Biofuel

| Part of a series on |
| Renewable energy |
|---|
 |
Biofuel is a fuel that is produced over a short time span from biomass, rather than by the very slow natural processes involved in the formation of fossil fuels such as oil. Biofuel can be produced from plants or from agricultural, domestic or industrial biowaste.[1] Biofuels are mostly used for transportation, but can also be used for heating and electricity.[2]: 173 [3] Biofuels (and bioenergy in general) are regarded as a renewable energy source.[4]: 11 The use of biofuel has been subject to criticism regarding the "food vs fuel" debate, varied assessments of their sustainability, and possible deforestation and biodiversity loss as a result of biofuel production.
In general, biofuels emit fewer greenhouse gas emissions when burned in an engine and are generally considered carbon-neutral fuels as the carbon emitted has been captured from the atmosphere by the crops used in production.[5] However, life-cycle assessments of biofuels have shown large emissions associated with the potential land-use change required to produce additional biofuel feedstocks.[6][7] The outcomes of lifecycle assessments (LCAs) for biofuels are highly situational and dependent on many factors including the type of feedstock, production routes, data variations, and methodological choices. This could be added to emphasize the complexity and variability in assessing the environmental impacts of biofuels.[8] Estimates about the climate impact from biofuels vary widely based on the methodology and exact situation examined.[6] Therefore, the climate change mitigation potential of biofuel varies considerably: in some scenarios emission levels are comparable to fossil fuels, and in other scenarios the biofuel emissions result in negative emissions.
Global demand for biofuels is predicted to increase by 56% over 2022–2027.[9] By 2027 worldwide biofuel production is expected to supply 5.4% of the world's fuels for transport including 1% of aviation fuel.[10] Demand for aviation biofuel is forecast to increase.[11][12]
The two most common types of biofuel are bioethanol and biodiesel. Brazil is the largest producer of bioethanol, while the EU is the largest producer of biodiesel. The energy content in the global production of bioethanol and biodiesel is 2.2 and 1.8 EJ per year, respectively.[13]
Bioethanol is an alcohol made by fermentation, mostly from carbohydrates produced in sugar or starch crops such as maize, sugarcane, or sweet sorghum. Cellulosic biomass, derived from non-food sources, such as trees and grasses, is also being developed as a feedstock for ethanol production. Ethanol can be used as a fuel for vehicles in its pure form (E100), but it is usually used as a gasoline additive to increase octane ratings and improve vehicle emissions.
Biodiesel is produced from oils or fats using transesterification. It can be used as a fuel for vehicles in its pure form (B100), but it is usually used as a diesel additive to reduce levels of particulates, carbon monoxide, and hydrocarbons from diesel-powered vehicles.[14]
Terminology
[edit]
The term biofuel is used in different ways. One definition is "Biofuels are biobased products, in solid, liquid, or gaseous forms. They are produced from crops or natural products, such as wood, or agricultural residues, such as molasses and bagasse."[2]: 173
Other publications reserve the term biofuel for liquid or gaseous fuels, used for transportation.[3]
The IPCC Sixth Assessment Report defines biofuel as "A fuel, generally in liquid form, produced from biomass. Biofuels include bioethanol from sugarcane, sugar beet or maize, and biodiesel from canola or soybeans.".[15]: 1795 It goes on to define biomass in this context as "organic material excluding the material that is fossilised or embedded in geological formations".[15]: 1795 This means that coal or other fossil fuels is not a form of biomass in this context.
Conventional biofuels (first generation)
[edit]First-generation biofuels (also denoted as "conventional biofuels") are made from food crops grown on arable land.[16][17]: 447 The crop's sugar, starch, or oil content is converted into biodiesel or ethanol, using transesterification, or yeast fermentation.[18]
Advanced biofuels
[edit]To avoid a "food versus fuel" dilemma, second-generation biofuels and third-generation biofuels (also called advanced biofuels or sustainable biofuels or drop-in biofuels) are made from feedstocks which do not directly compete with food or feed crop such as waste products and energy crops.[19] A wide range of waste feedstocks such as those derived from agriculture and forestry activities like rice straw, rice husk, wood chips, and sawdust can be used to produce advanced biofuels through biochemical and thermochemical processes.[17]: 448
The feedstock used to make the fuels either grow on arable land but are byproducts of the main crop, or they are grown on marginal land. Second-generation feedstocks also include straw, bagasse, perennial grasses, jatropha, waste vegetable oil, municipal solid waste and so forth.[20]
Types
[edit]Liquid
[edit]Ethanol
[edit]Biologically produced alcohols, most commonly ethanol, and less commonly propanol and butanol, are produced by the action of microorganisms and enzymes through the fermentation of sugars or starches (easiest to produce) or cellulose (more difficult to produce).The IEA estimates that ethanol production used 20% of sugar supplies and 13% of corn supplies in 2021.[21]
Ethanol fuel is the most common biofuel worldwide, particularly in Brazil. Alcohol fuels are produced by fermentation of sugars derived from wheat, corn, sugar beets, sugar cane, molasses and any sugar or starch from which alcoholic beverages such as whiskey, can be made (such as potato and fruit waste, etc.). Production methods used are enzyme digestion (to release sugars from stored starches), fermentation of the sugars, distillation and drying. The distillation process requires significant energy input to generate heat. Heat is sometimes generated with unsustainable natural gas fossil fuel, but cellulosic biomass such as bagasse is the most common fuel in Brazil, while pellets, wood chips and also waste heat are more common in Europe. Corn-to-ethanol and other food stocks has led to the development of cellulosic ethanol.[22]
Other bioalcohols
[edit]Methanol is currently produced from natural gas, a non-renewable fossil fuel. In the future it is hoped to be produced from biomass as biomethanol. This is technically feasible, but the production is currently being postponed for concerns that the economic viability is still pending.[23] The methanol economy is an alternative to the hydrogen economy to be contrasted with today's hydrogen production from natural gas.
Butanol (C
4H
9OH) is formed by ABE fermentation (acetone, butanol, ethanol) and experimental modifications of the process show potentially high net energy gains with biobutanol as the only liquid product. Biobutanol is often claimed to provide a direct replacement for gasoline, because it will produce more energy than ethanol and allegedly can be burned "straight" in existing gasoline engines (without modification to the engine or car),[24] is less corrosive and less water-soluble than ethanol, and could be distributed via existing infrastructures. Escherichia coli strains have also been successfully engineered to produce butanol by modifying their amino acid metabolism.[25] One drawback to butanol production in E. coli remains the high cost of nutrient rich media, however, recent work has demonstrated E. coli can produce butanol with minimal nutritional supplementation.[26] Biobutanol is sometimes called biogasoline, which is incorrect as it is chemically different, being an alcohol and not a hydrocarbon like gasoline.
