Tabernanthine
Tools
Actions
General
Print/export
Print/export
In other projects
From Wikipedia, the free encyclopedia
This is an old revision of this page, as edited by DMacks (talk | contribs) at 21:10, 19 June 2020 (Remove malformatted |molecular_weight= when infobox can autocalculate it, per Wikipedia talk:WikiProject Pharmacology#Molecular weights in drugboxes (via WP:JWB)). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 21:10, 19 June 2020 by DMacks (talk | contribs) (Remove malformatted |molecular_weight= when infobox can autocalculate it, per Wikipedia talk:WikiProject Pharmacology#Molecular weights in drugboxes (via WP:JWB))
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
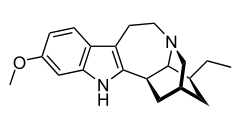
| Formula | C20H26N2O |
| Molar mass | 310.441 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Tabernanthine is an alkaloid found in Tabernanthe iboga.[1]
It has been used in laboratory experiments to study how addiction affects the brain.[2]
Tabernanthine persistently reduced the self-administration of cocaine and morphine in rats.[3]
See also
References
- ^ Bartlett, M. F.; Dickel, D. F.; Taylor, W. I. (1958). "The Alkaloids of Tabernanthe iboga. Part IV.1 The Structures of Ibogamine, Ibogaine, Tabernanthine and Voacangine". Journal of the American Chemical Society. 80: 126–136. doi:10.1021/ja01534a036.
- ^ Levi MS, Borne RF (October 2002). "A review of chemical agents in the pharmacotherapy of addiction". Curr. Med. Chem. 9 (20): 1807–18. doi:10.2174/0929867023368980. PMID 12369879.
- ^ Glick SD, Kuehne ME, Raucci J, Wilson TE, Larson D, Keller RW Jr, Carlson JN (September 1994). "Effects of iboga alkaloids on morphine and cocaine self-administration in rats: relationship to tremorigenic effects and to effects on dopamine release in nucleus accumbens and striatum". Brain Res. 657 (1–2): 14–22. doi:10.1016/0006-8993(94)90948-2. PMID 7820611.
Treatment of drug dependence (N07B) | |
|---|---|
| Nicotine dependence |
|
| Alcohol dependence | |
| Opioid dependence | |
| Benzodiazepine dependence |
|
This drug article relating to the nervous system is a stub. You can help Wikipedia by expanding it. |
