Norgestrel
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Ovral, others |
| Other names | dl-Norgestrel; DL-Norgestrel; (±)-Norgestrel; WY-3707; SH-70850; SH-850; FH 122-A; rac-13-Ethyl-17α-ethynyl-19-nortestosterone; rac-13-Ethyl-17α-ethynylestr-4-en-17β-ol-3-one |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a602008 |
| Routes of administration | By mouth |
| Drug class | Progestin; Progestogen |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.026.758 |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Norgestrel, sold under the brand name Ovral among others, is a progestin medication which is used in birth control pills and in menopausal hormone therapy.[1][2][3][4] It is available both in combination with an estrogen and alone.[5] It is taken by mouth.[3][4]
Side effects of norgestrel include menstrual irregularities, headaches, nausea, breast tenderness, mood changes, acne, increased hair growth, and others.[citation needed] Norgestrel is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[4] It has weak androgenic activity and no other important hormonal activity.[4]
Norgestrel was introduced for medical use, specifically in birth control pills, in 1966.[6][7] It was subsequently introduced for use in menopausal hormone therapy as well.[5] Norgestrel is sometimes referred to as a "second-generation" progestin.[8] It is marketed widely throughout the world.[5][2] Norgestrel is available as a generic medication.[9]
Medical uses
Norgestrel is used in combination with ethinylestradiol or quinestrol in combined birth control pills, alone in progestogen-only birth control pills, and in combination with estradiol or conjugated estrogens in menopausal hormone therapy.[5] It has also been used as an emergency contraceptive in the Yuzpe regimen.[10]
Side effects
Pharmacology
Pharmacodynamics
Norgestrel is a progestogen, or an agonist of the progesterone receptor.[4] The biological activity of norgestrel lies in the levo enantiomer, levonorgestrel, whereas the dextro isomer is inactive.[4] As such, norgestrel is identical in its hormonal activity to levonorgestrel except that it is half as potent by weight.[4] Levonorgestrel, and by extension norgestrel, have some androgenic activity, but no estrogenic, antimineralocorticoid, or glucocorticoid activity.[4]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG | |
|---|---|---|---|---|---|---|---|---|
| Levonorgestrel | 150–162 | 34a, 45 | 0 | 1–8 | 17–75 | 50 | 0 | |
| 5α-Dihydrolevonorgestrel | 50 | 38a | 0 | ? | ? | ? | ? | |
| 3α,5α-Tetrahydrolevonorgestrel | ? | ? | 0.4 | ? | ? | ? | ? | |
| 3β,5α-Tetrahydrolevonorgestrel | ? | ? | 2.4 | ? | ? | ? | ? | |
| Values are percentages (%). Reference ligands (100%) were promegestone for the PR, metribolone (a = mibolerone) for the AR, E2 for the ER, DEXA for the GR, aldosterone for the MR, DHT for SHBG, and cortisol for CBG. Sources:[4][11][12][13] | ||||||||
Pharmacokinetics
The pharmacokinetics of norgestrel have been reviewed.[14]
Chemistry
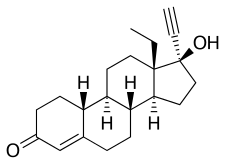
Norgestrel, also known as rac-13-ethyl-17α-ethynyl-19-nortestosterone or as rac-13-ethyl-17α-ethynylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone.[1][2] It is a racemic mixture of stereoisomers dextronorgestrel (the C13α isomer; l-norgestrel, L-norgestrel, or (+)-norgestrel) and levonorgestrel (the C13β isomer; d-norgestrel, D-norgestrel, or (–)-norgestrel), the former of which is inactive (making norgestrel exactly half as potent as levonorgestrel).[15][16] Norgestrel is more specifically a derivative of norethisterone (17α-ethynyl-19-nortestosterone) and is a member of the gonane (18-methylestrane) subgroup of the 19-nortestosterone family of progestins.[17]
Synthesis
Chemical syntheses of norgestrel have been published.[14]
History
Norgestrel was first introduced, as a birth control pill in combination with ethinylestradiol, under the brand name Eugynon in Germany in 1966.[6][7] It was subsequently marketed as a combined birth control pill with ethinylestradiol in the United States under the brand name Ovral in 1968, and was marketed in many other countries as well.[18][19][5]
Society and culture
Generic names
Norgestrel is the generic name of the drug and its INN, USAN, USP, BAN, DCF, DCIT, and JAN.[1][2][3][5] It is also known as dl-norgestrel, DL-norgestrel, or (±)-norgestrel.[1][2][3][5]
Brand names
Norgestrel has been marketed under a variety of brand names including Cyclacur, Cryselle, Cyclo-Progynova, Duoluton, Elinest, Eugynon, Microgynon, Lo/Ovral, Low-Ogestrel, Logynon, Microlut, Minicon, Nordette, Neogest, Ogestrel, Ovral, Ovran, Ovranette, Ovrette, Planovar, Prempak, Progyluton, and Trinordiol among others.[1][2][5][18]
See also
References
- ^ a b c d e J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 887–. ISBN 978-1-4757-2085-3.