Biodiesel
[edit]
Biodiesel is the most common biofuel in Europe. It is produced from oils or fats using transesterification and is a liquid similar in composition to fossil/mineral diesel. Chemically, it consists mostly of fatty acid methyl (or ethyl) esters (FAMEs).[27] Feedstocks for biodiesel include animal fats, vegetable oils, soy, rapeseed, jatropha, mahua, mustard, flax, sunflower, palm oil, hemp, field pennycress, Pongamia pinnata and algae. Pure biodiesel (B100, also known as "neat" biodiesel) currently reduces emissions with up to 60% compared to diesel Second generation B100.[28] As of 2020[update], researchers at Australia's CSIRO have been studying safflower oil as an engine lubricant, and researchers at Montana State University's Advanced Fuels Center in the US have been studying the oil's performance in a large diesel engine, with results described as a "breakthrough".[29]

Biodiesel can be used in any diesel engine and modified equipment when mixed with mineral diesel. It can also be used in its pure form (B100) in diesel engines, but some maintenance and performance problems may occur during wintertime utilization, since the fuel becomes somewhat more viscous at lower temperatures, depending on the feedstock used.[30]
Electronically controlled 'common rail' and 'Unit Injector' type systems from the late 1990s onwards can only use biodiesel blended with conventional diesel fuel. These engines have finely metered and atomized multiple-stage injection systems that are very sensitive to the viscosity of the fuel. Many current-generation diesel engines are designed to run on B100 without altering the engine itself, although this depends on the fuel rail design. Since biodiesel is an effective solvent and cleans residues deposited by mineral diesel, engine filters may need to be replaced more often, as the biofuel dissolves old deposits in the fuel tank and pipes. It also effectively cleans the engine combustion chamber of carbon deposits, helping to maintain efficiency.
Biodiesel is an oxygenated fuel, meaning it contains a reduced amount of carbon and higher hydrogen and oxygen content than fossil diesel. This improves the combustion of biodiesel and reduces the particulate emissions from unburnt carbon. However, using pure biodiesel may increase NOx-emissions[31] Biodiesel is also safe to handle and transport because it is non-toxic and biodegradable, and has a high flash point of about 300 °F (148 °C) compared to petroleum diesel fuel, which has a flash point of 125 °F (52 °C).[32]
In many European countries, a 5% biodiesel blend is widely used and is available at thousands of gas stations.[33][34] In France, biodiesel is incorporated at a rate of 8% in the fuel used by all French diesel vehicles.[35] Avril Group produces under the brand Diester, a fifth of 11 million tons of biodiesel consumed annually by the European Union.[36] It is the leading European producer of biodiesel.[35]
Green diesel
[edit]Green diesel can be produced from a combination of biochemical and thermochemical processes. Conventional green diesel is produced through hydroprocessing biological oil feedstocks, such as vegetable oils and animal fats.[37][38] Recently, it is produced using series of thermochemical processes such as pyrolysis and hydroprocessing. In the thermochemical route, syngas produced from gasification, bio-oil produced from pyrolysis or biocrude produced from hydrothermal liquefaction is upgraded to green diesel using hydroprocessing.[39][40][41] Hydroprocessing is the process of using hydrogen to reform a molecular structure. For example, hydrocracking which is a widely used hydroprocessing technique in refineries is used at elevated temperatures and pressure in the presence of a catalyst to break down larger molecules, such as those found in vegetable oils, into shorter hydrocarbon chains used in diesel engines.[42] Green diesel may also be called renewable diesel, drop-in biodiesel, hydrotreated vegetable oil (HVO fuel)[42] or hydrogen-derived renewable diesel.[38] Unlike biodiesel, green diesel has exactly the same chemical properties as petroleum-based diesel.[42][43] It does not require new engines, pipelines or infrastructure to distribute and use, but has not been produced at a cost that is competitive with petroleum.[38] Gasoline versions are also being developed.[44] Green diesel is being developed in Louisiana and Singapore by ConocoPhillips, Neste Oil, Valero, Dynamic Fuels, and Honeywell UOP[38][45] as well as Preem in Gothenburg, Sweden, creating what is known as Evolution Diesel.[46]
Straight vegetable oil
[edit]
Straight unmodified edible vegetable oil is generally not used as fuel, but lower-quality oil has been used for this purpose. Used vegetable oil is increasingly being processed into biodiesel, or (more rarely) cleaned of water and particulates and then used as a fuel. The IEA estimates that biodiesel production used 17% of global vegetable oil supplies in 2021.[21]
Oils and fats reacted with 10 pounds of a short-chain alcohol (usually methanol) in the presence of a catalyst (usually sodium hydroxide [NaOH] can be hydrogenated to give a diesel substitute.[48] The resulting product is a straight-chain hydrocarbon with a high cetane number, low in aromatics and sulfur and does not contain oxygen. Hydrogenated oils can be blended with diesel in all proportions. They have several advantages over biodiesel, including good performance at low temperatures, no storage stability problems and no susceptibility to microbial attack.[49]
Biogasoline
[edit]Biogasoline can be produced biologically and themochemically. Using biologicalical methods, a study led by Professor Lee Sang-yup at the Korea Advanced Institute of Science and Technology (KAIST) and published in the international science journal Nature used modified E. coli fed with glucose found in plants or other non-food crops to produce biogasoline with the produced enzymes. The enzymes converted the sugar into fatty acids and then turned these into hydrocarbons that were chemically and structurally identical to those found in commercial gasoline fuel.[50] The thermochemical approach of producing biogasoline are similar to those used to produced biodiesel.[39][40][41] Biogasoline may also be called drop-in gasoline or renewable gasoline.
Bioethers
[edit]
Bioethers (also referred to as fuel ethers or oxygenated fuels) are cost-effective compounds that act as octane rating enhancers. "Bioethers are produced by the reaction of reactive iso-olefins, such as iso-butylene, with bioethanol."[51][attribution needed] Bioethers are created from wheat or sugar beets, and also be produced from the waste glycerol that results from the production of biodiesel.[52] They also enhance engine performance, while significantly reducing engine wear and toxic exhaust emissions. By greatly reducing the amount of ground-level ozone emissions, they contribute to improved air quality.[53][54]
In transportation fuel there are six ether additives: dimethyl ether (DME), diethyl ether (DEE), methyl tert-butyl ether (MTBE), ethyl tert-butyl ether (ETBE), tert-amyl methyl ether (TAME), and tert-amyl ethyl ether (TAEE).[55]
The European Fuel Oxygenates Association identifies MTBE and ETBE as the most commonly used ethers in fuel to replace lead. Ethers were introduced in Europe in the 1970s to replace the highly toxic compound.[56] Although Europeans still use bioether additives, the U.S. Energy Policy Act of 2005 lifted a requirement for reformulated gasoline to include an oxygenate, leading to less MTBE being added to fuel.[57] Although bioethers are likely to replace ethers produced from petroleum in the UK, it is highly unlikely they will become a fuel in and of itself due to the low energy density.[58]
Aviation biofuel
[edit]
An aviation biofuel (also known as bio-jet fuel[59] or bio-aviation fuel (BAF)[60]) is a biofuel used to power aircraft and is a sustainable aviation fuel (SAF). The International Air Transport Association (IATA) considers it a key element in reducing the environmental impact of aviation.[61] Aviation biofuel is used to decarbonize medium and long-haul air travel. These types of travel generate the most emissions, and could extend the life of older aircraft types by lowering their carbon footprint. Synthetic paraffinic kerosene (SPK) refers to any non-petroleum-based fuel designed to replace kerosene jet fuel, which is often, but not always, made from biomass.
