Intermediate filament
| Intermediate filament tail domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
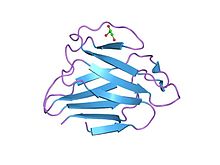 Structure of lamin a/c globular domain | |||||||||
| Identifiers | |||||||||
| Symbol | IF_tail | ||||||||
| Pfam | PF00932 | ||||||||
| InterPro | IPR001322 | ||||||||
| PROSITE | PDOC00198 | ||||||||
| SCOP2 | 1ivt / SCOPe / SUPFAM | ||||||||
| |||||||||
| Intermediate filament rod domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Human vimentin coil 2b fragment (cys2) | |||||||||
| Identifiers | |||||||||
| Symbol | Filament | ||||||||
| Pfam | PF00038 | ||||||||
| InterPro | IPR016044 | ||||||||
| PROSITE | PDOC00198 | ||||||||
| SCOP2 | 1gk7 / SCOPe / SUPFAM | ||||||||
| |||||||||
| Intermediate filament head (DNA binding) region | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Filament_head | ||||||||
| Pfam | PF04732 | ||||||||
| InterPro | IPR006821 | ||||||||
| SCOP2 | 1gk7 / SCOPe / SUPFAM | ||||||||
| |||||||||
| Peripherin neuronal intermediate filament protein | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | PRPH | ||||||
| Alt. symbols | NEF4 | ||||||
| NCBI gene | 5630 | ||||||
| HGNC | 9461 | ||||||
| OMIM | 170710 | ||||||
| RefSeq | NM_006262.3 | ||||||
| UniProt | P41219 | ||||||
| Other data | |||||||
| Locus | Chr. 12 q13.12 | ||||||
| |||||||
| Nestin neuronal stem cell intermediate filament protein | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | NES | ||||||
| NCBI gene | 10763 | ||||||
| HGNC | 7756 | ||||||
| OMIM | 600915 | ||||||
| RefSeq | NP_006608 | ||||||
| UniProt | P48681 | ||||||
| Other data | |||||||
| Locus | Chr. 1 q23.1 | ||||||
| |||||||
Intermediate filaments (IFs) are cytoskeletal structural components found in the cells of vertebrates, and many invertebrates.[1][2][3] Homologues of the IF protein have been noted in an invertebrate, the cephalochordate Branchiostoma.[4]
Intermediate filaments are composed of a family of related proteins sharing common structural and sequence features. Initially designated 'intermediate' because their average diameter (10 nm) is between those of narrower microfilaments (actin) and wider myosin filaments found in muscle cells, the diameter of intermediate filaments is now commonly compared to actin microfilaments (7 nm) and microtubules (25 nm).[1][5] Animal intermediate filaments are subcategorized into six types based on similarities in amino acid sequence and protein structure.[6] Most types are cytoplasmic, but one type, Type V is a nuclear lamin. Unlike microtubules, IF distribution in cells show no good correlation with the distribution of either mitochondria or endoplasmic reticulum.[7]
Structure

The structure of proteins that form intermediate filaments (IF) was first predicted by computerized analysis of the amino acid sequence of a human epidermal keratin derived from cloned cDNAs.[8] Analysis of a second keratin sequence revealed that the two types of keratins share only about 30% amino acid sequence homology but share similar patterns of secondary structure domains.[9] As suggested by the first model, all IF proteins appear to have a central alpha-helical rod domain that is composed of four alpha-helical segments (named as 1A, 1B, 2A and 2B) separated by three linker regions.[9][10]
The central building block of an intermediate filament is a pair of two intertwined proteins that is called a coiled-coil structure. This name reflects the fact that the structure of each protein is helical, and the intertwined pair is also a helical structure. Structural analysis of a pair of keratins shows that the two proteins that form the coiled-coil bind by hydrophobic interactions.[11][12] The charged residues in the central domain do not have a major role in the binding of the pair in the central domain.[11]
Cytoplasmic IFs assemble into non-polar unit-length filaments (ULFs). Identical ULFs associate laterally into staggered, antiparallel, soluble tetramers, which associate head-to-tail into protofilaments that pair up laterally into protofibrils, four of which wind together into an intermediate filament.[13] Part of the assembly process includes a compaction step, in which ULF tighten and assume a smaller diameter. The reasons for this compaction are not well understood, and IF are routinely observed to have diameters ranging between 6 and 12 nm.
