GABAB receptor
| gamma-aminobutyric acid (GABA) B receptor, 1 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | GABBR1 | ||||||
| NCBI gene | 2550 | ||||||
| HGNC | 4070 | ||||||
| OMIM | 603540 | ||||||
| RefSeq | NM_021905 | ||||||
| UniProt | Q9UBS5 | ||||||
| Other data | |||||||
| Locus | Chr. 6 p21.3 | ||||||
| |||||||
| gamma-aminobutyric acid (GABA) B receptor, 2 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | GABBR2 | ||||||
| Alt. symbols | GPR51 | ||||||
| NCBI gene | 9568 | ||||||
| HGNC | 4507 | ||||||
| OMIM | 607340 | ||||||
| RefSeq | NM_005458 | ||||||
| UniProt | O75899 | ||||||
| Other data | |||||||
| Locus | Chr. 9 q22.1-22.3 | ||||||
| |||||||
GABAB receptors (GABABR) are G-protein coupled receptors for gamma-aminobutyric acid (GABA), therefore making them metabotropic receptors, that are linked via G-proteins to potassium channels.[1] The changing potassium concentrations hyperpolarize the cell at the end of an action potential. The reversal potential of the GABAB-mediated IPSP (inhibitory postsynaptic potential) is −100 mV, which is much more hyperpolarized than the GABAA IPSP. GABAB receptors are found in the central nervous system and the autonomic division of the peripheral nervous system.[2]
The receptors were first named in 1981 when their distribution in the CNS was determined, which was determined by Norman Bowery and his team using radioactively labelled baclofen.[3]
Functions
[edit]GABABRs stimulate the opening of K+ channels, specifically GIRKs, which brings the neuron closer to the equilibrium potential of K+. This reduces the frequency of action potentials which reduces neurotransmitter release.[citation needed] Thus GABAB receptors are inhibitory receptors.
GABAB receptors also reduces the activity of adenylyl cyclase and Ca2+ channels by using G-proteins with Gi/G0 α subunits.[4]
GABAB receptors are involved in behavioral actions of ethanol,[5][6] gamma-hydroxybutyric acid (GHB),[7] and possibly in pain.[8] Recent research suggests that these receptors may play an important developmental role.[9]
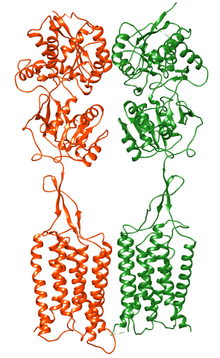
Structure
[edit]GABAB Receptors are similar in structure to and in the same receptor family with metabotropic glutamate receptors.[10] There are two subunits of the receptor, GABAB1 and GABAB2,[11] and these appear to assemble as obligate heterodimers in neuronal membranes by linking up by their intracellular C termini.[10] In the mammalian brain, two predominant, differentially expressed isoforms of the GABAB1 are transcribed from the Gabbr1 gene, GABAB(1a) and GABAB(1b), which are conserved in different species including humans.[12] This might potentially offer more complexity in terms of the function due to different composition of the receptor.[12] Cryo-electron microscopy structures of the full length GABAB receptor in different conformational states from inactive apo to fully active have been obtained. Unlike Class A and B GPCRs, phospholipids bind within the transmembrane bundles and allosteric modulators bind at the interface of GABAB1 and GABAB2 subunits.[13][14][15][16][17][18][19]
Ligands
[edit]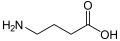

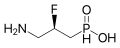
Agonists
[edit]- GABA
- Baclofen is a GABA analogue which acts as a selective agonist of GABAB receptors, and is used as a muscle relaxant. However, it can aggravate absence seizures, and so is not used in epilepsy.
- gamma-Hydroxybutyrate (GHB)
- Phenibut
- 4-Fluorophenibut
- Isovaline
- 3-Aminopropylphosphinic acid
- Lesogaberan
- SKF-97541: 3-Aminopropyl(methyl)phosphinic acid, 10× more potent than baclofen as GABAB agonist, but also GABAA-ρ antagonist
- Taurine
- CGP-44532

Positive Allosteric Modulators
[edit]

Antagonists
[edit]- Homotaurine[24]
- Ginsenosides[25]
- 2-OH-saclofen
- Saclofen
- Phaclofen
- SCH-50911
- 2-Phenethylamine
- CGP-35348
- CGP-52432: 3-([(3,4-Dichlorophenyl)methyl]amino]propyl) diethoxymethyl)phosphinic acid, CAS# 139667-74-6
- CGP-55845: (2S)-3-([(1S)-1-(3,4-Dichlorophenyl)ethyl]amino-2-hydroxypropyl)(phenylmethyl)phosphinic acid, CAS# 149184-22-5
- SGS-742[26][27]
See also
[edit]References
[edit]- ^ Chen K, Li HZ, Ye N, Zhang J, Wang JJ (October 2005). "Role of GABAB receptors in GABA and baclofen-induced inhibition of adult rat cerebellar interpositus nucleus neurons in vitro". Brain Research Bulletin. 67 (4): 310–8. doi:10.1016/j.brainresbull.2005.07.004. PMID 16182939. S2CID 6433030.