Biofuels are biomass-derived fuels from plants, animals, or waste; depending on which type of biomass is used, they could lower CO2 emissions by 20–98% compared to conventional jet fuel.[62] The first test flight using blended biofuel was in 2008, and in 2011, blended fuels with 50% biofuels were allowed on commercial flights. In 2023 SAF production was 600 million liters, representing 0.2% of global jet fuel use.[63]
Aviation biofuel can be produced from plant or animal sources such as Jatropha, algae, tallows, waste oils, palm oil, Babassu, and Camelina (bio-SPK); from solid biomass using pyrolysis processed with a Fischer–Tropsch process (FT-SPK); with an alcohol-to-jet (ATJ) process from waste fermentation; or from synthetic biology through a solar reactor. Small piston engines can be modified to burn ethanol.
Sustainable biofuels are an alternative to electrofuels.[64] Sustainable aviation fuel is certified as being sustainable by a third-party organisation.
SAF technology faces significant challenges due to feedstock constraints. The oils and fats known as hydrotreated esters and fatty acids (Hefa), crucial for SAF production, are in limited supply as demand increases. Although advanced e-fuels technology, which combines waste CO2 with clean hydrogen, presents a promising solution, it is still under development and comes with high costs. To overcome these issues, SAF developers are exploring more readily available feedstocks such as woody biomass and agricultural and municipal waste, aiming to produce lower-carbon jet fuel more sustainably and efficiently.[65]Gaseous
[edit]Biogas and biomethane
[edit]
Biogas is a mixture composed primarily of methane and carbon dioxide produced by the process of anaerobic digestion of organic material by micro-organisms. Other trace components of this mixture includes water vapor, hydrogen sulfide, siloxanes, hydrocarbons, ammonia, oxygen, carbon monoxide, and nitrogen.[66][67] It can be produced either from biodegradable waste materials or by the use of energy crops fed into anaerobic digesters to supplement gas yields. The solid byproduct, digestate, can be used as a biofuel or a fertilizer. When CO2 and other impurities are removed from biogas, it is called biomethane. The CO2 can also be combined with hydrogen in methanation to form more methane.
Biogas can be recovered from mechanical biological treatment waste processing systems. Landfill gas, a less clean form of biogas, is produced in landfills through naturally occurring anaerobic digestion. If it escapes into the atmosphere, it acts as a greenhouse gas.
In Sweden, "waste-to-energy" power plants capture methane biogas from garbage and use it to power transport systems.[68] Farmers can produce biogas from cattle manure via anaerobic digesters.[69]
Syngas
[edit]Syngas, a mixture of carbon monoxide, hydrogen and various hydrocarbons, is produced by partial combustion of biomass (combustion with an amount of oxygen that is not sufficient to convert the biomass completely to carbon dioxide and water).[49] Before partial combustion the biomass is dried and sometimes pyrolysed. Syngas is more efficient than direct combustion of the original biofuel; more of the energy contained in the fuel is extracted.
Syngas may be burned directly in internal combustion engines, turbines or high-temperature fuel cells.[70] The wood gas generator, a wood-fueled gasification reactor, can be connected to an internal combustion engine.
Syngas can be used to produce methanol, dimethyl ether and hydrogen, or converted via the Fischer–Tropsch process to produce a diesel substitute, or a mixture of alcohols that can be blended into gasoline. Gasification normally relies on temperatures greater than 700 °C.
Lower-temperature gasification is desirable when co-producing biochar, but results in syngas polluted with tar.
Solid
[edit]The term "biofuels" is also used for solid fuels that are made from biomass, even though this is less common.[3]
Research into other types
[edit]Algae-based biofuels
[edit]Algae can be produced in ponds or tanks on land, and out at sea.[71][72] Algal fuels have high yields,[73] a high ignition point,[74] can be grown with minimal impact on fresh water resources,[75][76][77] can be produced using saline water and wastewater, and are biodegradable and relatively harmless to the environment if spilled.[78][79] However, production requires large amounts of energy and fertilizer, the produced fuel degrades faster than other biofuels, and it does not flow well in cold temperatures.[71][80]
By 2017, due to economic considerations, most efforts to produce fuel from algae have been abandoned or changed to other applications.[81]
Third and fourth-generation biofuels also include biofuels that are produced by bioengineered organisms i.e. algae and cyanobacteria.[82] Algae and cyanobacteria will use water, carbon dioxide, and solar energy to produce biofuels.[82] This method of biofuel production is still at the research level. The biofuels that are secreted by the bioengineered organisms are expected to have higher photon-to-fuel conversion efficiency, compared to older generations of biofuels.[82] One of the advantages of this class of biofuels is that the cultivation of the organisms that produce the biofuels does not require the use of arable land.[83] The disadvantages include the cost of cultivating the biofuel-producing organisms being very high.[83]
Electrofuels and solar fuels
[edit]Electrofuels[citation needed] and solar fuels may or may not be biofuels, depending on whether they contain biological elements. Electrofuels are made by storing electrical energy in the chemical bonds of liquids and gases. The primary targets are butanol, biodiesel, and hydrogen, but include other alcohols and carbon-containing gases such as methane and butane. A solar fuel is a synthetic chemical fuel produced from solar energy. Light is converted to chemical energy, typically by reducing protons to hydrogen, or carbon dioxide to organic compounds.[84]
Bio-digesters
[edit]A bio-digester is a mechanized toilet that uses decomposition and sedimentation to turn human waste into a renewable fuel called biogas. Biogas can be made from substances like agricultural waste and sewage.[85][86] The bio-digester uses a process called anaerobic digestion to produce biogas. Anaerobic digestion uses a chemical process to break down organic matter with the use of microorganisms in the absence of oxygen to produce biogas.[87] The processes involved in anaerobic respiration are hydrolysis, acidogenesis, acetogenesis, and methanogenesis.[88]
Extent of production and use
[edit]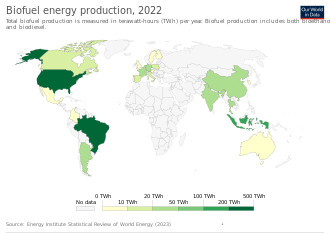
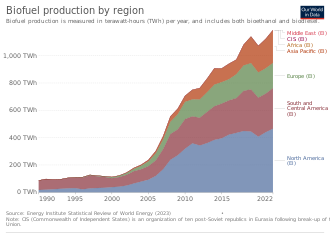
Global biofuel production was 81 Mtoe in 2017 which represented an annual increase of about 3% compared to 2010.[4]: 12 In 2017, the US was the largest biofuel producer in the world producing 37 Mtoe, followed by Brazil and South America at 23 Mtoe and Europe (mainly Germany) at 12 Mtoe.[4]: 12
An assessment from 2017 found that: "Biofuels will never be a major transport fuel as there is just not enough land in the world to grow plants to make biofuel for all vehicles. It can however, be part of an energy mix to take us into a future of renewable energy."[4]: 11