The N-terminus and the C-terminus of IF proteins are non-alpha-helical regions and show wide variation in their lengths and sequences across IF families. The N-terminal "head domain" binds DNA.[14] Vimentin heads are able to alter nuclear architecture and chromatin distribution, and the liberation of heads by HIV-1 protease may play an important role in HIV-1 associated cytopathogenesis and carcinogenesis.[15] Phosphorylation of the head region can affect filament stability.[16] The head has been shown to interact with the rod domain of the same protein.[17]
C-terminal "tail domain" shows extreme length variation between different IF proteins.[18]
The anti-parallel orientation of tetramers means that, unlike microtubules and microfilaments, which have a plus end and a minus end, IFs lack polarity and cannot serve as basis for cell motility and intracellular transport.
Also, unlike actin or tubulin, intermediate filaments do not contain a binding site for a nucleoside triphosphate.
Cytoplasmic IFs do not undergo treadmilling like microtubules and actin fibers, but are dynamic.[19]
Biomechanical properties
IFs are rather deformable proteins that can be stretched several times their initial length.[20] The key to facilitate this large deformation is due to their hierarchical structure, which facilitates a cascaded activation of deformation mechanisms at different levels of strain.[12] Initially the coupled alpha-helices of unit-length filaments uncoil as they're strained, then as the strain increases they transition into beta-sheets, and finally at increased strain the hydrogen bonds between beta-sheets slip and the ULF monomers slide along each other.[12]
Types
There are about 70 different human genes coding for various intermediate filament proteins. However, different kinds of IFs share basic characteristics: In general, they are all polymers that measure between 9–11 nm in diameter when fully assembled.
Animal IFs are subcategorized into six types based on similarities in amino acid sequence and protein structure:[6]
Types I and II – acidic and basic keratins
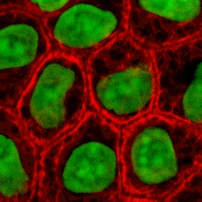
These proteins are the most diverse among IFs and constitute type I (acidic) and type II (basic) IF proteins. The many isoforms are divided in two groups:
- epithelial keratins (about 20) in epithelial cells (image to right)
- trichocytic keratins (about 13) (hair keratins), which make up hair, nails, horns and reptilian scales.
Regardless of the group, keratins are either acidic or basic. Acidic and basic keratins bind each other to form acidic-basic heterodimers and these heterodimers then associate to make a keratin filament.[6]
Cytokeratin filaments laterally associate with each other to create a thick bundle of ~50 nm radius. The optimal radius of such bundles is determined by the interplay between the long range electrostatic repulsion and short range hydrophobic attraction.[21] Subsequently, these bundles would intersect through junctions to form a dynamic network, spanning the cytoplasm of epithelial cells.
Type III

There are four proteins classed as type III intermediate filament proteins, which may form homo- or heteropolymeric proteins.
- Desmin IFs are structural components of the sarcomeres in muscle cells and connect different cell organells like the desmosomes with the cytoskeleton.[22]
- Glial fibrillary acidic protein (GFAP) is found in astrocytes and other glia.
- Peripherin found in peripheral neurons.
- Vimentin, the most widely distributed of all IF proteins, can be found in fibroblasts, leukocytes, and blood vessel endothelial cells. They support the cellular membranes, keep some organelles in a fixed place within the cytoplasm, and transmit membrane receptor signals to the nucleus.[6]
- Syncoilin is an atypical type III IF protein.[23]
Type IV
- Alpha-internexin
- Neurofilaments - the type IV family of intermediate filaments that is found in high concentrations along the axons of vertebrate neurons.
- Synemin
- Syncoilin
Type V – nuclear lamins
Lamins are fibrous proteins having structural function in the cell nucleus.