- ^ a b c d e f Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 751–. ISBN 978-3-88763-075-1.
- ^ a b c d I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 202–. ISBN 978-94-011-4439-1.
- ^ a b c d e f g h i Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947.
- ^ a b c d e f g h https://www.drugs.com/international/norgestrel.html
- ^ a b Teresa Ortiz-Gómez; María Jesús Santesmases (22 April 2016). Gendered Drugs and Medicine: Historical and Socio-Cultural Perspectives. Taylor & Francis. pp. 175–. ISBN 978-1-317-12981-3.
The 1966 marketing campaign for Schering's second contraceptive, Eugynon, [...] (Schering AG Berline 1966, 11). [...] In 1970 [Schering] had already conducted an opinion poll among doctors in the run up to the marketing campaign for the newly introduced Neogynon. [...]
- ^ a b W. Gerhard Pohl (2004). Die wissenschaftliche Welt von gestern: die Preisträger des Ignaz L. Lieben-Preises 1865-1937 und des Richard Lieben-Preises 1912-1928 : ein Kapitel österreichischer Wissenschaftsgeschichte in Kurzbiografien. Böhlau Verlag Wien. pp. 150–. ISBN 978-3-205-77303-0.
[The contraceptive EUGYNON is launched in 1966. NEOGYNON follows in 1970.]
- ^ Howard J.A. Carp (9 April 2015). Progestogens in Obstetrics and Gynecology. Springer. p. 112. ISBN 978-3-319-14385-9.
- ^ https://www.drugs.com/availability/generic-lo-ovral-28.html
- ^ "A Multicenter Clinical Investigation Employing Ethinyl Estradiol Combined with dl-Norgestrel as Postcoital Contraceptive Agent". Fertility and Sterility. 37 (4): 508–513. 1982. PMID 7040117.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ Philibert D, Bouchoux F, Degryse M, Lecaque D, Petit F, Gaillard M (October 1999). "The pharmacological profile of a novel norpregnance progestin (trimegestone)". Gynecol. Endocrinol. 13 (5): 316–26. doi:10.3109/09513599909167574. PMID 10599548.
- ^ Santillán R, Pérez-Palacios G, Reyes M, Damián-Matsumura P, García GA, Grillasca I, Lemus AE (September 2001). "Assessment of the oestrogenic activity of the contraceptive progestin levonorgestrel and its non-phenolic metabolites". Eur. J. Pharmacol. 427 (2): 167–74. doi:10.1016/S0014-2999(01)01263-8. PMID 11557270.
- ^ Cabeza M, Vilchis F, Lemus AE, Díaz de León L, Pérez-Palacios G (September 1995). "Molecular interactions of levonorgestrel and its 5 alpha-reduced derivative with androgen receptors in hamster flanking organs". Steroids. 60 (9): 630–5. doi:10.1016/0039-128X(95)00075-2. PMID 8545853.
- ^ a b Die Gestagene. Springer-Verlag. 27 November 2013. pp. 16–17, 284–. ISBN 978-3-642-99941-3.
- ^ Brian K. Alldredge; Robin L. Corelli; Michael E. Ernst (1 February 2012). Koda-Kimble and Young's Applied Therapeutics: The Clinical Use of Drugs. Lippincott Williams & Wilkins. pp. 1072–. ISBN 978-1-60913-713-7.
- ^ J.P. Lavery; J.S. Sanfilippo (6 December 2012). Pediatric and Adolescent Obstetrics and Gynecology. Springer Science & Business Media. pp. 248–. ISBN 978-1-4612-5064-7.
- ^ Stefan Offermanns; W. Rosenthal (14 August 2008). Encyclopedia of Molecular Pharmacology. Springer Science & Business Media. pp. 390–. ISBN 978-3-540-38916-3.
- ^ a b William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 2935–. ISBN 978-0-8155-1856-3.
- ^ Lara Marks (2010). Sexual Chemistry: A History of the Contraceptive Pill. Yale University Press. pp. 73–. ISBN 978-0-300-16791-7.