In 2021, worldwide biofuel production provided 4.3% of the world's fuels for transport, including a very small amount of aviation biofuel.[10] By 2027, worldwide biofuel production is expected to supply 5.4% of the world's fuels for transport including 1% of aviation fuel.[10]
The US, Europe, Brazil and Indonesia are driving the majority of biofuel consumption growth. This demand for biodiesel, renewable diesel and biojet fuel is projected to increase by 44% (21 billion litres) over 2022-2027.[90]
Issues
[edit]
This article needs to be updated. (March 2024) |
This article has an unclear citation style. (July 2023) |
Issues relating to biofuel are social, economic, environmental and technical problems that may arise from biofuel production and use. Social and economic issues include the "food vs fuel" debate and the need to develop responsible policies and economic instruments to ensure sustainable biofuel production. Farming for biofuels feedstock can be detrimental to the environment if not done sustainably. Environmental concerns include deforestation, biodiversity loss and soil erosion as a result of land clearing for biofuels agriculture. While biofuels can contribute to reduction in global carbon emissions, indirect land use change for biofuel production can have the inverse effect. Technical issues include possible modifications necessary to run the engine on biofuel, as well as energy balance and efficiency.
The International Resource Panel outlined the wider and interrelated factors that need to be considered when deciding on the relative merits of pursuing one biofuel over another.[91] The IRP concluded that not all biofuels perform equally in terms of their effect on climate, energy security and ecosystems, and suggested that environmental and social effects need to be assessed throughout the entire life-cycle.Environmental impacts
[edit]
Estimates about the climate impact from biofuels vary widely based on the methodology and exact situation examined.[6]
In general, biofuels emit fewer greenhouse gas emissions when burned in an engine and are generally considered carbon-neutral fuels as the carbon they emit has been captured from the atmosphere by the crops used in biofuel production.[5] They can have greenhouse gas emissions ranging from as low as -127.1 gCO2eq per MJ when carbon capture is incorporated into their production to those exceeding 95 gCO2eq per MJ when land-use change is significant. [40][41] Several factors are responsible for the variation in emission numbers of biofuel such as feedstock and its origin, fuel production technique, system boundary definitions, and energy sources.[41] However, many government policies such as those by European Union and the UK require that biofuels have at least 65% greenhouse gas emissions savings (or 70% if it is renewable fuels of non-biological origins) relative to fossil fuels.[93][94]
Life-cycle assessments of first generation biofuels have shown large emissions associated with the potential land-use change required to produce additional biofuel feedstocks.[6][7] If no land-use change is involved, first-generation biofuels can—on average—have lower emissions than fossil fuels.[6] However, biofuel production can compete with food crop production. Up to 40% of corn produced in the United States is used to make ethanol[95] and worldwide 10% of all grain is turned into biofuel.[96] A 50% reduction in grain used for biofuels in the US and Europe would replace all of Ukraine's grain exports.[97] Several studies have shown that reductions in emissions from biofuels are achieved at the expense of other impacts, such as acidification, eutrophication, water footprint and biodiversity loss.[6]
The use of second generation biofuels is thought to increase environmental sustainability, since the non-food part of plants is being used to produce second-generation biofuels, instead of being disposed.[98] But the use of second-generation biofuels increases the competition for lignocellulosic biomass, increasing the cost of these biofuels.[99]
Third generation biofuels, produced from Algae, in theory shouldn't have a negative impact on the environment than the first or second generation biofuels, due to lower changes in land use and not requiring pesticide use for production.[100] When looking at the data however, it has been shown that the environmental cost to produce the infrastructure and energy required for third generation biofuel production, are higher than the benefits provided from the biofuels use.[101]
The European Commission has officially approved a measure to phase out palm oil-based biofuels by 2030.[102][103] Unsustainable palm oil agriculture has caused significant environmental and social problems, including deforestation and pollution.
The production of biofuels can be very energy intensive, which if generated from non-renewable sources can heavily mitigate the benefits gained through biofuel use. A solution proposed to solve this issue is to supply biofuel production facilities with excess nuclear energy, which can supplement the energy provided by fossil fuels.[104] This can provide a carbon inexpensive solution to help reduce the environmental impacts of biofuel production.
Indirect land use change impacts of biofuels
[edit]This article needs to be updated. (August 2021) |


The indirect land use change impacts of biofuels, also known as ILUC or iLUC (pronounced as i-luck), relates to the unintended consequence of releasing more carbon emissions due to land-use changes around the world induced by the expansion of croplands for ethanol or biodiesel production in response to the increased global demand for biofuels.[105][106]
As farmers worldwide respond to higher crop prices in order to maintain the global food supply-and-demand balance, pristine lands are cleared to replace the food crops that were diverted elsewhere to biofuels' production. Because natural lands, such as rainforests and grasslands, store carbon in their soil and biomass as plants grow each year, clearance of wilderness for new farms translates to a net increase in greenhouse gas emissions. Due to this off-site change in the carbon stock of the soil and the biomass, indirect land use change has consequences in the greenhouse gas (GHG) balance of a biofuel.[105][106][107][108]
Other authors have also argued that indirect land use changes produce other significant social and environmental impacts, affecting biodiversity, water quality, food prices and supply, land tenure, worker migration, and community and cultural stability.[107][109][110][111]See also
[edit]- Aviation biofuel
- Bioenergy Europe
- BioEthanol for Sustainable Transport
- Biofuels Center of North Carolina
- Biogas powerplant
- International Renewable Energy Agency
- List of biofuel companies and researchers
- List of vegetable oils used for biofuel
- Renewable energy by country
- Renewable Energy Transition
- Residue-to-product ratio
- Sustainable aviation fuel
- Sustainable transport
- Table of biofuel crop yields
References
[edit]- ^ "Biofuel | Definition, Types, & Pros and Cons | Britannica". www.britannica.com. 18 March 2024. Retrieved 2 April 2024.
