of Sciences of the United States of America. 94 (10): 5273–5277. Bibcode:1997PNAS...94.5273B. doi:10.1073/pnas.94.10.5273. PMC 24668. PMID 9144227.
- ^ a b "Entrez Gene: IL16 interleukin 16 (lymphocyte chemoattractant factor)".
- ^ a b Cruikshank W, Center DM (June 1982). "Modulation of lymphocyte migration by human lymphokines. II. Purification of a lymphotactic factor (LCF)". Journal of Immunology. 128 (6): 2569–2574. doi:10.4049/jimmunol.128.6.2569. PMID 7042841. S2CID 33890529.
- ^ Cruikshank WW, Kornfeld H, Center DM (June 2000). "Interleukin-16". Journal of Leukocyte Biology. 67 (6): 757–766. doi:10.1002/jlb.67.6.757. PMID 10857846.
- ^ Cruikshank WW, Center DM, Nisar N, Wu M, Natke B, Theodore AC, Kornfeld H (May 1994). "Molecular and functional analysis of a lymphocyte chemoattractant factor: association of biologic function with CD4 expression". Proceedings of the National Academy of Sciences of the United States of America. 91 (11): 5109–5113. Bibcode:1994PNAS...91.5109C. doi:10.1073/pnas.91.11.5109. PMC 43941. PMID 7910967.
- ^ a b c d e Kurschner C, Yuzaki M (September 1999). "Neuronal interleukin-16 (NIL-16): a dual function PDZ domain protein". The Journal of Neuroscience. 19 (18): 7770–7780. doi:10.1523/JNEUROSCI.19-18-07770.1999. PMC 6782450. PMID 10479680.
- ^ a b Bannert N, Vollhardt K, Asomuddinov B, Haag M, König H, Norley S, Kurth R (October 2003). "PDZ Domain-mediated interaction of interleukin-16 precursor proteins with myosin phosphatase targeting subunits". The Journal of Biological Chemistry. 278 (43): 42190–42199. doi:10.1074/jbc.M306669200. PMID 12923170.
Further reading
[edit]- Wilson KC, Center DM, Cruikshank WW (June 2004). "The effect of interleukin-16 and its precursor on T lymphocyte activation and growth". Growth Factors. 22 (2): 97–104. doi:10.1080/08977190410001704679. PMID 15253385. S2CID 16860935.
- Copeland KF (December 2005). "Modulation of HIV-1 transcription by cytokines and chemokines". Mini Reviews in Medicinal Chemistry. 5 (12): 1093–1101. doi:10.2174/138955705774933383. PMID 16375755.
- Rand TH, Cruikshank WW, Center DM, Weller PF (June 1991). "CD4-mediated stimulation of human eosinophils: lymphocyte chemoattractant factor and other CD4-binding ligands elicit eosinophil migration". The Journal of Experimental Medicine. 173 (6): 1521–1528. doi:10.1084/jem.173.6.1521. PMC 2190841. PMID 1851800.
- Ryan TC, Cruikshank WW, Kornfeld H, Collins TL, Center DM (July 1995). "The CD4-associated tyrosine kinase p56lck is required for lymphocyte chemoattractant factor-induced T lymphocyte migration". The Journal of Biological Chemistry. 270 (29): 17081–17086. doi:10.1074/jbc.270.29.17081. PMID 7615501.
- Cruikshank WW, Center DM, Nisar N, Wu M, Natke B, Theodore AC, Kornfeld H (May 1994). "Molecular and functional analysis of a lymphocyte chemoattractant factor: association of biologic function with CD4 expression". Proceedings of the National Academy of Sciences of the United States of America. 91 (11): 5109–5113. Bibcode:1994PNAS...91.5109C. doi:10.1073/pnas.91.11.5109. PMC 43941. PMID 7910967.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–174. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Parada NA, Cruikshank WW, Danis HL, Ryan TC, Center DM (February 1996). "IL-16- and other CD4 ligand-induced migration is dependent upon protein kinase C". Cellular Immunology. 168 (1): 100–106. doi:10.1006/cimm.1996.0054. PMID 8599832.