In metazoan cells, there are A and B type lamins, which differ in their length and pI. Human cells have three differentially regulated genes. B-type lamins are present in every cell. B type lamins, lamin B1 and B2, are expressed from the LMNB1 and LMNB2 genes on 5q23 and 19q13, respectively. A-type lamins are only expressed following gastrulation. Lamin A and C are the most common A-type lamins and are splice variants of the LMNA gene found at 1q21.
These proteins localize to two regions of the nuclear compartment, the nuclear lamina—a proteinaceous structure layer subjacent to the inner surface of the nuclear envelope and throughout the nucleoplasm in the nucleoplasmic veil.
Comparison of the lamins to vertebrate cytoskeletal IFs shows that lamins have an extra 42 residues (six heptads) within coil 1b. The c-terminal tail domain contains a nuclear localization signal (NLS), an Ig-fold-like domain, and in most cases a carboxy-terminal CaaX box that is isoprenylated and carboxymethylated (lamin C does not have a CAAX box). Lamin A is further processed to remove the last 15 amino acids and its farnesylated cysteine.
During mitosis, lamins are phosphorylated by MPF, which drives the disassembly of the lamina and the nuclear envelope.[6]
Type VI
- Beaded filaments: Filensin, Phakinin.[6]
- Nestin (was once proposed for reclassification but due to differences, remains as a type VI IF protein)[24]
Vertebrate-only. Related to type I-IV. Used to contain other newly-discovered IF proteins not yet assigned to a type.[25]
Function
Cell adhesion
At the plasma membrane, some keratins or desmin interact with desmosomes (cell-cell adhesion) and hemidesmosomes (cell-matrix adhesion) via adapter proteins.
Associated proteins
Filaggrin binds to keratin fibers in epidermal cells. Plectin links vimentin to other vimentin fibers, as well as to microfilaments, microtubules, and myosin II. Kinesin is being researched and is suggested to connect vimentin to tubulin via motor proteins.
Keratin filaments in epithelial cells link to desmosomes (desmosomes connect the cytoskeleton together) through plakoglobin, desmoplakin, desmogleins, and desmocollins; desmin filaments are connected in a similar way in heart muscle cells.
Diseases arising from mutations in IF genes
- Dilated cardiomyoathy (DCM), mutations in the DES gene[26]
- Arrhythmogenic cardiomyopathy (ACM), mutations in the DES gene[27][28][29][30]
- Restrictive cardiomyopathy (RCM), mutations in the DES gene[31]
- Non-compaction cardiomyopathy, mutations in the DES genes[32][33]
- Cardiomyopathy in combination with skeletal myopathy (DES)[34]
- Epidermolysis bullosa simplex; keratin 5 or keratin 14 mutation
- Laminopathies are a family of diseases caused by mutations in nuclear lamins and include Hutchinson-Gilford progeria syndrome and various lipodystrophies and cardiomyopathies among others.
In other organisms
IF proteins are universal among animals in the form of a nuclear lamin. The Hydra has an additional "nematocilin" derived from the lamin. Cytoplasmic IFs (type I-IV) are only found in Bilateria; they also arose from a gene duplication event involving "type V" nuclear lamin. In addition, a few other diverse types of eukaryotes have lamins, suggesting an early origin of the protein.[25]
There was not really a concrete definition of an "intermediate filament protein", in the sense that the size or shape-based definition does not cover a monophyletic group. With the inclusion of unusual proteins like the network-forming beaded lamins (type VI), the current classification is moving to a clade containing nuclear lamin and its many descendents, characterized by sequence similarity as well as the exon structure. Functionally-similar proteins out of this clade, like crescentins, alveolins, tetrins, and epiplasmins, are therefore only "IF-like". They likely arose through convergent evolution.[25]
References
- ^ a b Herrmann H, Bär H, Kreplak L, Strelkov SV, Aebi U (July 2007). "Intermediate filaments: from cell architecture to nanomechanics". Nature Reviews. Molecular Cell Biology. 8 (7): 562–73. doi:10.1038/nrm2197. PMID 17551517. S2CID 27115011.
- ^ Chang L, Goldman RD (August 2004). "Intermediate filaments mediate cytoskeletal crosstalk". Nature Reviews. Molecular Cell Biology. 5 (8): 601–13. doi:10.1038/nrm1438. PMID 15366704. S2CID 31835055.