- ^ a b T. M. Letcher, ed. (2020). "Chapter 9: Biofuels for transport". Future energy : improved, sustainable and clean options for our planet (3rd ed.). Amsterdam, Netherlands. ISBN 978-0-08-102887-2. OCLC 1137604985.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b c "Biofuels explained - U.S. Energy Information Administration (EIA)". www.eia.gov. Retrieved 24 January 2023.
- ^ a b c d T. M. Letcher, ed. (2020). "Chapter1: Introduction With a Focus on Atmospheric Carbon Dioxide and Climate Change". Future energy : improved, sustainable and clean options for our planet (3rd ed.). Amsterdam, Netherlands. ISBN 978-0-08-102887-2. OCLC 1137604985.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b Lewandrowski J, Rosenfeld J, Pape D, Hendrickson T, Jaglo K, Moffroid K (25 March 2019). "The greenhouse gas benefits of corn ethanol – assessing recent evidence". Biofuels. 11 (3). Informa UK Limited: 361–375. doi:10.1080/17597269.2018.1546488. ISSN 1759-7269. S2CID 134824935.
- ^ a b c d e f Jeswani HK, Chilvers A, Azapagic A (November 2020). "Environmental sustainability of biofuels: a review". Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences. 476 (2243): 20200351. Bibcode:2020RSPSA.47600351J. doi:10.1098/rspa.2020.0351. PMC 7735313. PMID 33363439.
- ^ a b Lark TJ, Hendricks NP, Smith A, Pates N, Spawn-Lee SA, Bougie M, et al. (March 2022). "Environmental outcomes of the US Renewable Fuel Standard". Proceedings of the National Academy of Sciences of the United States of America. 119 (9). Bibcode:2022PNAS..11901084L. doi:10.1073/pnas.2101084119. PMC 8892349. PMID 35165202.
- ^ Jeswani HK, Chilvers A, Azapagic A (November 2020). "Environmental sustainability of biofuels: a review". Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences. 476 (2243). Bibcode:2020RSPSA.47600351J. doi:10.1098/rspa.2020.0351. ISSN 1364-5021. PMC 7735313. PMID 33363439.
- ^ "Biofuel is approaching a feedstock crunch. How bad? And what must be done?". Energy Post. 23 January 2023. Retrieved 14 March 2024.
- ^ a b c "Transport biofuels – Renewables 2022 – Analysis". IEA. Retrieved 30 January 2023.
- ^ "Biofuel is approaching a feedstock crunch. How bad? And what must be done?". Energy Post. 23 January 2023. Retrieved 30 January 2023.
- ^ "How to scale Sustainable Aviation Fuel in the next decade". World Economic Forum. Retrieved 30 January 2023.
- ^ "Renewables Report 2022". IEA. 6 December 2022.
- ^ Bayetero CM, Yépez CM, Cevallos IB, Rueda EH (January 2022). "Effect of the use of additives in biodiesel blends on the performance and opacity of a diesel engine". Materials Today: Proceedings. Advances in Mechanical Engineering Trends. 49: 93–99. doi:10.1016/j.matpr.2021.07.478. ISSN 2214-7853. S2CID 238787289.
- ^ a b IPCC, 2022: Annex I: Glossary [van Diemen, R., J.B.R. Matthews, V. Möller, J.S. Fuglestvedt, V. Masson-Delmotte, C. Méndez, A. Reisinger, S. Semenov (eds)]. In IPCC, 2022: Climate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [P.R. Shukla, J. Skea, R. Slade, A. Al Khourdajie, R. van Diemen, D. McCollum, M. Pathak, S. Some, P. Vyas, R. Fradera, M. Belkacemi, A. Hasija, G. Lisboa, S. Luz, J. Malley, (eds.)]. Cambridge University Press, Cambridge, UK and New York, NY, USA. doi: 10.1017/9781009157926.020
- ^ Cavelius P, Engelhart-Straub S, Mehlmer N, Lercher J, Awad D, Brück T (30 March 2023). "The potential of biofuels from first to fourth generation". PLOS Biology. 21 (3): e3002063. doi:10.1371/journal.pbio.3002063. ISSN 1545-7885. PMC 10063169. PMID 36996247.
- ^ a b T. M. Letcher, ed. (2020). "Chapter 21: Energy from biomass". Future energy : improved, sustainable and clean options for our planet (3rd ed.). Amsterdam, Netherlands. ISBN 978-0-08-102887-2. OCLC 1137604985.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ "What are – and who's making – 2G, 3G and 4G biofuels? : Biofuels Digest - biofuels, biodiesel, ethanol, algae, jatropha, green gasoline, green diesel, and biocrude daily news". 21 May 2010. Archived from the original on 21 May 2010.
- ^ European Parliament. "Advanced biofuels" (PDF). Retrieved 19 April 2024.
- ^ "Biofuels – Second Generation Biofuels". biofuel.org.uk. Archived from the original on 15 July 2019. Retrieved 18 January 2018.
- ^ a b "Is the biofuel industry approaching a feedstock crunch? – Analysis". IEA. 6 December 2022. Retrieved 2 January 2023.
- ^ Houghton J, Weatherwax S, Ferrell J (7 June 2006). Breaking the biological barriers to cellulosic ethanol: a joint research agenda (Report). Washington, DC (United States): EERE Publication and Product Library. doi:10.2172/1218382.
- ^ Börjesson P, Lundgren J, Ahlgren S, Nyström I (18 June 2013). Dagens och framtidens hållbara biodrivmedel: underlagsrapport från f3 till utredningen om fossilfri fordonstrafik [Today's and the future's sustainable biofuels: background report from f3 to the inquiry into fossil-free vehicle traffic.] (Report) (in Swedish). Vol. 13. The Swedish Knowledge Centre for Renewable Transportation Fuels. p. 170.
- ^ "ButylFuel, LLC Main Page". Butanol.com. 15 August 2005. Archived from the original on 10 July 2019. Retrieved 14 July 2010.
- ^ Evans J (14 January 2008). "Biofuels aim higher". Biofuels, Bioproducts and Biorefining (BioFPR). Archived from the original on 10 August 2009. Retrieved 3 December 2008.
- ^ Pontrelli S, Fricke RC, Sakurai SS, Putri SP, Fitz-Gibbon S, Chung M, et al. (September 2018). "Directed strain evolution restructures metabolism for 1-butanol production in minimal media". Metabolic Engineering. 49: 153–163. doi:10.1016/j.ymben.2018.08.004. PMID 30107263.
