Alprazolam
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ælˈpræzəlæm/ or /ælˈpreɪzəlæm/ |
| Trade names | Xanax, Xanor, Niravam, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684001 |
| License data |
|
| Pregnancy category |
|
| Dependence liability | Very high[2] |
| Addiction liability | High[2] |
| Routes of administration | Oral |
| Drug class | Benzodiazepine |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80–90% |
| Protein binding | 80% |
| Metabolism | Liver, via cytochrome P450 3A4 |
| Metabolites | • Alpha-hydroxyalprazolam • 4-hydroxyalprazolam |
| Onset of action | 20~60 minutes[8] |
| Elimination half-life | Full release: 11~13 hours[9] Extended release: 11~16 hours[9] |
| Duration of action | Full release: 6 hours[8] Extended release: 11.3 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.044.849 |
| Chemical and physical data | |
| Formula | C17H13ClN4 |
| Molar mass | 308.77 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Alprazolam, sold under the brand name Xanax among others, is a fast-acting, potent tranquilizer of moderate duration within the triazolobenzodiazepine group of chemicals called benzodiazepines.[10] Alprazolam is most commonly used in management of anxiety disorders, specifically panic disorder or generalized anxiety disorder (GAD).[6] Other uses include the treatment of chemotherapy-induced nausea, together with other treatments.[9] GAD improvement occurs generally within a week.[11][12] Alprazolam is generally taken orally (by mouth).[9]
Common side effects include sleepiness, depression or suppressed emotions, mild to severe decreases in motor skills, hiccups, dulling or declining of cognition as well as alertness or general awareness of one's surroundings or even behavior, dry mouth (mildly), decreased heart rate, suppression (physiological inhibition) of general central nervous system activity (opposite of physiological excitation), impairment of judgment (usually in higher than therapeutic doses), marginal to severe decreases in memory formation, decreased ability to process new information, as well as partial to complete amnesia (anterograde amnesia[13]) depending on dosage.[9] Some of the sedation and tiredness may improve within a few days.[14]
Alprazolam withdrawal may occur if use is suddenly decreased.[9][11]
Alprazolam was invented by Jackson Hester Jr. at the Upjohn Company[15] and patented in 1971 and approved for medical use in the United States in 1981.[9][16] Alprazolam is a Schedule IV controlled substance and is a common drug of abuse.[2][17] It is available as a generic medication.[6] In 2021, it was the 42nd most-commonly-prescribed medication in the United States, with more than 15 million prescriptions.[18][19]
Medical uses[edit]


Alprazolam is used in the management of anxiety disorders, panic disorders, and nausea due to chemotherapy.[9] Alprazolam is indicated for the treatment of generalized anxiety disorder and panic disorder with or without agoraphobia in adults.[6]
Panic disorder[edit]
Alprazolam is effective in the relief of moderate to severe anxiety and panic attacks.[6] In Australia, alprazolam is not recommended for the treatment of panic disorder because of concerns regarding tolerance, dependence, and abuse.[20] Most evidence shows that the benefits of alprazolam in treating panic disorder last only four to ten weeks. However, people with panic disorder have been treated on an open basis for up to eight months without apparent loss of benefit.[6]
Alprazolam is recommended by the World Federation of Societies of Biological Psychiatry (WFSBP) for treatment-resistant cases of panic disorder where there is no history of tolerance or dependence.[21]
A 2023 meta-analysis of published and unpublished FDA-submitted regulatory trials of alprazolam extended-release for panic disorder found that only one of five clinical trials showed a positive efficacy outcome (20%) while the rest were negative and did not demonstrate effectiveness.[22] In the published literature, three trials were published conveying a positive outcome (100%), but of these, only one was actually positive and the other two were considered to have been inappropriately spun as positive.[22] The effect size (Hedges's g) of alprazolam for treatment of panic disorder based on the five clinical trials was 0.33 (a small effect) and based on the published trials was 0.47 (a moderate effect), equating to an increase of 0.14 or 42%.[22] The authors concluded that publication bias substantially inflated the effectiveness of alprazolam for panic disorder.[22]
Anxiety disorders[edit]
Anxiety associated with depression is responsive to alprazolam. Clinical studies have shown that the effectiveness is limited to four months for anxiety disorders.[6] However, the research into antidepressant properties of alprazolam is poor and has only assessed its short-term effects against depression.[23] In one study, some long term, high-dosage users of alprazolam developed reversible depression.[24]
In the US, alprazolam is indicated for the treatment of generalized anxiety disorder and panic disorder with or without agoraphobia.[6][7]
In the UK, alprazolam is indicated for short-term symptomatic treatment of anxiety in adults.[4]
Nausea due to chemotherapy[edit]
Alprazolam may be used in combination with other medications for chemotherapy-induced nausea and vomiting.[9]
Contraindications[edit]
Benzodiazepines require special precaution if used in children and in alcohol- or drug-dependent individuals. Particular care should be taken in pregnant or elderly people, people with substance use disorder history, particularly alcohol dependence, and people with comorbid psychiatric disorders.[25]
Alprazolam should be avoided or carefully monitored by medical professionals in individuals with myasthenia gravis, acute narrow-angle glaucoma, severe liver deficiencies such as cirrhosis, severe sleep apnea, pre-existing respiratory depression, marked neuromuscular respiratory, acute pulmonary insufficiency, chronic psychosis, hypersensitivity, allergy to alprazolam or other benzodiazepines, and borderline personality disorder, where it may induce suicidality and dyscontrol.[26][27][28]
Like all central nervous system depressants, alprazolam in larger-than-normal doses can cause significant deterioration in alertness and increase drowsiness, especially in those unaccustomed to the drug's effects.[29]
Side effects[edit]

Sedative drugs, including alprazolam, have been associated with an increased risk of death.[30]
Possible side effects include:
- Anterograde amnesia[31] and concentration problems
- Ataxia and Dysarthria[32]
- Disinhibition[33]
- Drowsiness, dizziness, lightheadedness, fatigue, unsteadiness, impaired coordination, and vertigo[34][35]
- Dry mouth (infrequent)[36]
- Hallucinations (rare)[37]
- Jaundice (very rare)[38]
- Seizure (less common)[39]
- Skin rash, respiratory depression, and constipation[34][35]
- Suicidal ideation or suicide[40]
- Urinary retention (infrequent)[37]
- Muscle weakness[medical citation needed]
In September 2020, the US Food and Drug Administration (FDA) required the boxed warning be updated for all benzodiazepine medicines to describe the risks of abuse, misuse, addiction, physical dependence, and withdrawal reactions consistently across all the medicines in the class.[41]
Paradoxical reactions[edit]
Although unusual, the following paradoxical reactions have been shown to occur:
- Aggression[42]
- Mania, agitation, hyperactivity, and restlessness[43][44][45]
- Rage and hostility[33]
- Twitches and tremor[46]
Food and drug interactions[edit]
Alprazolam is primarily metabolized via CYP3A4.[47] Combining CYP3A4 inhibitors such as cimetidine, erythromycin, norfluoxetine, fluvoxamine, itraconazole, ketoconazole, nefazodone, propoxyphene, and ritonavir delay the hepatic clearance of alprazolam, which may result in its accumulation[48] and increased severity of its side effects.[49][50]
Imipramine and desipramine have been reported to increase an average of 31% and 20% respectively by the concomitant administration of alprazolam tablets.[6] Combined oral contraceptive pills reduce the clearance of alprazolam, which may lead to increased plasma levels of alprazolam and accumulation.[51]
Alcohol is one of the most common interactions; alcohol and alprazolam taken in combination have a synergistic effect on one another, which can cause severe sedation, behavioral changes, and intoxication. The more alcohol and alprazolam taken, the worse the interaction.[33] Combination of alprazolam with the herb kava can result in the development of a semi-comatose state.[52] Plants in the genus Hypericum, including St. John's wort, conversely can lower the plasma levels of alprazolam and reduce its therapeutic effect.[53][54][55]