- ^ Intermediate Filaments: A Review, Springer Berlin Heidelberg, 2012, p. 33, ISBN 9783642702303
{{citation}}: Unknown parameter|authors=ignored (help) - ^ Karabinos A, Riemer D, Erber A, Weber K (October 1998). "Homologues of vertebrate type I, II and III intermediate filament (IF) proteins in an invertebrate: the IF multigene family of the cephalochordate Branchiostoma". FEBS Letters. 437 (1–2): 15–8. doi:10.1016/S0014-5793(98)01190-9. PMID 9804163. S2CID 7886395.
- ^ Ishikawa H, Bischoff R, Holtzer H (September 1968). "Mitosis and intermediate-sized filaments in developing skeletal muscle". The Journal of Cell Biology. 38 (3): 538–55. doi:10.1083/jcb.38.3.538. PMC 2108373. PMID 5664223.
- ^ a b c d e f Szeverenyi I, Cassidy AJ, Chung CW, Lee BT, Common JE, Ogg SC, et al. (March 2008). "The Human Intermediate Filament Database: comprehensive information on a gene family involved in many human diseases". Human Mutation. 29 (3): 351–360. doi:10.1002/humu.20652. PMID 18033728. S2CID 20760837.
- ^ Soltys BJ, Gupta RS (1992). "Interrelationships of endoplasmic reticulum, mitochondria, intermediate filaments, and microtubules--a quadruple fluorescence labeling study". Biochemistry and Cell Biology. 70 (10–11): 1174–86. doi:10.1139/o92-163. PMID 1363623.
- ^ Hanukoglu I, Fuchs E (November 1982). "The cDNA sequence of a human epidermal keratin: divergence of sequence but conservation of structure among intermediate filament proteins". Cell. 31 (1): 243–52. doi:10.1016/0092-8674(82)90424-X. PMID 6186381. S2CID 35796315.
- ^ a b Hanukoglu I, Fuchs E (July 1983). "The cDNA sequence of a Type II cytoskeletal keratin reveals constant and variable structural domains among keratins". Cell. 33 (3): 915–24. doi:10.1016/0092-8674(83)90034-X. PMID 6191871. S2CID 21490380.
- ^ Lee CH, Kim MS, Chung BM, Leahy DJ, Coulombe PA (June 2012). "Structural basis for heteromeric assembly and perinuclear organization of keratin filaments". Nature Structural & Molecular Biology. 19 (7): 707–15. doi:10.1038/nsmb.2330. PMC 3864793. PMID 22705788.
- ^ a b Hanukoglu I, Ezra L (Jan 2014). "Proteopedia entry: coiled-coil structure of keratins". Biochemistry and Molecular Biology Education. 42 (1): 93–4. doi:10.1002/bmb.20746. PMID 24265184. S2CID 30720797.
- ^ a b c Qin Z, Kreplak L, Buehler MJ (October 2009). "Hierarchical structure controls nanomechanical properties of vimentin intermediate filaments". PLOS ONE. 4 (10): e7294. Bibcode:2009PLoSO...4.7294Q. doi:10.1371/journal.pone.0007294. PMC 2752800. PMID 19806221.
- ^ Lodish H, Berk A, Zipursky SL, et al. (2000). Molecular Cell Biology. New York: W. H. Freeman. p. Section 19.6, Intermediate Filaments. ISBN 978-0-07-243940-3.
- ^ Wang Q, Tolstonog GV, Shoeman R, Traub P (August 2001). "Sites of nucleic acid binding in type I-IV intermediate filament subunit proteins". Biochemistry. 40 (34): 10342–9. doi:10.1021/bi0108305. PMID 11513613.
- ^ Shoeman RL, Hüttermann C, Hartig R, Traub P (January 2001). "Amino-terminal polypeptides of vimentin are responsible for the changes in nuclear architecture associated with human immunodeficiency virus type 1 protease activity in tissue culture cells". Molecular Biology of the Cell. 12 (1): 143–54. doi:10.1091/mbc.12.1.143. PMC 30574. PMID 11160829.