- ^ Fukuda H, Kondo A, Noda H (January 2001). "Biodiesel fuel production by transesterification of oils". Journal of Bioscience and Bioengineering. 92 (5): 405–416. doi:10.1016/s1389-1723(01)80288-7. PMID 16233120.
- ^ "Perstop Press release: Verdis Polaris Aura – second generation B100 – The advanced green one". Archived from the original on 4 August 2014. Retrieved 21 June 2014.
- ^ Lee T (7 June 2020). "Safflower oil hailed by scientists as possible recyclable, biodegradable replacement for petroleum". ABC News. Landline. Australian Broadcasting Corporation. Archived from the original on 7 June 2020. Retrieved 7 June 2020.
- ^ "Alternative Fuels Data Center: Biodiesel Blends". afdc.energy.gov. Retrieved 31 March 2022.
- ^ Nylund NO, Koponen K (2012). Fuel and Technology Alternatives for Buses. Overall Energy Efficiency and Emission Performance. IEA Bioenergy Task 46 (PDF) (Report). VTT Technical Research Centre of Finland. Archived from the original (PDF) on 16 February 2020.. Possibly the new emission standards Euro VI/EPA 10 will lead to reduced NOx-levels also when using B100.
- ^ "Biofuels Facts". Hempcar.org. Archived from the original on 20 May 2011. Retrieved 14 July 2010.
- ^ "ADM Biodiesel: Hamburg, Leer, Mainz". Biodiesel.de. Archived from the original on 2 August 2009. Retrieved 14 July 2010.
- ^ RRI Limited for Biodiesel Filling Stations. "Welcome to Biodiesel Filling Stations". Biodieselfillingstations.co.uk. Archived from the original on 14 July 2018. Retrieved 14 July 2010.
- ^ a b Avril Group : Activity Report 2014, p. 58
- ^ EurObserv 2014, p. 4
- ^ Brown R, Holmgren J. "Fast Pyrolysis and Bio-Oil Upgrading" (PDF). Archived (PDF) from the original on 5 January 2012. Retrieved 15 March 2012.
- ^ a b c d "Alternative & Advanced Fuels". US Department of Energy. Archived from the original on 27 October 2012. Retrieved 7 March 2012.
- ^ a b "Technology | Comsyn". www.comsynproject.eu. Retrieved 19 April 2024.
- ^ a b c Lilonfe S, Dimitriou I, Davies B, Abdul-Manan AF, McKechnie J (1 January 2024). "Comparative techno-economic and life cycle analyses of synthetic "drop-in" fuel production from UK wet biomass". Chemical Engineering Journal. 479: 147516. Bibcode:2024ChEnJ.47947516L. doi:10.1016/j.cej.2023.147516. ISSN 1385-8947.
- ^ a b c d Lilonfe S, Davies B, Abdul-Manan AF, Dimitriou I, McKechnie J (17 April 2024). "A review of techno-economic analyses and life cycle greenhouse gas emissions of biomass-to-hydrocarbon "drop-in" fuels". Sustainable Production and Consumption. 47: 425–444. Bibcode:2024SusPC..47..425L. doi:10.1016/j.spc.2024.04.016. ISSN 2352-5509.
- ^ a b c Knothe G (June 2010). "Biodiesel and renewable diesel: a comparison". Progress in Energy and Combustion Science. 36 (3): 364–373. Bibcode:2010PECS...36..364K. doi:10.1016/j.pecs.2009.11.004. Archived from the original on 6 November 2012. Retrieved 23 August 2012.
- ^ "Green Diesel v. Biodiesel". Archived from the original on 5 August 2018. Retrieved 5 August 2018.
- ^ Jessica E. "Breakthroughs in Green Gasoline Production". Biomass Magazine. Archived from the original on 11 March 2012. Retrieved 14 August 2012.
- ^ Albrecht KO, Hallen RT (March 2011). A Brief Literature Overview of Various Routes to Biorenewable Fuels from Lipids for the National Alliance of Advanced Biofuels and Bio-products NAAB Consortium (PDF) (Report). Prepared by the US Department of Energy. Archived (PDF) from the original on 12 July 2012. Retrieved 23 August 2012.
- ^ "Preem makes major investment in green diesel at the Port of Gothenburg – Port of Gothenburg". August 2014. Archived from the original on 1 August 2014.
- ^ "Wal-Mart To Test Hybrid Trucks". Sustainable Business. 3 February 2009. Archived from the original on 8 May 2014. Retrieved 8 May 2014.
- ^ "Alternative Fuels Data Center: Biodiesel Production and Distribution". afdc.energy.gov. Retrieved 31 March 2022.
- ^ a b Evans G (14 April 2008). Liquid Transport Biofuels – Technology Status Report (Report). National Non-Food Crops Centre. Archived from the original on 11 June 2008.
- ^ Liquid Transport Fuels&Lubes - South Korean scientists use E. coli to make gasoline (Report). Fuels&Lubes Daily. 4 November 2013. Archived from the original on 7 September 2022.
- ^ Rock K, Korpelshoek M (2007). "Bioethers Impact on the Gasoline Pool". Digital Refining. Archived from the original on 14 November 2016. Retrieved 15 February 2014.
- ^ "Biofuels - Types of Biofuels - Bioethers". biofuel.org.uk. Archived from the original on 1 February 2016.
- ^ "Council Directive 85/536/EEC of 5 December 1985 on crude-oil savings through the use of substitute fuel components in petrol". Eur-lex.europa.eu. Archived from the original on 21 May 2011. Retrieved 14 July 2010.
- ^ "Impact Assessment of the Proposal for a Directive of the European Parliament and of the Council modifying Directive 98/70/EC relating to the quality of petrol and diesel fuels" (PDF). Brussels: Commission of the European Communities. 31 January 2007. Archived (PDF) from the original on 15 July 2011. Retrieved 14 July 2010.
- ^ Sukla MK, Bhaskar T, Jain AK, Singal SK, Garg MO. "Bio-Ethers as Transportation Fuel: A Review" (PDF). Indian Institute of Petroleum Dehradun. Archived (PDF) from the original on 14 October 2011. Retrieved 15 February 2014.
- ^ "What are Bio-Ethers?" (PDF). . The European Fuel Oxygenates Association. Archived from the original (PDF) on 6 March 2014.
- ^ "Gasoline". Environmental Protection Agency. Archived from the original on 6 December 2013. Retrieved 6 March 2014.
- ^ "Biofuels – Types of Biofuels – Bioethers". Archived from the original on 1 February 2016. Retrieved 30 May 2015.
