Interleukin 5
Interleukin 5 (IL-5) is an interleukin produced by type-2 T helper cells and mast cells.
Function
[edit]Through binding to the interleukin-5 receptor, interleukin 5 stimulates B cell growth and increases immunoglobulin secretion—primarily IgA. It is also a key mediator in eosinophil activation.
Structure
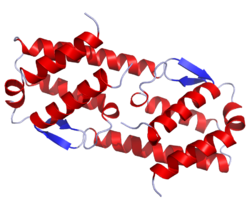
[edit]IL-5 is a 115-amino acid (in human, 133 in the mouse) -long Th2 cytokine that is part of the hematopoietic family. Unlike other members of this cytokine family (namely interleukin 3 and GM-CSF), this glycoprotein in its active form is a homodimer.[5]
Tissue expression
[edit]The IL-5 gene is located on chromosome 11 in the mouse, and chromosome 5 in humans, in close proximity to the genes encoding IL-3, IL-4, and granulocyte-macrophage colony-stimulating factor (GM-CSF),[6][7] which are often co-expressed in Th2 cells. IL-5 is also expressed by eosinophils[8] and has been observed in the mast cells of asthmatic airways by immunohistochemistry.[9] IL-5 expression is regulated by several transcription factors including GATA3.[10]
Clinical significance
[edit]IL-5 has long been associated with the cause of several allergic diseases including allergic rhinitis and asthma, wherein a large increase in the number of circulating, airway tissue, and induced sputum eosinophils have been observed.[11] Given the high concordance of eosinophils and, in particular, allergic asthma pathology, it has been widely speculated that eosinophils have an important role in the pathology of this disease.[12]
As of 2019, there are two FDA-approved monoclonal antibodies that inhibit IL-5, mepolizumab and reslizumab. Additionally, the antibody benralizumab blocks the interleukin-5 receptor. All three drugs are used to treat severe eosinophilic asthma[13] and eosinophilic granulomatosis with polyangiitis (EGPA).[14] Another antibody, depemokimab (GSK3511294), is under development.[15]
Some hydroxyethylaminomethylbenzimidazole analogs have shown IL-5 inhibition in vitro.[16]
Effect on eosinophils
[edit]Eosinophils are terminally differentiated granulocytes found in most mammals. The principal role of these cells, in a healthy host, is the elimination of antibody bound parasites through the release of cytotoxic granule proteins.[17] Given that eosinophils are the primary IL-5Rα-expressing cells, it is not surprising that this cell type responds to IL-5. In fact, IL-5 was originally discovered as an eosinophil colony-stimulating factor,[18] is a major regulator of eosinophil accumulation in tissues, and can modulate eosinophil behavior at every stage from maturation to survival. Mepolizumab is a monoclonal antibody antagonist IL-5 which can reduce excessive eosinophilia.
In Hodgkin lymphoma, the typically-observed eosinophilia is thought to be attributable to an increased production of IL-5.[19]
Interactions
[edit]IL-5 has been shown to interact with interleukin 5 receptor alpha subunit.[20][21][22]
Receptors
[edit]The IL-5 receptor is composed of an α and a βc chain.[23] The α subunit is specific for the IL-5 molecule, whereas the βc subunit also recognised by interleukin 3 (IL-3) and granulocyte-macrophage colony-stimulating factor (GM-CSF).[23][24] Glycosylation of the Asn196 residue of the Rα subunit appears to be essential for binding of IL-5.[25]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000113525 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000036117 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Milburn MV, Hassell AM, Lambert MH, Jordan SR, Proudfoot AE, Graber P, Wells TN (May 1993). "A novel dimer configuration revealed by the crystal structure at 2.4 A resolution of human interleukin-5". Nature. 363 (6425): 172–176. Bibcode:1993Natur.363..172M. doi:10.1038/363172a0. PMID 8483502. S2CID 4254991.
- ^ Lee JS, Campbell HD, Kozak CA, Young IG (March 1989). "The IL-4 and IL-5 genes are closely linked and are part of a cytokine gene cluster on mouse chromosome 11". Somatic Cell and Molecular Genetics. 15 (2): 143–152. doi:10.1007/BF01535075. PMID 2784591. S2CID 41719900.
- ^ van Leeuwen BH, Martinson ME, Webb GC, Young IG (April 1989). "Molecular organization of the cytokine gene cluster, involving the human IL-3, IL-4, IL-5, and GM-CSF genes, on human chromosome 5". Blood. 73 (5): 1142–1148. doi:10.1182/blood.V73.5.1142.1142. PMID 2564789.
- ^ Dubucquoi S, Desreumaux P, Janin A, Klein O, Goldman M, Tavernier J, et al. (February 1994). "Interleukin 5 synthesis by eosinophils: association with granules and immunoglobulin-dependent secretion". The Journal of Experimental Medicine. 179 (2): 703–708. doi:10.1084/jem.179.2.703. PMC 2191391. PMID 8294877.
- ^ Bradding P, Roberts JA, Britten KM, Montefort S, Djukanovic R, Mueller R, et al. (May 1994). "Interleukin-4, -5, and -6 and tumor necrosis factor-alpha in normal and asthmatic airways: evidence for the human mast cell as a source of these cytokines". American Journal of Respiratory Cell and Molecular Biology. 10 (5): 471–480. doi:10.1165/ajrcmb.10.5.8179909. PMID 8179909.