Pregnancy and breastfeeding[edit]
Benzodiazepines cross the placenta, enter the fetus, and are also excreted in breast milk.[6]
The use of alprazolam during pregnancy is associated with congenital abnormalities,[6][56] and use in the last trimester may cause fetal drug dependence and withdrawal symptoms in the post-natal period as well as neonatal flaccidity and respiratory problems.[57][58] However, in long-term users of benzodiazepines, abrupt discontinuation due to concerns of teratogenesis has a high risk of causing extreme withdrawal symptoms and a severe rebound effect of the underlying mental health disorder. Spontaneous abortions may also result from abrupt withdrawal of psychotropic medications, including benzodiazepines.[59]
Overdose[edit]

The maximum recommended daily dose is 10 milligrams per day.[6]
Overdoses of alprazolam can be mild to severe depending on the quantity ingested and if other drugs are taken in combination.[60]
Alprazolam overdoses cause excess central nervous system (CNS) depression.[37]
Dependence and withdrawal[edit]
The potential for misuse among those taking it for medical reasons is controversial, with some expert reviews stating that the risk is low and similar to that of other benzodiazepine drugs.[61] Others state that there is a substantial risk of misuse and dependence in both patients and non-medical users and that the short half-life and rapid onset of action may increase the risk of misuse.[9][20] Compared to the large number of prescriptions, relatively few individuals increase their dose on their own initiative or engage in drug-seeking behavior.[62]
Alprazolam, like other benzodiazepines, binds to specific sites on the GABAA (gamma-aminobutyric acid) receptor. When bound to these sites, which are referred to as benzodiazepine receptors, it modulates the effect of GABAA receptors and, thus, of GABAergic neurons.[63][unreliable medical source?] Long-term use causes adaptive changes in the benzodiazepine receptors, making them less sensitive to stimulation and thus making the drugs less potent.[64]
Withdrawal and rebound symptoms commonly occur and necessitate a gradual reduction in dosage to minimize withdrawal effects when discontinuing.[11]
Not all withdrawal effects are evidence of true dependence or withdrawal. Recurrence of symptoms such as anxiety may simply indicate that the drug was having its expected anti-anxiety effect and that, in the absence of the drug, the symptom has returned to pretreatment levels. If the symptoms are more severe or frequent, the person may be experiencing a rebound effect due to the removal of the drug. Either of these can occur without the person actually being drug dependent.[64]
Alprazolam and other benzodiazepines may also cause the development of physical dependence, tolerance, and benzodiazepine withdrawal symptoms during rapid dose reduction or cessation of therapy after long-term treatment.[65][66] There is a higher chance of withdrawal reactions if the drug is administered in a higher dosage than recommended, or if a person stops taking the medication altogether without slowly allowing the body to adjust to a lower-dosage regimen.[67][68]
In 1992, Romach and colleagues reported that dose escalation is not a characteristic of long-term alprazolam users and that the majority of long-term alprazolam users change their initial pattern of regular use to one of symptom control only when required.[69]
Some common symptoms of alprazolam discontinuation include malaise, weakness, insomnia, tachycardia, lightheadedness, and dizziness.[70]
Those taking more than 4 mg per day have an increased potential for dependence. This medication may cause withdrawal symptoms upon abrupt withdrawal or rapid tapering, which in some cases have been known to cause seizures, as well as marked delirium similar to that produced by the anticholinergic tropane alkaloids of Datura (scopolamine and atropine).[71][72][73] The discontinuation of this medication may also cause a reaction called rebound anxiety.[medical citation needed]
In a 1983 study, only 5% of patients who had abruptly stopped taking long-acting benzodiazepines after less than 8 months demonstrated withdrawal symptoms, but 43% of those who had been taking them for more than 8 months did. With alprazolam – a short-acting benzodiazepine – taken for 8 weeks, 65% of patients experienced significant rebound anxiety. To some degree, these older benzodiazepines are self-tapering.[74]
The benzodiazepines diazepam and oxazepam have been found to produce fewer withdrawal reactions than alprazolam, temazepam, or lorazepam. Factors that determine the risk of psychological dependence or physical dependence and the severity of the benzodiazepine withdrawal symptoms during dose reduction of alprazolam include: dosage used, length of use, frequency of dosing, personality characteristics of the individual, previous use of cross-dependent/cross-tolerant drugs (alcohol or other sedative-hypnotic drugs), current use of cross-dependent/-tolerant drugs, use of other short-acting, high-potency benzodiazepines,[75][76] and method of discontinuation.[67]
Pharmacology[edit]

Alprazolam is a positive allosteric modulator of the gamma-aminobutyric acid (GABA) type A receptor.[63][unreliable medical source?] When it binds to the receptor, effects of GABA are enhanced leading to inhibition of neurons in the brain. This results in effects including reduced anxiety, muscle relaxant, antidepressant and anticonvulsant activity.[78] The activity of alprazolam in the central nervous system is dose dependent.[6]
Mechanism of action[edit]
Alprazolam is classed as a high-potency triazolobenzodiazepine:[79][80] a benzodiazepine with a triazole ring attached to its structure. As a benzodiazepine, alprazolam produces a variety of therapeutic and adverse effects by binding to the GABAA benzodiazepine receptor site and modulating its function; GABA receptors are the most prolific inhibitory receptor within the brain. The GABA chemical and receptor system mediates inhibitory or calming effects of alprazolam on the nervous system. Binding of alprazolam to the GABAA receptor, a chloride ion channel, enhances the effects of GABA, a neurotransmitter. When GABA binds the GABAA receptor the channel opens and chloride enters the cell which makes it more resistant to depolarisation. Therefore, alprazolam has a depressant effect on synaptic transmission to reduce anxiety.[81][82]
The GABAA receptor is made up of 5 subunits out of a possible 19, and GABAA receptors made up of different combinations of subunits have different properties, different locations within the brain, and, importantly, different activities with regard to benzodiazepines. Alprazolam and other triazolobenzodiazepines such as triazolam that have a triazole ring fused to their diazepine ring appear to have antidepressant properties.[31][83] This is perhaps due to the similarities shared with tricyclic antidepressants, as they have two benzene rings fused to a diazepine ring. Alprazolam causes a marked suppression of the hypothalamic–pituitary–adrenal axis. The therapeutic properties of alprazolam are similar to other benzodiazepines and include anxiolytic, anticonvulsant, muscle relaxant, hypnotic[84] and amnesic; however, it is used mainly as an anxiolytic.[85]
Giving alprazolam, as compared to lorazepam, has been demonstrated to elicit a statistically significant increase in extracellular dopamine D1 and D2 concentrations in the striatum.[86][87]
Pharmacokinetics[edit]

Alprazolam is taken orally, and is absorbed well – 80% of alprazolam binds to proteins in the serum (the majority binding to albumin). The concentration of alprazolam peaks after one to two hours.[6]
Alprazolam is metabolized in the liver, mostly by the enzyme cytochrome P450 3A4 (CYP3A4). Two major metabolites are produced: 4-hydroxyalprazolam and α-hydroxyalprazolam, as well as an inactive benzophenone. The low concentrations and low potencies of 4-hydroxyalprazolam and α-hydroxyalprazolam indicate that they have little to no contribution to the effects of alprazolam.[6]
The metabolites and some unmetabolized alprazolam are filtered out by the kidneys and are excreted in the urine.[6]
Chemistry[edit]
Physical properties[edit]
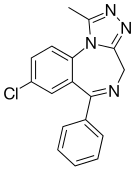