- ^ Takemura M, Gomi H, Colucci-Guyon E, Itohara S (August 2002). "Protective role of phosphorylation in turnover of glial fibrillary acidic protein in mice". The Journal of Neuroscience. 22 (16): 6972–9. doi:10.1523/JNEUROSCI.22-16-06972.2002. PMC 6757867. PMID 12177195.
- ^ Parry DA, Marekov LN, Steinert PM, Smith TA (2002). "A role for the 1A and L1 rod domain segments in head domain organization and function of intermediate filaments: structural analysis of trichocyte keratin". Journal of Structural Biology. 137 (1–2): 97–108. doi:10.1006/jsbi.2002.4437. PMID 12064937.
- ^ Quinlan R, Hutchison C, Lane B (1995). "Intermediate filament proteins". Protein Profile. 2 (8): 795–952. PMID 8771189.
- ^ Helfand BT, Chang L, Goldman RD (January 2004). "Intermediate filaments are dynamic and motile elements of cellular architecture". Journal of Cell Science. 117 (Pt 2): 133–41. doi:10.1242/jcs.00936. PMID 14676269.
- ^ Herrmann H, Bär H, Kreplak L, Strelkov SV, Aebi U (July 2007). "Intermediate filaments: from cell architecture to nanomechanics". Nature Reviews. Molecular Cell Biology. 8 (7): 562–73. doi:10.1038/nrm2197. PMID 17551517. S2CID 27115011.Qin Z, Kreplak L, Buehler MJ (October 2009). "Hierarchical structure controls nanomechanical properties of vimentin intermediate filaments". PLOS ONE. 4 (10): e7294. Bibcode:2009PLoSO...4.7294Q. doi:10.1371/journal.pone.0007294. PMC 2752800. PMID 19806221.Kreplak L, Fudge D (January 2007). "Biomechanical properties of intermediate filaments: from tissues to single filaments and back". BioEssays. 29 (1): 26–35. doi:10.1002/bies.20514. PMID 17187357. S2CID 6560740.Qin Z, Buehler MJ, Kreplak L (January 2010). "A multi-scale approach to understand the mechanobiology of intermediate filaments". Journal of Biomechanics. 43 (1): 15–22. doi:10.1016/j.jbiomech.2009.09.004. PMID 19811783.Qin Z, Kreplak L, Buehler MJ (October 2009). "Nanomechanical properties of vimentin intermediate filament dimers". Nanotechnology. 20 (42): 425101. Bibcode:2009Nanot..20P5101Q. doi:10.1088/0957-4484/20/42/425101. PMID 19779230. S2CID 6870454.
- ^ Haimov E, Windoffer R, Leube RE, Urbakh M, Kozlov MM (July 2020). "Model for Bundling of Keratin Intermediate Filaments". Biophysical Journal. 119 (1): 65–74. Bibcode:2020BpJ...119...65H. doi:10.1016/j.bpj.2020.05.024. PMC 7335914. PMID 32533940.
- ^ Brodehl A, Gaertner-Rommel A, Milting H (August 2018). "Molecular insights into cardiomyopathies associated with desmin (DES) mutations". Biophysical Reviews. 10 (4): 983–1006. doi:10.1007/s12551-018-0429-0. PMC 6082305. PMID 29926427.
- ^ "SYNC - Syncoilin - Homo sapiens (Human) - SYNC gene & protein". www.uniprot.org. Retrieved 20 December 2021.
- ^ Bernal A, Arranz L (June 2018). "Nestin-expressing progenitor cells: function, identity and therapeutic implications". Cellular and Molecular Life Sciences. 75 (12): 2177–2195. doi:10.1007/s00018-018-2794-z. PMC 5948302. PMID 29541793.
- ^ a b c Kollmar M (May 2015). "Polyphyly of nuclear lamin genes indicates an early eukaryotic origin of the metazoan-type intermediate filament proteins". Scientific Reports. 5: 10652. Bibcode:2015NatSR...510652K. doi:10.1038/srep10652. PMC 4448529. PMID 26024016.