- Bannert N, Baier M, Werner A, Kurth R (May 1996). "Interleukin-16 or not?". Nature. 381 (6577): 30. Bibcode:1996Natur.381...30B. doi:10.1038/381030a0. PMID 8609984. S2CID 4347508.
- Maciaszek JW, Parada NA, Cruikshank WW, Center DM, Kornfeld H, Viglianti GA (January 1997). "IL-16 represses HIV-1 promoter activity". Journal of Immunology. 158 (1): 5–8. doi:10.4049/jimmunol.158.1.5. PMID 8977168. S2CID 28041095.
- Laberge S, Ernst P, Ghaffar O, Cruikshank WW, Kornfeld H, Center DM, Hamid Q (August 1997). "Increased expression of interleukin-16 in bronchial mucosa of subjects with atopic asthma". American Journal of Respiratory Cell and Molecular Biology. 17 (2): 193–202. CiteSeerX 10.1.1.319.2157. doi:10.1165/ajrcmb.17.2.2750. PMID 9271307.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–156. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Zhang Y, Center DM, Wu DM, Cruikshank WW, Yuan J, Andrews DW, Kornfeld H (January 1998). "Processing and activation of pro-interleukin-16 by caspase-3". The Journal of Biological Chemistry. 273 (2): 1144–1149. doi:10.1074/jbc.273.2.1144. PMID 9422780.
- Mühlhahn P, Zweckstetter M, Georgescu J, Ciosto C, Renner C, Lanzendörfer M, et al. (August 1998). "Structure of interleukin 16 resembles a PDZ domain with an occluded peptide binding site". Nature Structural Biology. 5 (8): 682–686. doi:10.1038/1376. hdl:11858/00-001M-0000-002B-A4A1-7. PMID 9699630. S2CID 39286954.
- Chupp GL, Wright EA, Wu D, Vallen-Mashikian M, Cruikshank WW, Center DM, et al. (September 1998). "Tissue and T cell distribution of precursor and mature IL-16". Journal of Immunology. 161 (6): 3114–3119. doi:10.4049/jimmunol.161.6.3114. PMID 9743378. S2CID 11896037.
- Bannert N, Avots A, Baier M, Serfling E, Kurth R (February 1999). "GA-binding protein factors, in concert with the coactivator CREB binding protein/p300, control the induction of the interleukin 16 promoter in T lymphocytes". Proceedings of the National Academy of Sciences of the United States of America. 96 (4): 1541–1546. Bibcode:1999PNAS...96.1541B. doi:10.1073/pnas.96.4.1541. PMC 15509. PMID 9990060.
- Kim HS (1999). "Assignment of human interleukin 16 (IL16) to chromosome 15q26.3 by radiation hybrid mapping". Cytogenetics and Cell Genetics. 84 (1–2): 93. doi:10.1159/000015224. PMID 10343113. S2CID 83880527.
- Liu Y, Cruikshank WW, O'Loughlin T, O'Reilly P, Center DM, Kornfeld H (August 1999). "Identification of a CD4 domain required for interleukin-16 binding and lymphocyte activation". The Journal of Biological Chemistry. 274 (33): 23387–23395. doi:10.1074/jbc.274.33.23387. PMID 10438516.
- Kaser A, Dunzendorfer S, Offner FA, Ryan T, Schwabegger A, Cruikshank WW, et al. (September 1999). "A role for IL-16 in the cross-talk between dendritic cells and T cells". Journal of Immunology. 163 (6): 3232–3238. doi:10.4049/jimmunol.163.6.3232. PMID 10477592. S2CID 36053665.
- Kurschner C, Yuzaki M (September 1999). "Neuronal interleukin-16 (NIL-16): a dual function PDZ domain protein". The Journal of Neuroscience. 19 (18): 7770–7780. doi:10.1523/JNEUROSCI.19-18-07770.1999. PMC 6782450. PMID 10479680.
External links
[edit]- Overview of all the structural information available in the PDB for UniProt: Q14005 (Interleukin-16) at the PDBe-KB.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.