Alprazolam is a triazole and benzodiazepine derivative substituted with a phenyl group at position 6, with a chlorine atom at position 8 and with a methyl group at position 1. It is an analogue of triazolam, the difference between them being the absence of a chlorine atom in the ortho position of the phenyl ring.[88] It is slightly soluble in chloroform, soluble in alcohol, slightly soluble in acetone and insoluble in water.[89] It has a melting point of 228–229.5 °C (442.4–445.1 °F; 501.1–502.6 K).[89][90]
Synthesis[edit]
For the synthesis of alprazolam the same method can be used as for triazolam, excepting that it starts from 2-amino-5-chlorobenzophenone.[91] However, an alternative easier synthesis starting with 2,6-dichloro-4-phenylquinoline has been suggested, in which it reacts with hydrazine giving 6-chloro-2-hydrazino-4-phenylquinoline. Boiling the mixture with triethyl orthoacetate results in cyclization with the formation of the triazole ring. The product undergoes oxidative degradation in the presence of periodate and ruthenium dioxide in acetone solution, giving 2-[4-(3'-methyl-1,2,4-triazolo)]-5-chlorobenzophenone. Oxy-methylation with formaldehyde results in a product that is treated with phosphorus tribromide, when 2-[4-(3'-methyl-5'-bromomethyl-1,2,4-triazolo)]-5-chlorobenzophenone is obtained. By substituting the bromine atom with an amino group conferred by ammonia, it forms alprazolam triazolobenzophenone, following which an intermolecular heterocyclization takes place to obtain alprazolam.[88][92][90]
Detection[edit]
Quantification of alprazolam in blood and plasma samples may be necessary to confirm a diagnosis of intoxication in hospitalized patients, or to provide evidence in the case of crimes e.g., impaired driving arrest, or to assist in a thorough forensic investigation, e.g., in a medicolegal death investigation. Blood or plasma alprazolam concentrations are usually in a range of 10–100 μg/L in persons receiving the drug therapeutically, 100–300 μg/L in those arrested for impaired driving, and 300–2,000 μg/L in victims of acute overdosage. Most of the commercial immunoassays used for the benzodiazepine class of drugs cross-react with alprazolam, but confirmation and quantitative determination are usually done by chromatographic techniques.[93]
Forms of alprazolam[edit]
Alprazolam regular release and orally disintegrating tablets are available as 0.25 mg, 0.5 mg, 1 mg, and 2 mg tablets,[6] while extended release tablets are available as 0.5 mg, 1 mg, 2 mg, and 3 mg.[7] Liquid alprazolam is available in a 1 mg/mL oral concentrate. Inactive ingredients in alprazolam tablets and solutions include microcrystalline cellulose, corn starch, docusate sodium, povidone, sodium starch glycolate, lactose monohydrate, magnesium stearate, colloidal silicon dioxide, and sodium benzoate. In addition, the 0.25 mg tablet contains D&C Yellow No. 10 and the 0.5 mg tablet contains FD&C Yellow No. 6 and D&C Yellow No. 10.[6]
Society and culture[edit]
Patent[edit]
Alprazolam is covered under U.S. patent 3,987,052, which was filed in October 1969, granted in October 1976, and expired in September 1993.[citation needed]
Recreational use[edit]
There is a risk of misuse and dependence in both patients and non-medical users of alprazolam; alprazolam's high affinity binding, high potency, and rapid onset increase its abuse potential. The physical dependence and withdrawal syndrome of alprazolam also add to its addictive nature. In the small subgroup of individuals who escalate their doses there is usually a history of alcohol or other substance use disorders.[20] Despite this, most prescribed alprazolam users do not use their medication recreationally, and the long-term use of benzodiazepines does not generally correlate with the need for dose escalation.[94] However, based on US findings from the Treatment Episode Data Set (TEDS), an annual compilation of patient characteristics in substance abuse treatment facilities in the United States, admissions due to "primary tranquilizer" (including, but not limited to, benzodiazepine-type) drug use increased 79% from 1992 to 2002, suggesting that misuse of benzodiazepines may be on the rise.[95]
The US Centers for Disease Control and Prevention (CDC) reported an 89 percent increase in emergency room visits nationwide related to nonmedical benzodiazepine use between 2004 and 2008.[96][97]
Alprazolam is one of the most commonly prescribed and misused benzodiazepines in the United States.[98][99] A large-scale nationwide U.S. government study conducted by the Substance Abuse and Mental Health Services Administration concluded that, in the US, benzodiazepines are recreationally the most frequently used pharmaceuticals due to their widespread availability, accounting for 35% of all drug-related visits to hospital emergency and urgent care facilities. Men and women are equally likely to use benzodiazepines recreationally. The report found that alprazolam is the most common benzodiazepine for recreational use, followed by clonazepam, lorazepam, and diazepam. The number of emergency department visits due to benzodiazepines increased by 36% between 2004 and 2006.[99]
Regarding the significant increases detected, it is worthwhile to consider that the number of pharmaceuticals dispensed for legitimate therapeutic uses may be increasing over time, and DAWN estimates are not adjusted to take such increases into account. Nor do DAWN estimates take into account the increases in the population or in ED use between 2004 and 2006.[99]
Those at a particularly high risk for misuse and dependence are people with a history of alcoholism or drug abuse and/or dependence[100][101] and people with borderline personality disorder.[102]
The poly-drug use of powerful depressant drugs poses the highest level of health concerns due to a significant increase in the likelihood of experiencing an overdose, which may cause fatal respiratory depression.[103][104]
A 1990 study found that diazepam has a higher misuse potential relative to many other benzodiazepines and that some data suggest that alprazolam and lorazepam resemble diazepam in this respect.[105]
Anecdotally, injection of alprazolam has been reported, causing dangerous damage to blood vessels, closure of blood vessels (embolization) and decay of muscle tissue (rhabdomyolysis).[106] Alprazolam is not very soluble in water—when crushed in water it does not fully dissolve (40 μg/ml of H2O at pH 7).[107] There are also reports of alprazolam being snorted.[108] Due to the low weight of a dose, alprazolam, in one case, was distributed on blotter paper in a manner similar to LSD.[109]
Misuse of alprazolam and other benzodiazepines has been shown to cause cognitive impairment.[110] Alprazolam has typically caused anterograde amnesia effects (inability to recall new events), but a study conducted on mice by the Department of Pharmaceutical Sciences and Drug Research at Punjabi University has also determined that alprazolam can produce retrograde amnesic effects (inability to remember events occurring before amnesia). One 2016 study found that "chronic administration of alprazolam affects memory but attentive and psychomotor performance remained unaffected".[111]
A 2016 randomized controlled trial stated that "overall, long-term benzodiazepine users may not be in their full cognitive state upon withdrawal".[110] Similarly, a 2017 meta-analysis concluded that "a range of neuropsychological functions are impaired as a result of long-term benzodiazepine use, and that these are likely to persist even following withdrawal".[112]
Popular culture[edit]
Slang terms for alprazolam vary from place to place. Some of the more common terms are modified versions of the trade name "Xanax", such as Xannies (or Xanies) and the phonetic equivalent of Zannies;[113][114] references to their drug classes, such as benzos or downers; or remark upon their shape or color (most commonly a straight, perforated tablet or an oval-shaped pill): bars, ladders, Xanbars, Xans, Z-bars, handle bars, beans, footballs, planks, poles, sticks, blues, or blue footballs.[115][116][117][118]
The 2022 film Take Your Pills: Xanax provides an overview of the drug's history and usage.[119]
Availability[edit]
Alprazolam is available in English-speaking countries under the following brand names:[120]
- Alprax, Alprocontin, Alzam, Alzolam, Anzilum, Apo-Alpraz, Helex, Kalma, Mylan-Alprazolam, Niravam, Novo-Alprazol, Nu-Alpraz, Pacyl, Restyl, Tranax, Trika, Xycalm, Xanax, Xanor, Zolam, Zopax.