- ^ "Sustainable aviation fuel market demand drives new product launches". Investable Universe. 4 December 2020. Retrieved 12 December 2022. Note: Investable Universe>About
- ^ Doliente SS, et al. (10 July 2020). "Bio-aviation Fuel: A Comprehensive Review and Analysis of the Supply Chain Components" (PDF). Frontiers in Energy Research. 8. doi:10.3389/fenrg.2020.00110.
- ^ "Developing Sustainable Aviation Fuel (SAF)". IATA.
- ^ Bauen A, Howes J, Bertuccioli L, Chudziak C (August 2009). "Review of the potential for biofuels in aviation". CiteSeerX 10.1.1.170.8750.
- ^ IATA (December 2023). "Net zero 2050: sustainable aviation fuels – December 2023". www.iata.org/flynetzero. Archived from the original on 24 February 2024.
- ^ Mark Pilling (25 March 2021). "How sustainable fuel will help power aviation's green revolution". Flight Global.
- ^ "New Technology Helps Advance Non-Hefa SAF Projects". Energy Intelligence. 10 May 2024. Retrieved 14 May 2024.
- ^ Ryckebosch E, Drouillon M, Vervaeren H (1 May 2011). "Techniques for transformation of biogas to biomethane". Biomass and Bioenergy. 35 (5): 1633–1645. Bibcode:2011BmBe...35.1633R. doi:10.1016/j.biombioe.2011.02.033. ISSN 0961-9534.
- ^ "A Detailed Economic Assessment of Anaerobic Digestion Technology and its Suitability to UK Farming and Waste Systems (Andersons)". National Non-Food Crops Centre. 4 October 2008. NNFCC 08-006. Archived from the original on 4 October 2008. Retrieved 2 January 2023.
- ^ Yee A (21 September 2018). "In Sweden, Trash Heats Homes, Powers Buses and Fuels Taxi Fleets". The New York Times. ISSN 0362-4331. Retrieved 14 March 2024.
- ^ "BIOGAS: No bull, manure can power your farm." Farmers Guardian (25 September 2009): 12. General OneFile. Gale.
- ^ Nagel F (2008). Electricity from wood through the combination of gasification and solid oxide fuel cells (PhD thesis). Swiss Federal Institute of Technology Zurich. Archived from the original on 13 March 2011.
- ^ a b "Biofuel from Algae: The Pros and Cons of Pond Scum". Thomasnet®. Archived from the original on 6 April 2020. Retrieved 25 October 2020.
- ^ "Biomass - Offshore wind farms = seaweed = biofuel". Renewable Energy Magazine, at the heart of clean energy journalism. 14 September 2020. Archived from the original on 27 July 2020. Retrieved 16 October 2020.
- ^ Greenwell HC, Laurens LM, Shields RJ, Lovitt RW, Flynn KJ (May 2010). "Placing microalgae on the biofuels priority list: a review of the technological challenges". Journal of the Royal Society, Interface. 7 (46): 703–726. doi:10.1098/rsif.2009.0322. PMC 2874236. PMID 20031983.
- ^ Dinh LT, Guo Y, Mannan MS (2009). "Sustainability evaluation of biodiesel production using multicriteria decision-making". Environmental Progress & Sustainable Energy. 28 (1): 38–46. Bibcode:2009EPSE...28...38D. doi:10.1002/ep.10335. S2CID 111115884.
- ^ Ajayebi A, Gnansounou E, Kenthorai Raman J (1 December 2013). "Comparative life cycle assessment of biodiesel from algae and jatropha: A case study of India". Bioresource Technology. 150: 429–437. Bibcode:2013BiTec.150..429A. doi:10.1016/j.biortech.2013.09.118. ISSN 0960-8524. PMID 24140355.
- ^ Yang J, Xu M, Zhang X, Hu Q, Sommerfeld M, Chen Y (January 2011). "Life-cycle analysis on biodiesel production from microalgae: water footprint and nutrients balance" (PDF). Bioresource Technology. 102 (1): 159–165. Bibcode:2011BiTec.102..159Y. doi:10.1016/j.biortech.2010.07.017. PMID 20675125. Archived from the original (PDF) on 27 February 2012.
- ^ Cornell CB (29 March 2008). "First Algae Biodiesel Plant Goes Online: 1 April 2008". Gas 2.0. Archived from the original on 18 June 2019. Retrieved 10 June 2008.
- ^ Demirbas AH (2011). "Biodiesel from oilgae, biofixation of carbon dioxide by microalgae: A solution to pollution problems". Applied Energy. 88 (10): 3541–3547. Bibcode:2011ApEn...88.3541D. doi:10.1016/j.apenergy.2010.12.050. hdl:11503/1330.
- ^ Demirbas AH (2009). "Inexpensive oil and fats feedstocks for production of biodiesel". Energy Education Science and Technology Part A: Energy Science and Research. 23: 1–13.
- ^ Rodionova M, Poudyal R, Tiwari I, Voloshin R, Zharmukhamedov S, Nam H, et al. (March 2017). "Biofuel production: Challenges and opportunities". International Journal of Hydrogen Energy. 42 (12): 8450–8461. Bibcode:2017IJHE...42.8450R. doi:10.1016/j.ijhydene.2016.11.125.
- ^ Wesoff E (19 April 2017). "Hard Lessons From the Great Algae Biofuel Bubble". Archived from the original on 5 July 2017. Retrieved 5 August 2017.
- ^ a b c Aro EM (January 2016). "From first generation biofuels to advanced solar biofuels". Ambio. 45 (Supplement 1): S24–S31. Bibcode:2016Ambio..45S..24A. doi:10.1007/s13280-015-0730-0. PMC 4678123. PMID 26667057.
- ^ a b Abdullah B, Muhammad SA, Shokravi Z, Ismail S, Kassim KA, Mahmood AN, et al. (June 2019). "Fourth generation biofuel: A review on risks and mitigation strategies". Renewable and Sustainable Energy Reviews. 107: 37–50. Bibcode:2019RSERv.107...37A. doi:10.1016/j.rser.2019.02.018. S2CID 116245776.
- ^ Lü J, Sheahan C, Fu P (2011). "Metabolic engineering of algae for fourth generation biofuels production". Energy & Environmental Science. 4 (7): 2451. doi:10.1039/c0ee00593b. ISSN 1754-5692.
- ^ Xu F, Li Y, Ge X, Yang L, Li Y (1 January 2018). "Anaerobic digestion of food waste – Challenges and opportunities". Bioresource Technology. 247: 1047–1058. Bibcode:2018BiTec.247.1047X. doi:10.1016/j.biortech.2017.09.020. ISSN 0960-8524. PMID 28965912.