- ^ Hyland NP, Cryan JF (2010). "A Gut Feeling about GABA: Focus on GABA(B) Receptors". Frontiers in Pharmacology. 1: 124. doi:10.3389/fphar.2010.00124. PMC 3153004. PMID 21833169.
- ^ Hill DR, Bowery NG (March 1981). "3H-baclofen and 3H-GABA bind to bicuculline-insensitive GABA B sites in rat brain". Nature. 290 (5802): 149–52. Bibcode:1981Natur.290..149H. doi:10.1038/290149a0. PMID 6259535. S2CID 4335907.
- ^ Rang HP, Dale MM, Ritter JM, Flower RJ, Henderson G (2016). Rang and Dale's Pharmacology (8th ed.). Elsevier, Churchill Livingstone. p. 462. ISBN 978-0-7020-5362-7. OCLC 903234097.
- ^ Dzitoyeva S, Dimitrijevic N, Manev H (April 2003). "Gamma-aminobutyric acid B receptor 1 mediates behavior-impairing actions of alcohol in Drosophila: adult RNA interference and pharmacological evidence". Proceedings of the National Academy of Sciences of the United States of America. 100 (9): 5485–90. Bibcode:2003PNAS..100.5485D. doi:10.1073/pnas.0830111100. PMC 154371. PMID 12692303.
- ^ Ariwodola OJ, Weiner JL (November 2004). "Ethanol potentiation of GABAergic synaptic transmission may be self-limiting: role of presynaptic GABA(B) receptors". The Journal of Neuroscience. 24 (47): 10679–86. doi:10.1523/JNEUROSCI.1768-04.2004. PMC 6730127. PMID 15564584.
- ^ Dimitrijevic N, Dzitoyeva S, Satta R, Imbesi M, Yildiz S, Manev H (September 2005). "Drosophila GABA(B) receptors are involved in behavioral effects of gamma-hydroxybutyric acid (GHB)". European Journal of Pharmacology. 519 (3): 246–52. doi:10.1016/j.ejphar.2005.07.016. PMID 16129424.
- ^ Manev H, Dimitrijevic N (May 2004). "Drosophila model for in vivo pharmacological analgesia research". European Journal of Pharmacology. 491 (2–3): 207–8. doi:10.1016/j.ejphar.2004.03.030. PMID 15140638.
- ^ Dzitoyeva S, Gutnov A, Imbesi M, Dimitrijevic N, Manev H (August 2005). "Developmental role of GABAB(1) receptors in Drosophila". Brain Research. Developmental Brain Research. 158 (1–2): 111–4. doi:10.1016/j.devbrainres.2005.06.005. PMID 16054235.
- ^ a b MRC (Medical Research Council). 2003. Glutamate receptors: Structures and functions. University of Brisotol Centre for Synaptic Plasticity.
- ^ Purves D, Augustine GJ, Fitzpatrick D, Katz LC, LaMantia AS, McNamara JO, Williams SM (2001). "7. Neurotransmitter Receptors and Their Effects". Neuroscience (Second ed.). Sinauer Associates, Inc.
- ^ a b Kaupmann K, Huggel K, Heid J, Flor PJ, Bischoff S, Mickel SJ, et al. (March 1997). "Expression cloning of GABA(B) receptors uncovers similarity to metabotropic glutamate receptors". Nature. 386 (6622): 239–46. Bibcode:1997Natur.386..239K. doi:10.1038/386239a0. PMID 9069281. S2CID 4345443.
- ^ Shaye H, Stauch B, Gati C, Cherezov V (May 2021). "Molecular mechanisms of metabotropic GABAB receptor function". Science Advances. 7 (22): eabg3362. Bibcode:2021SciA....7.3362S. doi:10.1126/sciadv.abg3362. PMC 8163086. PMID 34049877.
- ^ Shaye H, Ishchenko A, Lam JH, Han GW, Xue L, Rondard P, et al. (August 2020). "Structural basis of the activation of a metabotropic GABA receptor". Nature. 584 (7820): 298–303. Bibcode:2020Natur.584..298S. doi:10.1038/s41586-020-2408-4. PMC 8020835. PMID 32555460.