In December 2013, in anticipation of the rescheduling of alprazolam to Schedule 8 in Australia, Pfizer Australia announced they would be discontinuing the Xanax brand in Australia as it was no longer commercially viable.[121]
Legal status[edit]
Alprazolam has varied legal status depending on jurisdiction:
- In the United States, alprazolam is a prescription drug and is assigned to Schedule IV of the Controlled Substances Act by the Drug Enforcement Administration.[122]
- Under the UK drug misuse classification system, alprazolam is a Class C drug.[5][123] In the UK, alprazolam is not available on the National Health Service (NHS) and can only be obtained on a private prescription.[124]
- In Ireland, alprazolam is a Schedule 4 medicine.[125]
- In Sweden, alprazolam is a prescription drug in List IV (Schedule 4) under the Narcotics Drugs Act (1968).[126]
- In the Netherlands, alprazolam is a List 2 substance of the Opium Law and is available for prescription.[citation needed]
- In Germany, alprazolam can be prescribed normally in doses up to 1 mg. Higher doses are scheduled as Anlage III drugs and require a special prescription form.[citation needed]
- In Australia, alprazolam, as of February 2014, is a Schedule 8 medication.[127][128][129]
- In the Philippines, alprazolam is legally classified as a "dangerous drug" under the Comprehensive Dangerous Drugs Act of 2002, along with other schedule drugs listed in the 1971 Convention on Psychotropic Substances.[130] The importation of dangerous drugs including alprazolam, requires authorization from the Philippine Drug Enforcement Agency.[131]
Internationally, alprazolam is included under the United Nations Convention on Psychotropic Substances as Schedule IV.[132]
References[edit]
- ^ "Alprazolam Use During Pregnancy". Drugs.com. 4 May 2020. Archived from the original on 20 June 2020. Retrieved 8 June 2020.
- ^ a b c Ait-Daoud N, Hamby AS, Sharma S, Blevins D (2018). "A Review of Alprazolam Use, Misuse, and Withdrawal". Journal of Addiction Medicine. 12 (1): 4–10. doi:10.1097/ADM.0000000000000350. PMC 5846112. PMID 28777203.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b "Xanax Summary of Product Characteristics (SmPC)". (emc). 28 July 2023. Retrieved 20 November 2023.
- ^ a b "List of most commonly encountered drugs currently controlled under the misuse of drugs legislation". GOV.UK. 20 October 2016. Retrieved 20 November 2023.
- ^ a b c d e f g h i j k l m n o p q r "Xanax- alprazolam tablet". DailyMed. Archived from the original on 17 January 2021. Retrieved 19 February 2021.
- ^ a b c "Xanax XR- alprazolam tablet, extended release". DailyMed. 12 March 2021. Archived from the original on 17 April 2021. Retrieved 2 June 2022.
- ^ a b Lilley LL, Snyder JS, Collins SR (2016). Pharmacology for Canadian Health Care Practice. Elsevier Health Sciences. p. 329. ISBN 9781771720663. Archived from the original on 10 May 2022. Retrieved 13 September 2020.
- ^ a b c d e f g h i j American Society of Health-System Pharmacists (13 November 2017). "Alprazolam Monograph for Professionals". Drugs.com. Archived from the original on 7 December 2010. Retrieved 25 October 2018.
- ^ Goldberg R (2009). Drugs Across the Spectrum. Cengage Learning. p. 195. ISBN 9781111782009. Archived from the original on 4 June 2020. Retrieved 24 August 2017.
- ^ a b c Verster JC, Volkerts ER (2004). "Clinical pharmacology, clinical efficacy, and behavioral toxicity of alprazolam: a review of the literature". CNS Drug Reviews. 10 (1): 45–76. doi:10.1111/j.1527-3458.2004.tb00003.x. PMC 6741717. PMID 14978513.
- ^ Tampi RR, Muralee S, Weder ND, Penland H, eds. (2008). Comprehensive Review of Psychiatry. Philadelphia, PA: Wolters Kluwer/ Lippincott Williams & Wilkins Health. p. 226. ISBN 978-0-7817-7176-4. Archived from the original on 19 March 2017. Retrieved 13 March 2016.
- ^ Chowdhury ZS, Morshed MM, Shahriar M, Bhuiyan MA, Islam SM, Bin Sayeed MS (2016). "The Effect of Chronic Alprazolam Intake on Memory, Attention, and Psychomotor Performance in Healthy Human Male Volunteers". Behavioural Neurology. 2016: 3730940. doi:10.1155/2016/3730940. PMC 4947648. PMID 27462136.
- ^ Pavuluri MN, Janicak PG, Marder SR (2010). Principles and Practice of Psychopharmacotherapy (5th ed.). Philadelphia, PA: Wolters Kluwer Health/ Lippincott Williams & Wilkins. p. 535. ISBN 978-1-60547-565-3. Archived from the original on 17 July 2020. Retrieved 13 March 2016.
- ^ Ainsworth SJ (9 December 2013). "Hester Dies At 80". Chemical & Engineering News. Retrieved 21 April 2024.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 536. ISBN 9783527607495. Archived from the original on 28 August 2021. Retrieved 13 September 2020.
- ^ Malamed SF (2009). Sedation: A Guide to Patient Management. Elsevier Health Sciences. p. 105. ISBN 978-0323075961. Archived from the original on 10 May 2022. Retrieved 13 September 2020.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Alprazolam - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ a b c Moylan S, Giorlando F, Nordfjærn T, Berk M (March 2012). "The role of alprazolam for the treatment of panic disorder in Australia" (PDF). The Australian and New Zealand Journal of Psychiatry. 46 (3): 212–24. doi:10.1177/0004867411432074. PMID 22391278. S2CID 11006795. Archived (PDF) from the original on 22 April 2017. Retrieved 27 October 2012.
- ^ Bandelow B, Zohar J, Hollander E, Kasper S, Möller HJ (October 2002). "World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and posttraumatic stress disorders". The World Journal of Biological Psychiatry. 3 (4): 171–99. doi:10.3109/15622970209150621. PMID 12516310.
- ^ a b c d Ahn-Horst RY, Turner EH (October 2023). "Unpublished trials of alprazolam XR and their influence on its apparent efficacy for panic disorder". Psychol Med. 54 (5): 1026–1033. doi:10.1017/S0033291723002830. PMID 37853797.
