Arecoline: Difference between revisions
m Reverted edits by 220.132.102.226 (talk) to last version by DePiep |
|||
| Line 67: | Line 67: | ||
== Biological action == |
== Biological action == |
||
In many Asian cultures, |
In many Asian cultures, [[areca nut|buai]] is chewed along with [[betel leaf]] to obtain a [[stimulant|stimulating]] effect.<ref>{{cite journal |author=Gupta Prakash Chandra, Ray Cecily S |title=Epidemiology of betel quid usage |journal=Ann. Acad. Med. Singap. |volume=33 |issue=4 Suppl |pages=31–6 |date=July 2004 |pmid=15389304 |doi= |url=http://www.annals.edu.sg/pdf200409/V33N4p31S.pdf}}</ref> Arecoline is the primary active ingredient responsible for the central nervous system effects of the areca nut. Arecoline has been compared to [[nicotine]]; however, nicotine acts primarily on the [[nicotinic acetylcholine receptor]]. Arecoline is known to be a partial agonist of [[Muscarinic acetylcholine receptor#Form of muscarinic receptors|muscarinic]] [[acetylcholine]] M<sub>1</sub>, M<sub>2</sub>, M<sub>3</sub> receptors and M<sub>4</sub>,<ref name="ArecolineM1-Ghelardini"/><ref name="ArecolineM2-Yang">{{cite journal | author=Yang YR, Chang KC, Chen CL, Chiu TH. | title=Arecoline excites rat locus coeruleus neurons by activating the M2-muscarinic receptor. | journal=Chin J Physiol. | year=2000 | pages=23–8 | volume=43 | issue=1 | pmid=10857465}}</ref><ref name="ArecolineM3-Xie">{{cite journal | author=Xie DP, Chen LB, Liu CY, Zhang CL, Liu KJ, Wang PS. | title=Arecoline excites the colonic smooth muscle motility via M3 receptor in rabbits. | journal=Chin J Physiol. | year=2004 | pages=89–94 | volume=47 | issue=2 | pmid=15481791}}</ref> which is believed to be the primary cause of its [[parasympathetic]] effects (such as pupillary constriction, bronchial constriction, etc.). |
||
{{LD50}}: 100 mg/kg, administered subcutaneously in mouse.<ref name = Merck/> |
{{LD50}}: 100 mg/kg, administered subcutaneously in mouse.<ref name = Merck/> |
||
Revision as of 02:02, 21 August 2015
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.514 |
| Chemical and physical data | |
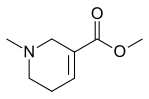
| Formula | C8H13NO2 |
| Molar mass | 155.194 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.0495 g/cm3 |
| Boiling point | 209 °C (408 °F) |
| |
| |
| | |
Arecoline (/əˈrɛkəliːn/) is a nicotinic acid-based alkaloid found in the areca nut, the fruit of the areca palm (Areca catechu).[1] It is an odourless oily liquid.
Chemistry
Arecoline is a base, and its conjugate acid has a pKa ~ 6.8.[2] Arecoline is volatile in steam, miscible with most organic solvents and water, but extractable from water by ether in presence of dissolved salts. Being basic, arecoline forms salts with acids. The salts are crystalline, but usually deliquescent: the hydrochloride, arecoline•HCl, forms needles, m.p. 158°C;[2] the hydrobromide, arecoline•HBr, forms slender prisms, mp. 177-179 °C from hot alcohol; the aurichloride, arecoline•HAuCl4, is an oil, but the platinichloride, arecoline2•H2PtCl6, mp. 176 °C, crystallizes from water in orange-red rhombohedrons. The methiodide forms glancing prisms, mp. 173-174 °C.
Biological action
In many Asian cultures, buai is chewed along with betel leaf to obtain a stimulating effect.[3] Arecoline is the primary active ingredient responsible for the central nervous system effects of the areca nut. Arecoline has been compared to nicotine; however, nicotine acts primarily on the nicotinic acetylcholine receptor. Arecoline is known to be a partial agonist of muscarinic acetylcholine M1, M2, M3 receptors and M4,[1][4][5] which is believed to be the primary cause of its parasympathetic effects (such as pupillary constriction, bronchial constriction, etc.).
LD50: 100 mg/kg, administered subcutaneously in mouse.[2]
Uses
Owing to its muscarinic and nicotinic agonist properties, arecoline has shown improvement in the learning ability of healthy volunteers. Since one of the hallmarks of Alzheimer's disease is a cognitive decline, arecoline was suggested as a treatment to slow down this process and arecoline administered intravenously did indeed show modest verbal and spatial memory improvement in Alzheimer's patients, though due to arecoline's possible carcinogenic properties,[6] it is not the first drug of choice for this degenerative disease.[7]
Arecoline has also been used medicinally as an antihelmintic (a drug against parasitic worms).[8]
References
- ^ a b Ghelardini C, Galeotti N, Lelli C, Bartolini A. (2001). "Arecoline M1 receptor activation is a requirement for arecoline analgesia". Farmaco. 56 (5–7): 383–5. doi:10.1016/S0014-827X(01)01091-6. PMID 11482763.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c The Merck Index, 10th Ed. (1983) p.113, Rahway: Merck & Co.
- ^ Gupta Prakash Chandra, Ray Cecily S (July 2004). "Epidemiology of betel quid usage" (PDF). Ann. Acad. Med. Singap. 33 (4 Suppl): 31–6. PMID 15389304.
- ^ Yang YR, Chang KC, Chen CL, Chiu TH. (2000). "Arecoline excites rat locus coeruleus neurons by activating the M2-muscarinic receptor". Chin J Physiol. 43 (1): 23–8. PMID 10857465.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Xie DP, Chen LB, Liu CY, Zhang CL, Liu KJ, Wang PS. (2004). "Arecoline excites the colonic smooth muscle motility via M3 receptor in rabbits". Chin J Physiol. 47 (2): 89–94. PMID 15481791.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Saikia JR, Schneeweiss FH, Sharan RN. (1999). "Arecoline-induced changes of poly-ADP-ribosylation of cellular proteins and its influence on chromatin organization". Cancer Letters. 139 (1): 59–65. doi:10.1016/S0304-3835(99)00008-7. PMID 10408909.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Christie JE, Shering A, Ferguson J (1981). "Physostigmine and arecoline: effects of intravenous infusions in Alzheimer's presenile dementia". British Journal of Psychiatry. 138 (1): 46–50. doi:10.1192/bjp.138.1.46. PMID 7023592.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Yusuf H, Yong SL (2002). "Oral submucous fibrosis in a 12-year-old Bangladeshi boy: a case report and review of literature". International journal of paediatric dentistry / the British Paedodontic Society [and] the International Association of Dentistry for Children. 12 (4): 271–6. PMID 12121538.