- ^ Papasergi-Scott MM, Robertson MJ, Seven AB, Panova O, Mathiesen JM, Skiniotis G (June 2020). "Structures of metabotropic GABAB receptor". Nature. 584 (7820): 310–314. Bibcode:2020Natur.584..310P. doi:10.1038/s41586-020-2469-4. PMC 7429364. PMID 32580208.
- ^ Mao C, Shen C, Li C, Shen DD, Xu C, Zhang S, et al. (June 2020). "B receptor". Cell Research. 30 (7): 564–573. doi:10.1038/s41422-020-0350-5. PMC 7343782. PMID 32494023. S2CID 219183617.
- ^ Park J, Fu Z, Frangaj A, Liu J, Mosyak L, Shen T, et al. (June 2020). "B receptor in an inactive state". Nature. 584 (7820): 304–309. doi:10.1038/s41586-020-2452-0. PMC 7725281. PMID 32581365. S2CID 220050861.
- ^ Kim Y, Jeong E, Jeong JH, Kim Y, Cho Y (November 2020). "Structural Basis for Activation of the Heterodimeric GABAB Receptor". Journal of Molecular Biology. 432 (22): 5966–5984. doi:10.1016/j.jmb.2020.09.023. PMID 33058878. S2CID 222841520.
- ^ Shen C, Mao C, Xu C, Jin N, Zhang H, Shen DD, et al. (June 2021). "Structural basis of GABAB receptor-Gi protein coupling". Nature. 594 (7864): 594–598. Bibcode:2021Natur.594..594S. doi:10.1038/s41586-021-03507-1. PMC 8222003. PMID 33911284.
- ^ Urwyler S, Mosbacher J, Lingenhoehl K, Heid J, Hofstetter K, Froestl W, et al. (November 2001). "Positive allosteric modulation of native and recombinant gamma-aminobutyric acid(B) receptors by 2,6-Di-tert-butyl-4-(3-hydroxy-2,2-dimethyl-propyl)-phenol (CGP7930) and its aldehyde analog CGP13501". Molecular Pharmacology. 60 (5): 963–71. doi:10.1124/mol.60.5.963. PMID 11641424.
- ^ Adams CL, Lawrence AJ (2007). "CGP7930: a positive allosteric modulator of the GABAB receptor". CNS Drug Reviews. 13 (3): 308–16. doi:10.1111/j.1527-3458.2007.00021.x. PMC 6494120. PMID 17894647.
- ^ Paterson NE, Vlachou S, Guery S, Kaupmann K, Froestl W, Markou A (July 2008). "Positive modulation of GABA(B) receptors decreased nicotine self-administration and counteracted nicotine-induced enhancement of brain reward function in rats". The Journal of Pharmacology and Experimental Therapeutics. 326 (1): 306–14. doi:10.1124/jpet.108.139204. PMC 2574924. PMID 18445779.
- ^ Urwyler S, Pozza MF, Lingenhoehl K, Mosbacher J, Lampert C, Froestl W, et al. (October 2003). "N,N'-Dicyclopentyl-2-methylsulfanyl-5-nitro-pyrimidine-4,6-diamine (GS39783) and structurally related compounds: novel allosteric enhancers of gamma-aminobutyric acidB receptor function". The Journal of Pharmacology and Experimental Therapeutics. 307 (1): 322–30. doi:10.1124/jpet.103.053074. PMID 12954816. S2CID 26152839.
- ^ Giotti A, Luzzi S, Spagnesi S, Zilletti L (August 1983). "Homotaurine: a GABAB antagonist in guinea-pig ileum". British Journal of Pharmacology. 79 (4): 855–62. doi:10.1111/j.1476-5381.1983.tb10529.x. PMC 2044932. PMID 6652358.
- ^ Kimura T, Saunders PA, Kim HS, Rheu HM, Oh KW, Ho IK (January 1994). "Interactions of ginsenosides with ligand-bindings of GABA(A) and GABA(B) receptors". General Pharmacology. 25 (1): 193–9. doi:10.1016/0306-3623(94)90032-9. PMID 8026706.
- ^ Froestl W, Gallagher M, Jenkins H, Madrid A, Melcher T, Teichman S, et al. (October 2004). "SGS742: the first GABA(B) receptor antagonist in clinical trials". Biochemical Pharmacology. 68 (8): 1479–87. doi:10.1016/j.bcp.2004.07.030. PMID 15451390.
- ^ Bullock R (January 2005). "SGS-742 Novartis". Current Opinion in Investigational Drugs. 6 (1): 108–13. PMID 15675610.