- ^ van Marwijk H, Allick G, Wegman F, Bax A, Riphagen II (July 2012). van Marwijk H (ed.). "Alprazolam for depression" (PDF). The Cochrane Database of Systematic Reviews. 7 (7): CD007139. doi:10.1002/14651858.CD007139.pub2. PMC 6769182. PMID 22786504. Archived (PDF) from the original on 11 August 2017. Retrieved 18 July 2017.
- ^ Lydiard RB, Laraia MT, Ballenger JC, Howell EF (May 1987). "Emergence of depressive symptoms in patients receiving alprazolam for panic disorder". The American Journal of Psychiatry. 144 (5): 664–5. doi:10.1176/ajp.144.5.664. PMID 3578580.
- ^ Authier N, Balayssac D, Sautereau M, Zangarelli A, Courty P, Somogyi AA, et al. (November 2009). "Benzodiazepine dependence: focus on withdrawal syndrome". Annales Pharmaceutiques Françaises. 67 (6): 408–13. doi:10.1016/j.pharma.2009.07.001. PMID 19900604.
- ^ "Alprazolam". British National Formulary. 2007. Archived from the original on 20 July 2012. Retrieved 3 August 2007.
- ^ Hori A (February 1998). "Pharmacotherapy for personality disorders". Psychiatry and Clinical Neurosciences. 52 (1): 13–9. doi:10.1111/j.1440-1819.1998.tb00967.x. PMID 9682928. S2CID 26667425.
- ^ Gardner DL, Cowdry RW (January 1985). "Alprazolam-induced dyscontrol in borderline personality disorder". The American Journal of Psychiatry. 142 (1): 98–100. doi:10.1176/ajp.142.1.98. PMID 2857071.
- ^ Kozená L, Frantik E, Horváth M (May 1995). "Vigilance impairment after a single dose of benzodiazepines". Psychopharmacology. 119 (1): 39–45. doi:10.1007/BF02246052. PMID 7675948. S2CID 2618084.
- ^ Kripke DF (February 2016). "Mortality Risk of Hypnotics: Strengths and Limits of Evidence" (PDF). Drug Safety. 39 (2): 93–107. doi:10.1007/s40264-015-0362-0. PMID 26563222. S2CID 7946506. Archived (PDF) from the original on 14 March 2020. Retrieved 29 August 2019.
- ^ a b Barbee JG (October 1993). "Memory, benzodiazepines, and anxiety: integration of theoretical and clinical perspectives". The Journal of Clinical Psychiatry. 54 (Suppl): 86–97, discussion 98–101. PMID 8262893.
- ^ Cassano GB, Toni C, Petracca A, Deltito J, Benkert O, Curtis G, et al. (March 1994). "Adverse effects associated with the short-term treatment of panic disorder with imipramine, alprazolam or placebo" (PDF). European Neuropsychopharmacology. 4 (1): 47–53. doi:10.1016/0924-977X(94)90314-X. hdl:2027.42/31721. PMID 8204996. S2CID 14557358. Archived (PDF) from the original on 20 February 2020. Retrieved 18 July 2017.
- ^ a b c Michel L, Lang JP (2003). "[Benzodiazepines and forensic aspects]" [Benzodiazepines and Forensic Aspects]. L'Encephale (in French). 29 (6): 479–85. PMID 15029082. Archived from the original on 2 June 2022. Retrieved 31 December 2020.
- ^ a b Rawson NS, Rawson MJ (1999). "Acute adverse event signalling scheme using the Saskatchewan Administrative health care utilization datafiles: results for two benzodiazepines". Canadian Journal of Clinical Pharmacology. 6 (3): 159–66. PMID 10495368.
- ^ a b "Alprazolam – Complete Medical Information Regarding This Treatment of Anxiety Disorders". Medicinenet.com. MedicineNet. Archived from the original on 9 March 2019. Retrieved 2 August 2007.
- ^ Elie R, Lamontagne Y (June 1984). "Alprazolam and diazepam in the treatment of generalized anxiety". Journal of Clinical Psychopharmacology. 4 (3): 125–9. doi:10.1097/00004714-198406000-00002. PMID 6145726. S2CID 735484.
- ^ a b c "Alprazolam Side Effects, Interactions and Information". Drugs.com. Archived from the original on 19 August 2007. Retrieved 2 August 2007.
- ^ Noyes R, DuPont RL, Pecknold JC, Rifkin A, Rubin RT, Swinson RP, et al. (May 1988). "Alprazolam in panic disorder and agoraphobia: results from a multicenter trial. II. Patient acceptance, side effects, and safety". Archives of General Psychiatry. 45 (5): 423–8. doi:10.1001/archpsyc.1988.01800290037005. PMID 3358644.
- ^ "Side Effects". Mayo Clinic. 1 May 2019. Archived from the original on 28 November 2020. Retrieved 22 May 2019.
- ^ Dodds TJ (March 2017). "Prescribed Benzodiazepines and Suicide Risk: A Review of the Literature". The Primary Care Companion for CNS Disorders. 19 (2). doi:10.4088/PCC.16r02037. PMID 28257172.
- ^ "FDA expands Boxed Warning to improve safe use of benzodiazepine drug". U.S. Food and Drug Administration (FDA). 23 September 2020. Archived from the original on 24 September 2020. Retrieved 23 September 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Rapaport M, Braff DL (January 1985). "Alprazolam and hostility". The American Journal of Psychiatry. 142 (1): 146a–146. doi:10.1176/ajp.142.1.146a. PMID 2857070.
- ^ Arana GW, Pearlman C, Shader RI (March 1985). "Alprazolam-induced mania: two clinical cases". The American Journal of Psychiatry. 142 (3): 368–9. doi:10.1176/ajp.142.3.368. PMID 2857534.
- ^ Strahan A, Rosenthal J, Kaswan M, Winston A (July 1985). "Three case reports of acute paroxysmal excitement associated with alprazolam treatment". The American Journal of Psychiatry. 142 (7): 859–61. CiteSeerX 10.1.1.461.6316. doi:10.1176/ajp.142.7.859. PMID 2861755.
- ^ Reddy J, Khanna S, Anand U, Banerjee A (August 1996). "Alprazolam-induced hypomania". The Australian and New Zealand Journal of Psychiatry. 30 (4): 550–2. CiteSeerX 10.1.1.941.5119. doi:10.3109/00048679609065031. PMID 8887708. S2CID 38542776.
- ^ Béchir M, Schwegler K, Chenevard R, Binggeli C, Caduff C, Büchi S, et al. (July 2007). "Anxiolytic therapy with alprazolam increases muscle sympathetic activity in patients with panic disorders". Autonomic Neuroscience. 134 (1–2): 69–73. doi:10.1016/j.autneu.2007.01.007. PMID 17363337. S2CID 44980758.
- ^ Otani K (2003). "[Cytochrome P450 3A4 and Benzodiazepines]". Seishin Shinkeigaku Zasshi = Psychiatria et Neurologia Japonica (in Japanese). 105 (5): 631–42. PMID 12875231.
- ^ Dresser GK, Spence JD, Bailey DG (January 2000). "Pharmacokinetic-pharmacodymic consequences and clinical relevance of cytochrome P450 3A4 inhibition". Clinical Pharmacokinetics. 38 (1): 41–57. doi:10.2165/00003088-200038010-00003. PMID 10668858. S2CID 37743328.
- ^ Greenblatt DJ, Wright CE (June 1993). "Clinical pharmacokinetics of alprazolam. Therapeutic implications". Clinical Pharmacokinetics. 24 (6): 453–71. doi:10.2165/00003088-199324060-00003. PMID 8513649. S2CID 46252882.
