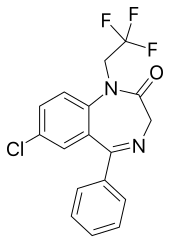
Halazepam
 | |
 | |
| Clinical data | |
|---|---|
| Other names | 9-chloro-6-phenyl-2-(2,2,2-trifluoroethyl)-2,5-diazabicyclo[5.4.0]undeca-5,8,10,12-tetraen-3-one |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a684001 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 14 hours (halazepam), 50–100 hours (metabolites). |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.041.281 |
| Chemical and physical data | |
| Formula | C17H12ClF3N2O |
| Molar mass | 352.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Halazepam is a benzodiazepine derivative that was marketed under the brand names Paxipam in the United States,[2] Alapryl in Spain,[3] and Pacinone in Portugal.[4]
Medical uses[edit]
Halazepam was used for the treatment of anxiety.[2]
Adverse effects[edit]
Adverse effects include drowsiness, confusion, dizziness, and sedation. Gastrointestinal side effects have also been reported including dry mouth and nausea.[2]
Pharmacokinetics and pharmacodynamics[edit]
Pharmacokinetics and pharmacodynamics were listed in Current Psychotherapeutic Drugs published on June 15, 1998 as follows:[5]
| Onset of action | Intermediate to slow |
| Plasma half life | 14 hr for parent drug and 30-100 hr for its metabolite |
| Peak plasma levels | 1-3 hr for parent drug and 3-6 hf for its metabolite |
| Metabolism | Metabolized into desmethyldiazepam and 3-hydroxyhalazepam (in the liver) |
| Excretion | Excreted through kidneys |
| Protein binding | 98% bound to plasma protein |
Regulatory Information[edit]
Halazepam is classified as a schedule 4 controlled substance with a corresponding code 2762 by the Drug Enforcement Administration (DEA).[6]
Commercial production[edit]
Halazepam was invented by Schlesinger Walter in the U.S. It was marketed as an anti-anxiety agent in 1981. However, Halazepam is not commercially available in the United States because it was withdrawn by its manufacturer for poor sales.[2]
See also[edit]
- Benzodiazepines
- Nordazepam
- Diazepam
- Chlordiazepoxide
- Quazepam, fletazepam, triflubazam — benzodiazepines with trifluoromethyl group attached
References[edit]
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d "halazepam". Drugs.com. Retrieved December 11, 2014.
- ^ "Alapryl". Drugs.com. Retrieved December 11, 2014.
- ^ "Pacinone". Drugs.com. Retrieved December 11, 2014.
- ^ Sellers EM (1998). "Antianxiety agents: benzodiazepine derivatives". In Quitkin FM, et al. (eds.). Current Psychotherapeutic Drugs (2nd ed.). Washington: American Psychiatric Press. p. 166. ISBN 978-0-88048-994-2.
- ^ "SCHEDULES OF CONTROLLED SUBSTANCES". Code of Federal Regulations. 2012-04-01. pp. § 1308.14 Schedule IV. Retrieved December 12, 2014.
External links[edit]