- ^ Fischer B, Dittmann S, Brodehl A, Unger A, Stallmeyer B, Paul M, et al. (December 2020). "Functional characterization of novel alpha-helical rod domain desmin (DES) pathogenic variants associated with dilated cardiomyopathy, atrioventricular block and a risk for sudden cardiac death". International Journal of Cardiology. 329: 167–174. doi:10.1016/j.ijcard.2020.12.050. PMID 33373648. S2CID 229719883.
- ^ Bermúdez-Jiménez FJ, Carriel V, Brodehl A, Alaminos M, Campos A, Schirmer I, et al. (April 2018). "Novel Desmin Mutation p.Glu401Asp Impairs Filament Formation, Disrupts Cell Membrane Integrity, and Causes Severe Arrhythmogenic Left Ventricular Cardiomyopathy/Dysplasia". Circulation. 137 (15): 1595–1610. doi:10.1161/CIRCULATIONAHA.117.028719. PMID 29212896. S2CID 4715358.
- ^ Protonotarios A, Brodehl A, Asimaki A, Jager J, Quinn E, Stanasiuk C, et al. (December 2020). "The novel desmin variant p.Leu115Ile is associated with a unique form of biventricular Arrhythmogenic Cardiomyopathy". The Canadian Journal of Cardiology. 37 (6): 857–866. doi:10.1016/j.cjca.2020.11.017. PMID 33290826. S2CID 228078648.
- ^ Klauke B, Kossmann S, Gaertner A, Brand K, Stork I, Brodehl A, et al. (December 2010). "De novo desmin-mutation N116S is associated with arrhythmogenic right ventricular cardiomyopathy". Human Molecular Genetics. 19 (23): 4595–607. doi:10.1093/hmg/ddq387. PMID 20829228.
- ^ Brodehl A, Hedde PN, Dieding M, Fatima A, Walhorn V, Gayda S, et al. (May 2012). "Dual color photoactivation localization microscopy of cardiomyopathy-associated desmin mutants". The Journal of Biological Chemistry. 287 (19): 16047–57. doi:10.1074/jbc.M111.313841. PMC 3346104. PMID 22403400.
- ^ Brodehl A, Pour Hakimi SA, Stanasiuk C, Ratnavadivel S, Hendig D, Gaertner A, et al. (November 2019). "Restrictive Cardiomyopathy is Caused by a Novel Homozygous Desmin (DES) Mutation p.Y122H Leading to a Severe Filament Assembly Defect". Genes. 10 (11): 918. doi:10.3390/genes10110918. PMC 6896098. PMID 31718026.
- ^ Kley RA, Hellenbroich Y, van der Ven PF, Fürst DO, Huebner A, Bruchertseifer V, et al. (December 2007). "Clinical and morphological phenotype of the filamin myopathy: a study of 31 German patients". Brain: A Journal of Neurology. 130 (Pt 12): 3250–64. doi:10.1093/brain/awm271. PMID 18055494.
- ^ Marakhonov AV, Brodehl A, Myasnikov RP, Sparber PA, Kiseleva AV, Kulikova OV, et al. (June 2019). "Noncompaction cardiomyopathy is caused by a novel in-frame desmin (DES) deletion mutation within the 1A coiled-coil rod segment leading to a severe filament assembly defect". Human Mutation. 40 (6): 734–741. doi:10.1002/humu.23747. PMID 30908796. S2CID 85515283.
- ^ Schirmer I, Dieding M, Klauke B, Brodehl A, Gaertner-Rommel A, Walhorn V, et al. (March 2018). "A novel desmin (DES) indel mutation causes severe atypical cardiomyopathy in combination with atrioventricular block and skeletal myopathy". Molecular Genetics & Genomic Medicine. 6 (2): 288–293. doi:10.1002/mgg3.358. PMC 5902401. PMID 29274115.
Further reading
- Herrmann H, Harris JR, eds. (1998). Intermediate filaments. Springer. ISBN 978-0-306-45854-5.
- Omary MB, Coulombe PA, eds. (2004). Intermediate filament cytoskeleton. Gulf Professional Publishing. ISBN 978-0-12-564173-9.
- Paramio JM, ed. (2006). Intermediate filaments. Springer. ISBN 978-0-387-33780-7.
External links
- Intermediate+Filament+Proteins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