- ^ Wang JS, DeVane CL (2003). "Pharmacokinetics and drug interactions of the sedative hypnotics" (PDF). Psychopharmacology Bulletin. 37 (1): 10–29. doi:10.1007/BF01990373. PMID 14561946. S2CID 1543185. Archived from the original (PDF) on 9 July 2007.
- ^ Back DJ, Orme ML (June 1990). "Pharmacokinetic drug interactions with oral contraceptives". Clinical Pharmacokinetics. 18 (6): 472–84. doi:10.2165/00003088-199018060-00004. PMID 2191822. S2CID 32523973.
- ^ Izzo AA, Ernst E (2001). "Interactions between herbal medicines and prescribed drugs: a systematic review". Drugs. 61 (15): 2163–75. doi:10.2165/00003495-200161150-00002. PMID 11772128. S2CID 46983699. Archived from the original on 25 May 2021. Retrieved 31 December 2020.
- ^ Izzo AA (March 2004). "Drug interactions with St. John's Wort (Hypericum perforatum): a review of the clinical evidence". International Journal of Clinical Pharmacology and Therapeutics. 42 (3): 139–48. doi:10.5414/CPP42139. PMID 15049433.
- ^ Madabushi R, Frank B, Drewelow B, Derendorf H, Butterweck V (March 2006). "Hyperforin in St. John's wort drug interactions". European Journal of Clinical Pharmacology. 62 (3): 225–33. doi:10.1007/s00228-006-0096-0. PMID 16477470. S2CID 22510479.
- ^ Izzo AA, Ernst E (2009). "Interactions between herbal medicines and prescribed drugs: an updated systematic review". Drugs. 69 (13): 1777–98. doi:10.2165/11317010-000000000-00000. PMID 19719333. S2CID 25720882.
- ^ Oo CY, Kuhn RJ, Desai N, Wright CE, McNamara PJ (September 1995). "Pharmacokinetics in lactating women: prediction of alprazolam transfer into milk". British Journal of Clinical Pharmacology. 40 (3): 231–6. doi:10.1111/j.1365-2125.1995.tb05778.x. PMC 1365102. PMID 8527284.
- ^ Iqbal MM, Sobhan T, Ryals T (January 2002). "Effects of commonly used benzodiazepines on the fetus, the neonate, and the nursing infant" (PDF). Psychiatric Services. 53 (1): 39–49. doi:10.1176/appi.ps.53.1.39. PMID 11773648. Archived (PDF) from the original on 26 March 2018. Retrieved 18 July 2017.
- ^ García-Algar O, López-Vílchez MA, Martín I, Mur A, Pellegrini M, Pacifici R, et al. (2007). "Confirmation of gestational exposure to alprazolam by analysis of biological matrices in a newborn with neonatal sepsis". Clinical Toxicology. 45 (3): 295–8. doi:10.1080/15563650601072191. PMID 17453885. S2CID 2155435.
- ^ Einarson A, Selby P, Koren G (January 2001). "Abrupt discontinuation of psychotropic drugs during pregnancy: fear of teratogenic risk and impact of counselling". Journal of Psychiatry & Neuroscience. 26 (1): 44–8. PMC 1408034. PMID 11212593.
- ^ Isbister GK, O'Regan L, Sibbritt D, Whyte IM (July 2004). "Alprazolam is relatively more toxic than other benzodiazepines in overdose". British Journal of Clinical Pharmacology. 58 (1): 88–95. doi:10.1111/j.1365-2125.2004.02089.x. PMC 1884537. PMID 15206998.
- ^ Work Group on Panic Disorder (January 2009). APA Practice Guideline for the Treatment of Patients With Panic Disorder (PDF). Vol. 1 (2nd ed.). doi:10.1176/appi.books.9780890423905.154688. ISBN 978-0-89042-385-1. Archived (PDF) from the original on 7 June 2019. Retrieved 25 October 2018.
- ^ "DEA Brief Benzodiazepines". Archived from the original on 12 March 2009. Retrieved 1 October 2011.
Given the millions of prescriptions written for benzodiazepines (about 100 million in 1999), relatively few individuals increase their dose on their own initiative or engage in drug-seeking behavior.
- ^ a b "Alprazolam". The Drug Classroom. Archived from the original on 22 January 2019. Retrieved 21 January 2019.
- ^ a b Stahl SM (June 2000). Essential psychopharmacology : neuroscientific basis and practical application (2nd ed.). Cambridge, United Kingdom: Cambridge University Press. pp. 501–502. ISBN 0-521-64154-3. OCLC 43051626.
- ^ Juergens SM, Morse RM (May 1988). "Alprazolam dependence in seven patients". The American Journal of Psychiatry. 145 (5): 625–7. doi:10.1176/ajp.145.5.625. PMID 3258735.
- ^ Klein E (2002). "The role of extended-release benzodiazepines in the treatment of anxiety: a risk-benefit evaluation with a focus on extended-release alprazolam". The Journal of Clinical Psychiatry. 63 (Suppl 14): 27–33. PMID 12562116. Archived from the original on 29 August 2017. Retrieved 18 July 2017.
- ^ a b Ashton H (August 2002). "The Ashton Manual – Benzodiazepines: How They Work and How to Withdraw". Benzo.org.uk. Archived from the original on 14 September 2008. Retrieved 31 October 2008.
- ^ Closser MH, Brower KJ (1994). "Treatment of alprazolam withdrawal with chlordiazepoxide substitution and taper" (PDF). Journal of Substance Abuse Treatment. 11 (4): 319–23. doi:10.1016/0740-5472(94)90042-6. hdl:2027.42/31476. PMID 7966502. Archived (PDF) from the original on 29 June 2020. Retrieved 18 July 2017.
- ^ Romach MK, Somer GR, Sobell LC, Sobell MB, Kaplan HL, Sellers EM (October 1992). "Characteristics of long-term alprazolam users in the community". Journal of Clinical Psychopharmacology. 12 (5): 316–321. doi:10.1097/00004714-199210000-00004. PMID 1479048. S2CID 7738529.
- ^ Fyer AJ, Liebowitz MR, Gorman JM, Campeas R, Levin A, Davies SO, et al. (March 1987). "Discontinuation of alprazolam treatment in panic patients". The American Journal of Psychiatry. 144 (3): 303–8. doi:10.1176/ajp.144.3.303. PMID 3826428. Archived from the original on 1 November 2019. Retrieved 10 December 2008.
- ^ Breier A, Charney DS, Nelson JC (December 1984). "Seizures induced by abrupt discontinuation of alprazolam". The American Journal of Psychiatry. 141 (12): 1606–7. doi:10.1176/ajp.141.12.1606. PMID 6150649.
- ^ Noyes R, Perry PJ, Crowe RR, Coryell WH, Clancy J, Yamada T, et al. (January 1986). "Seizures following the withdrawal of alprazolam". The Journal of Nervous and Mental Disease. 174 (1): 50–2. doi:10.1097/00005053-198601000-00009. PMID 2867122.
- ^ Levy AB (January 1984). "Delirium and seizures due to abrupt alprazolam withdrawal: case report". The Journal of Clinical Psychiatry. 45 (1): 38–9. PMID 6141159.
- ^ Schatzberg A, DeBattista C (2003). Manual of Clinical Psychopharmacology. Washington, DC: American Psychiatric Pub. p. 391. ISBN 978-1-58562-209-2. Archived from the original on 26 June 2014. Retrieved 22 January 2014.