- ^ Mahmudul HM, Rasul MG, Akbar D, Narayanan R, Mofijur M (20 January 2021). "A comprehensive review of the recent development and challenges of a solar-assisted biodigester system". Science of the Total Environment. 753: 141920. Bibcode:2021ScTEn.75341920M. doi:10.1016/j.scitotenv.2020.141920. ISSN 0048-9697. PMID 32889316.
- ^ Kougias PG, Angelidaki I (30 April 2018). "Biogas and its opportunities—A review". Frontiers of Environmental Science & Engineering. 12 (3): 14. doi:10.1007/s11783-018-1037-8. ISSN 2095-221X.
- ^ Zhang C, Su H, Baeyens J, Tan T (1 October 2014). "Reviewing the anaerobic digestion of food waste for biogas production". Renewable and Sustainable Energy Reviews. 38: 383–392. Bibcode:2014RSERv..38..383Z. doi:10.1016/j.rser.2014.05.038. ISSN 1364-0321.
- ^ "Biofuel energy production". Our World in Data. Retrieved 15 August 2023.
- ^ "Is the biofuel industry approaching a feedstock crunch? – Analysis". IEA. 6 December 2022. Retrieved 13 March 2024.
- ^ Towards sustainable production and use of resources: Assessing Biofuels Archived 2016-05-13 at the Portuguese Web Archive, 2009, International Resource Panel, United Nations Environment Programme
- ^ "Indonesia's biodiesel drive is leading to deforestation". BBC News. 8 December 2021.
- ^ "Press corner". European Commission - European Commission. Retrieved 19 April 2024.
- ^ "Biomass Strategy 2023". GOV.UK. Retrieved 19 April 2024.
- ^ "Food vs fuel: Ukraine war sharpens debate on use of crops for energy". Financial Times. 12 June 2022. Archived from the original on 10 December 2022.
- ^ "Guest view: Global hunger fight means no biofuel". Reuters. 6 June 2022.
- ^ "Cutting biofuels can help avoid global food shock from Ukraine war". New Scientist. 14 March 2022.
- ^ Antizar-Ladislao B, Turrion-Gomez JL (September 2008). "Second-generation biofuels and local bioenergy systems". Biofuels, Bioproducts and Biorefining. 2 (5): 455–469. doi:10.1002/bbb.97. S2CID 84426763.
- ^ Bryngemark E (December 2019). "Second generation biofuels and the competition for forest raw materials: A partial equilibrium analysis of Sweden". Forest Policy and Economics. 109: 102022. Bibcode:2019ForPE.10902022B. doi:10.1016/j.forpol.2019.102022. ISSN 1389-9341. S2CID 212954432.
- ^ Jacob-Lopes E, Zepka LQ, Severo IA, Maroneze MM, eds. (2022). 3rd generation biofuels: disruptive technologies to enable commercial production. Woodhead Publishing series in energy. Cambridge, MA Kidlington: Woodhead Publishing, an imprint of Elsevier. ISBN 978-0-323-90971-6.
- ^ Magazine H. "Biofuel Made from Algae Isn't the Holy Grail We Expected". Hakai Magazine. Retrieved 31 March 2024.
- ^ "Palm Oil Exporter Indonesia Concerned by EU's Deforestation Law". Jakarta Globe. 22 May 2022.
- ^ "EU palm oil use and imports seen plummeting by 2032". Reuters. 8 December 2022.
- ^ Forsberg C (January 2009). "The Real Path to Green Energy: Hybrid Nuclear-Renewable Power". Bulletin of the Atomic Scientists. 65 (6): 65–71. Bibcode:2009BuAtS..65f..65F. doi:10.2968/065006007. ISSN 0096-3402.
- ^ a b Timothy Searchinger, et al. (29 February 2008). "Use of U.S. Croplands for Biofuels Increases Greenhouse Gases Through Emissions from Land-Use Change". Science. 319 (5867): 1238–1240. Bibcode:2008Sci...319.1238S. doi:10.1126/science.1151861. PMID 18258860. S2CID 52810681. Originally published online in Science Express on 7 February 2008 available here Archived 2009-12-11 at the Wayback Machine
- ^ a b Michael Wang, Zia Haq (14 March 2008). "Letter to Science about Searchinger et al. article" (PDF). Argonne National Laboratory. Archived from the original (PDF) on 15 February 2013. Retrieved 7 June 2009. The published version on Science Letters is included in Searchinger E-Letter responses 2008-08-12
- ^ a b Gnansounou, et al. (March 2008). "Accounting for indirect land-use changes in GHG balances of biofuels: Review of current approaches" (PDF). École Polytechnique Fédérale de Lausanne. Retrieved 7 June 2009. Working Paper REF. 437.101
- ^ Alexander E. Farrell (13 February 2008). "Better biofuels before more biofuels". San Francisco Chronicle. Retrieved 7 June 2009.
- ^ Donald Sawyer (27 May 2008). "Climate change, biofuels and eco-social impacts in the Brazilian Amazon and Cerrado". Philosophical Transactions of the Royal Society. 363 (1498): 1747–1752. doi:10.1098/rstb.2007.0030. PMC 2373893. PMID 18267903. Published on line 2008-02-11.
- ^ Naylor, et al. (November 2007). "The Ripple Effect: Biofuels, Food Security, and the Environment". Environment. Retrieved 7 June 2009.
- ^ Renton Righelato, Dominick V. Spracklen (17 August 2007). "Carbon Mitigation by Biofuels or by Saving and Restoring Forests?". Science. 317 (5840): 902. doi:10.1126/science.1141361. PMID 17702929. S2CID 40785300.
Sources
[edit]- Avril Group, ed. (2015). A new springtime for the oils and proteins sectors : Activity Report 2014 (PDF) (Report). Paris: Avril. p. 65. Archived from the original (PDF) on 26 October 2020. Retrieved 11 August 2022.
- EurObserv (July 2014). Biofuel barometer (PDF) (Report).
External links
[edit]- Biofuels Journal
- Alternative Fueling Station Locator Archived 14 July 2008 at the Wayback Machine (EERE)
- Towards Sustainable Production and Use of Resources: Assessing Biofuels by the United Nations Environment Programme, October 2009.
- Biofuels guidance for businesses, including permits and licences required on NetRegs.gov.uk
- How Much Water Does It Take to Make Electricity?—Natural gas requires the least water to produce energy, some biofuels the most, according to a new study.
- International Conference on Biofuels Standards – European Union Biofuels Standardization
- Biofuels from Biomass: Technology and Policy Considerations Thorough overview from MIT
- The Guardian news on biofuels
- The US DOE Clean Cities Program – links to the 87 US Clean Cities coalitions, as of 2004.
- Biofuels Factsheet by the University of Michigan's Center for Sustainable Systems
- Learn Biofuels – Educational Resource for Students