- ^ Kaminuma O, Mori A, Kitamura N, Hashimoto T, Kitamura F, Inokuma S, Miyatake S (2005). "Role of GATA-3 in IL-5 gene transcription by CD4+ T cells of asthmatic patients". International Archives of Allergy and Immunology. 137 (Suppl 1): 55–59. doi:10.1159/000085433. PMID 15947486. S2CID 25517499.
- ^ Shen HH, Ochkur SI, McGarry MP, Crosby JR, Hines EM, Borchers MT, et al. (March 2003). "A causative relationship exists between eosinophils and the development of allergic pulmonary pathologies in the mouse". Journal of Immunology. 170 (6): 3296–3305. doi:10.4049/jimmunol.170.6.3296. PMID 12626589.
- ^ Sanderson CJ (June 1992). "Interleukin-5, eosinophils, and disease". Blood. 79 (12): 3101–3109. doi:10.1182/blood.V79.12.3101.3101. PMID 1596561.
- ^ "Anti-interleukin-5 therapy for severe asthma: A new therapeutic option". Mayo Clinic. 8 January 2019. Retrieved 14 December 2023.
- ^ Berti A, Atzeni F, Dagna L, Del Giacco S, Emmi G, Salvarani C, Vaglio A (February 2023). "Targeting the interleukin-5 pathway in EGPA: evidence, uncertainties and opportunities". Annals of the Rheumatic Diseases. 82 (2): 164–168. doi:10.1136/ard-2022-223044. PMID 36357156. S2CID 253457684.
- ^ Singh D, Fuhr R, Bird NP, Mole S, Hardes K, Man YL, et al. (February 2022). "A Phase 1 study of the long-acting anti-IL-5 monoclonal antibody GSK3511294 in patients with asthma". British Journal of Clinical Pharmacology. 88 (2): 702–712. doi:10.1111/bcp.15002. PMC 9290054. PMID 34292606.
- ^ Boggu PR, Kim Y, Jung SH (November 2019). "Discovery of benzimidazole analogs as a novel interleukin-5 inhibitors". European Journal of Medicinal Chemistry. 181: 111574. doi:10.1016/j.ejmech.2019.111574. PMID 31400705. S2CID 199527755.
- ^ Giembycz MA, Lindsay MA (June 1999). "Pharmacology of the eosinophil". Pharmacological Reviews. 51 (2): 213–340. PMID 10353986.
- ^ Lopez AF, Begley CG, Williamson DJ, Warren DJ, Vadas MA, Sanderson CJ (May 1986). "Murine eosinophil differentiation factor. An eosinophil-specific colony-stimulating factor with activity for human cells". The Journal of Experimental Medicine. 163 (5): 1085–1099. doi:10.1084/jem.163.5.1085. PMC 2188112. PMID 3486243.
- ^ Di Biagio E, Sánchez-Borges M, Desenne JJ, Suárez-Chacón R, Somoza R, Acquatella G (July 1996). "Eosinophilia in Hodgkin's disease: a role for interleukin 5". International Archives of Allergy and Immunology. 110 (3): 244–251. doi:10.1159/000237294. PMID 8688671.
- ^ Woodcock JM, Zacharakis B, Plaetinck G, Bagley CJ, Qiyu S, Hercus TR, et al. (November 1994). "Three residues in the common beta chain of the human GM-CSF, IL-3 and IL-5 receptors are essential for GM-CSF and IL-5 but not IL-3 high affinity binding and interact with Glu21 of GM-CSF". The EMBO Journal. 13 (21): 5176–5185. doi:10.1002/j.1460-2075.1994.tb06848.x. PMC 395466. PMID 7957082.
- ^ Johanson K, Appelbaum E, Doyle M, Hensley P, Zhao B, Abdel-Meguid SS, et al. (April 1995). "Binding interactions of human interleukin 5 with its receptor alpha subunit. Large scale production, structural, and functional studies of Drosophila-expressed recombinant proteins". The Journal of Biological Chemistry. 270 (16): 9459–9471. doi:10.1074/jbc.270.16.9459. PMID 7721873.
- ^ Murata Y, Takaki S, Migita M, Kikuchi Y, Tominaga A, Takatsu K (February 1992). "Molecular cloning and expression of the human interleukin 5 receptor". The Journal of Experimental Medicine. 175 (2): 341–351. doi:10.1084/jem.175.2.341. PMC 2119102. PMID 1732409.
- ^ a b Tavernier J, Devos R, Cornelis S, Tuypens T, Van der Heyden J, Fiers W, Plaetinck G (September 1991). "A human high affinity interleukin-5 receptor (IL5R) is composed of an IL5-specific alpha chain and a beta chain shared with the receptor for GM-CSF". Cell. 66 (6): 1175–1184. doi:10.1016/0092-8674(91)90040-6. PMID 1833065. S2CID 54277241.
- ^ Takaki S, Murata Y, Kitamura T, Miyajima A, Tominaga A, Takatsu K (June 1993). "Reconstitution of the functional receptors for murine and human interleukin 5". The Journal of Experimental Medicine. 177 (6): 1523–1529. doi:10.1084/jem.177.6.1523. PMC 2191058. PMID 8496674.
- ^ Ishino T, Economou NJ, McFadden K, Zaks-Zilberman M, Jost M, Baxter S, et al. (September 2011). "A protein engineering approach differentiates the functional importance of carbohydrate moieties of interleukin-5 receptor α". Biochemistry. 50 (35): 7546–7556. doi:10.1021/bi2009135. PMID 21770429.