- ^ Wolf B, Griffiths RR (December 1991). "Physical dependence on benzodiazepines: differences within the class". Drug and Alcohol Dependence. 29 (2): 153–6. doi:10.1016/0376-8716(91)90044-Y. PMID 1686752.
- ^ Higgitt A, Fonagy P, Lader M (1988). "The natural history of tolerance to the benzodiazepines". Psychological Medicine. Monograph Supplement. 13: 1–55. doi:10.1017/S0264180100000412. PMID 2908516. S2CID 38037200.
- ^ Masiulis S, Desai R, Uchański T, Serna Martin I, Laverty D, Karia D, et al. (January 2019). "GABAA receptor signalling mechanisms revealed by structural pharmacology". Nature. 565 (7740): 454–459. Bibcode:2019Natur.565..454M. doi:10.1038/s41586-018-0832-5. PMC 6370056. PMID 30602790.
- ^ Huybrechts I (1991). "The pharmacology of alprazolam: a review". Clinical Therapeutics. 13 (1): 100–117. PMID 2029716.
- ^ Skelton KH, Nemeroff CB, Owens MJ (October 2004). "Spontaneous withdrawal from the triazolobenzodiazepine alprazolam increases cortical corticotropin-releasing factor mRNA expression". The Journal of Neuroscience. 24 (42): 9303–9312. doi:10.1523/JNEUROSCI.1737-04.2004. PMC 6730084. PMID 15496666.
- ^ Chouinard G (2004). "Issues in the clinical use of benzodiazepines: potency, withdrawal, and rebound". The Journal of Clinical Psychiatry. 65 (Suppl 5): 7–12. PMID 15078112. Archived from the original on 29 August 2017. Retrieved 18 July 2017.
- ^ Andrew H, Lonsdale D, Burrage D, Baker E (2014). Top 100 Drugs: Clinical Pharmacology and Practical Prescribing. Edinburgh. ISBN 9780702055164. OCLC 864676781.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Huybrechts I (January 1991). "The pharmacology of alprazolam: a review". Clinical Therapeutics. 13 (1): 100–117. PMID 2029716.
- ^ White G, Gurley DA (February 1995). "Alpha subunits influence Zn block of gamma 2 containing GABAA receptor currents". NeuroReport. 6 (3): 461–464. doi:10.1097/00001756-199502000-00014. PMID 7766843.
- ^ Arvat E, Giordano R, Grottoli S, Ghigo E (September 2002). "Benzodiazepines and anterior pituitary function". Journal of Endocrinological Investigation. 25 (8): 735–47. doi:10.1007/bf03345110. PMID 12240908. S2CID 32002501.
- ^ "PubChem Compound Summary for CID 2118, Alprazolam". PubChem. Archived from the original on 19 December 2023. Retrieved 19 December 2023.
- ^ Bentué-Ferrer D, Reymann JM, Tribut O, Allain H, Vasar E, Bourin M (February 2001). "Role of dopaminergic and serotonergic systems on behavioral stimulatory effects of low-dose alprazolam and lorazepam". European Neuropsychopharmacology. 11 (1): 41–50. doi:10.1016/S0924-977X(00)00137-1. PMID 11226811. S2CID 24653686.
- ^ Giardino L, Zanni M, Pozza M, Bettelli C, Covelli V (March 1998). "Dopamine receptors in the striatum of rats exposed to repeated restraint stress and alprazolam treatment". European Journal of Pharmacology. 344 (2–3): 143–7. doi:10.1016/s0014-2999(97)01608-7. PMID 9600648.
- ^ a b Ruben V, Hruby V (2006). Synthesis of Essential Drugs (1 ed.). Elsevier Science. pp. 76–77. ISBN 9780444521668.
- ^ a b O'Neil MJ, Smith A, Heckelman PE, Budavari S (2013). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals (Fifteenth ed.). Whitehouse Station, N.J.: Merck. p. 58. ISBN 978-1-84973-670-1.
- ^ a b US 3709898, Hester JJ, "Process for the production of triazolobenzodiazepines and intermediates", issued 9 January 1973, assigned to Upjohn Company. "Process for the production of triazolobenzodiazepines and intermediates". Archived from the original on 21 February 2020. Retrieved 2 June 2022.
- ^ US 3987052, Hester Jr JB, "6-Phenyl-4H-s-triazolo(4,3-a)(1,4)benzodiazepines", issued 19 October 1976, assigned to Upjohn Company "6-Phenyl-4H-s-triazolo[4,3-a][1,4]benzodiazepines". Archived from the original on 20 February 2020. Retrieved 21 February 2020.
- ^ Walser, A, Zenchoff G (1977). "Quinazolines and 1,4-benzodiazepines. 81. s-Triazolo[4,3-a][1,4]benzodiazepines by oxidative cyclization of hydrazones". Journal of Medicinal Chemistry. 20 (12): 1694–1697. doi:10.1021/jm00222a035. PMID 592339.
- ^ Baselt R (2011). Disposition of Toxic Drugs and Chemicals in Man (9th ed.). Seal Beach, CA: Biomedical Publications. pp. 45–48. ISBN 978-0-9626523-8-7.
- ^ Soumerai SB, Simoni-Wastila L, Singer C, Mah C, Gao X, Salzman C, et al. (July 2003). "Lack of relationship between long-term use of benzodiazepines and escalation to high dosages". Psychiatric Services. 54 (7): 1006–11. doi:10.1176/appi.ps.54.7.1006. PMID 12851438.
- ^ Licata SC, Rowlett JK (July 2008). "Abuse and dependence liability of benzodiazepine-type drugs: GABA(A) receptor modulation and beyond". Pharmacology Biochemistry and Behavior. 90 (1): 74–89. doi:10.1016/j.pbb.2008.01.001. PMC 2453238. PMID 18295321.
- ^ Centers for Disease Control and Prevention (CDC) (June 2010). "Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008" (PDF). MMWR. Morbidity and Mortality Weekly Report. 59 (23): 705–9. PMID 20559200.
- ^ Goodnough A (14 September 2011). "Abuse of Xanax Leads a Clinic to Halt Supply". The New York Times. Archived from the original on 24 February 2017. Retrieved 26 February 2017.
- ^ Galanter M (2008). The American Psychiatric Publishing Textbook of Substance Abuse Treatment (4th ed.). American Psychiatric Publishing. p. 222. ISBN 978-1-58562-276-4. Retrieved 22 January 2014.
- ^ a b c "Drug Abuse Warning Network, 2006: National Estimates of Drug-Related Emergency Department Visits" (PDF). U.S. Department of Health and Human Services. Substance Abuse and Mental Health Services Administration. 2006. Archived from the original (PDF) on 12 March 2012. Retrieved 13 February 2012.
- ^ Ballenger JC (December 1984). "Psychopharmacology of the anxiety disorders". The Psychiatric Clinics of North America. 7 (4): 757–71. doi:10.1016/S0193-953X(18)30728-7. PMID 6151647.
- ^ Ciraulo DA, Barnhill JG, Greenblatt DJ, Shader RI, Ciraulo AM, Tarmey MF, et al. (September 1988). "Abuse liability and clinical pharmacokinetics of alprazolam in alcoholic men". The Journal of Clinical Psychiatry. 49 (9): 333–7. PMID 3417618.
- ^ Vorma H, Naukkarinen HH, Sarna SJ, Kuoppasalmi KI (2005). "Predictors of benzodiazepine discontinuation in subjects manifesting complicated dependence". Substance Use & Misuse. 40 (4): 499–510. doi:10.1081/JA-200052433. PMID 15830732. S2CID 1366333.
