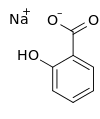
Sodium salicylate
| |
| Names | |
|---|---|
| IUPAC name
Sodium salicylate
| |
| Other names
Salsonin, Monosodium salicylate, Sodium o-hydroxybenzoate, Sodium 2-hydroxybenzoate, Salicylic acid sodium salt, Monosodium 2-hydroxybenzoate, Diuratin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.181 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H5NaO3 | |
| Molar mass | 160.104 g·mol−1 |
| Appearance | white crystals |
| Melting point | 200 °C (392 °F; 473 K) |
| 25.08 g/100 mL (-1.5 °C) 107.9 g/100 mL (15 °C) 124.6 g/100 mL (25 °C) 141.8 g/100 mL (78.5 °C) 179 g/100 g (114 °C)[1] | |
| Solubility | soluble in glycerol, 1,4-Dioxane, alcohol[1] |
| Solubility in methanol | 26.28 g/100 g (15 °C) 34.73 g/100 g (67.2 °C)[1] |
| Pharmacology | |
| N02BA04 (WHO) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Harmful |
Eye hazards
|
Irritant |
| GHS labelling: | |
 [2] [2]
| |
| Warning | |
| H314, H331, H400[2] | |
| P210, P261, P273, P280, P305+P351+P338, P310[2] | |
| NFPA 704 (fire diamond) | |
| 250 °C (482 °F; 523 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
930 mg/kg (rats, oral)[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium salicylate is a sodium salt of salicylic acid. It can be prepared from sodium phenolate and carbon dioxide under higher temperature and pressure. Historically, it has been synthesized from methyl salicylate (found in wintergreen plants or the bark of sweet birch tree) by reacting it with an excess of sodium hydroxide and heating it under reflux.[4]
Properties
Sodium salicylate is of the salicylate family and this compound is known to trigger Reye's Syndrome in children and adults, usually following a viral infection such as influenza or chicken pox. Products containing such salicylates should not be given to children under the age of 19.[citation needed]
Uses
It is used in medicine as an analgesic and antipyretic. Sodium salicylate also acts as non-steroidal anti-inflammatory drug (NSAID), and induces apoptosis in cancer cells [5][6][7] and also necrosis.[8] It is also a potential replacement for aspirin for people sensitive to it. It may also be used as a phosphor for the detection of vacuum ultraviolet radiation and electrons.[9]
References
- ^ a b c http://chemister.ru/Database/properties-en.php?dbid=1&id=2993
- ^ a b c Sigma-Aldrich Co., Sodium salicylate. Retrieved on 2014-05-26.
- ^ http://chem.sis.nlm.nih.gov/chemidplus/rn/54-21-7
- ^ Lehman, J.W., Operational Organich Chemistry, 4th ed., New Jersey, Prentice Hall, 2009
- ^ Klampfer, Lidija; Jörg Cammenga; Hans-Georg Wisniewski; Stephen D. Nimer (1999-04-01). "Sodium Salicylate Activates Caspases and Induces Apoptosis of Myeloid Leukemia Cell Lines". Blood. 93 (7): 2386–94. PMID 10090950. Retrieved 2011-02-21.
- ^ Rae, Colin; Susana Langa; Steven J. Tucker; David J. MacEwan (2007-07-31). "Elevated NF-κB responses and FLIP levels in leukemic but not normal lymphocytes: reduction by salicylate allows TNF-induced apoptosis". Proceedings of the National Academy of Sciences of the USA. 104 (31): 12790–5. doi:10.1073/pnas.0701437104. PMC 1937545. PMID 17646662. Retrieved 2011-02-21.
- ^ Stark, Lesley A.; et al. (May 2007). "Aspirin activates the NF-κB signalling pathway and induces apoptosis in intestinal neoplasia in two in vivo models of human colorectal cancer". Carcinogenesis. 28 (5): 968–76. doi:10.1093/carcin/bgl220. PMID 17132819. Retrieved 2011-02-21.
- ^ Schwenger, Paul; Edward Y. Skolnik; Jan Vilcek (1996-04-05). "Inhibition of Tumor Necrosis Factor-induced p42/p44 Mitogen-Activated Protein Kinase Activation by Sodium Salicylate". The Journal of Biological Chemistry. 271 (14): 8089–94. doi:10.1074/jbc.271.14.8089. PMID 8626494. Retrieved 2011-02-21.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Samson, James. "Vacuum Ultraviolet Spectroscopy" (PDF). Pied Publications. Retrieved July 26, 2012.