- ^ Walker BM, Ettenberg A (April 2003). "The effects of alprazolam on conditioned place preferences produced by intravenous heroin". Pharmacology Biochemistry and Behavior. 75 (1): 75–80. doi:10.1016/S0091-3057(03)00043-1. PMID 12759115. S2CID 23137094.
- ^ "OSAM-O-GRAM Highlights of Statewide Drug Use Trends" (PDF). Ohio, US: Wright State University and the University of Akron. January 2008. Archived from the original (PDF) on 7 August 2011. Retrieved 10 December 2008.
- ^ Griffiths RR, Wolf B (August 1990). "Relative abuse liability of different benzodiazepines in drug abusers". Journal of Clinical Psychopharmacology. 10 (4): 237–43. doi:10.1097/00004714-199008000-00002. PMID 1981067. S2CID 28209526.
- ^ Wang EC, Chew FS (2006). "MR Findings of Alprazolam Injection into the Femoral Artery with Microembolization and Rhabdomyolysis". Radiology Case Reports. 1 (3): 99–102. doi:10.2484/rcr.v1i3.33. PMC 4891562. PMID 27298694.
- ^ "DB00404 (Alprazolam)". Canada: DrugBank. 26 June 2008. Archived from the original on 29 January 2008. Retrieved 12 July 2011.
- ^ Sheehan MF, Sheehan DV, Torres A, Coppola A, Francis E (1991). "Snorting benzodiazepines". The American Journal of Drug and Alcohol Abuse. 17 (4): 457–68. doi:10.3109/00952999109001605. PMID 1684083.
- ^ "Intelligence Alert – Xanax Blotter Paper in Bartlesville, Oklahoma" (Microgram Bulletin). US Drug Enforcement Administration. May 2008. Archived from the original on 21 May 2008.
- ^ a b Chowdhury ZS, Morshed MM, Shahriar M, Bhuiyan MA, Islam SM, Bin Sayeed MS (2016). "The Effect of Chronic Alprazolam Intake on Memory, Attention, and Psychomotor Performance in Healthy Human Male Volunteers". Behavioural Neurology. 2016: 3730940. doi:10.1155/2016/3730940. PMC 4947648. PMID 27462136.
- ^ Chowdhury ZS, Morshed MM, Shahriar M, Bhuiyan MA, Islam SM, Bin Sayeed MS (2016). "The Effect of Chronic Alprazolam Intake on Memory, Attention, and Psychomotor Performance in Healthy Human Male Volunteers". Behavioural Neurology. 2016: 3730940. doi:10.1155/2016/3730940. PMC 4947648. PMID 27462136.
- ^ Crowe SF, Stranks EK (November 2018). "The Residual Medium and Long-term Cognitive Effects of Benzodiazepine Use: An Updated Meta-analysis". Archives of Clinical Neuropsychology. 33 (7): 901–911. doi:10.1093/arclin/acx120. PMID 29244060.
- ^ Mesibov G (2004). Outer Strength, Inner Strength. Xulon Press. p. 213. ISBN 9781594675041. Archived from the original on 26 April 2021. Retrieved 18 July 2017.
Some people sell Xanax on the street for ten or fifteen dollars a pill. They call them Xannies.
- ^ Curry M (2009). Dancing with the Devil: How Puff Burned the Bad Boys of Hip-hop. NewMark Books. p. 120. ISBN 9780615276502. Archived from the original on 27 April 2021. Retrieved 18 July 2017.
Puff would get so wired sometimes – his favorites were weed, ecstasy and xannies (Xanax) – that he wouldn't realize that he was speeding.
- ^ "Street Names for Xanax". Axis Residential Treatment. Archived from the original on 22 February 2014. Retrieved 14 February 2014.
- ^ "Street Names for Alprazolam". Alprazolamaddictionhelp.com. Archived from the original on 22 February 2014. Retrieved 14 February 2014.
- ^ FDA (2017). "Both opioids and benzodiazepines depress the central nervous system (CNS). FDA Drug Safety Communication: FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines; requires its strongest warning". U.S. Food and Drug Administration (FDA). ("Additional Information for Patients > bullet 5"). Archived from the original on 1 November 2017. Retrieved 15 October 2017.
Do not take opioid pain medicines with benzodiazepines or other medicines that depress the CNS without discussing it with your health Do not take opioid pain medicines with benzodiazepines or other medicines that depress the CNS without discussing it with your health care professional. Do not take opioid cough medicines with benzodiazepines or other medicines that depress the CNS professional. Do not take opioid cough medicines with benzodiazepines or other medicines that depress the CNS
- ^ "Xanax (Alprazolam) Drug Slang/Code Words". MPR. 2 August 2017. Archived from the original on 14 December 2019. Retrieved 17 March 2019.
- ^ Carroll L (8 December 2022). "New Netflix documentary calls out Xanax overuse: What doctors want you to know". Today. Retrieved 12 December 2022.
- ^ "Benzodiazepine Names". Non-benzodiazepines.org.uk. Archived from the original on 8 December 2008. Retrieved 31 October 2008.
- ^ "Discontinuation of Xanax" (PDF). Pfizer Australia. Archived from the original (PDF) on 4 February 2014.
- ^ "DEA, Drug Scheduling". Drug Enforcement Administration. Archived from the original on 4 November 2008. Retrieved 31 October 2008.
- ^ "Misuse of Drugs Act 1971 (c. 38)". The UK Statute Law database. 1991. Archived from the original on 27 November 2010. Retrieved 2 August 2007.
- ^ British Medical Association, Royal Pharmaceutical Society of Great Britain (September 2010). "4.1.2: Anxiolytics". British National Formulary (BNF 60). United Kingdom: BMJ Group and RPS Publishing. p. 212. ISBN 978-0-85369-931-6.
- ^ "Misuse of Drugs (Amendment) Regulations". Irish Statute Book. Office of the Attorney General. 1993. Archived from the original on 30 January 2015. Retrieved 7 September 2009.
- ^ "Läkemedelsverkets föreskrifter (LVFS 2011:10) om förteckningar över narkotika" [Medical Products Agency on the lists of drugs] (PDF) (in Swedish). Sweden: Läkemedelsverket. October 2011. Archived from the original (PDF) on 16 May 2013. Retrieved 6 August 2012.
- ^ "Alprazolam to be rescheduled from next year". 2013. Archived from the original on 27 March 2014. Retrieved 8 November 2013.
- ^ "2.6 Flubromazolam". Therapeutic Goods Administration (TGA). 3 February 2016. Archived from the original on 17 May 2018. Retrieved 2 June 2022.
- ^ "Up-scheduling of alprazolam from Schedule 4 to Schedule 8". NSW Health Pharmaceutical services. 2 June 2022. Archived from the original on 25 March 2022. Retrieved 2 June 2022.
- ^ "Comprehensive Dangerous Drugs Act of 2002". The LawPhil Project. 2002. Archived from the original on 15 September 2018. Retrieved 17 August 2018.
- ^ "BOC seizes regulated and prohibited drugs at NAIA". GMA News. 15 October 2015. Archived from the original on 27 June 2020. Retrieved 17 August 2018.
- ^ "List of Psychotropic Substances under International Control" (PDF). International Narcotics Control Board. August 2003. Archived from the original (PDF) on 17 December 2008. Retrieved 7 December 2008.
