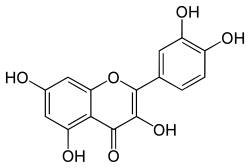
Quercetin
| |

| |
| Names | |
|---|---|
| Pronunciation | /ˈkwɜːrsɪtɪn/ |
| IUPAC name
3,3′,4′,5,7-Pentahydroxyflavone
| |
| Preferred IUPAC name
2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-4H-1-benzopyran-4-one | |
| Other names
5,7,3′,4′-flavon-3-ol, Sophoretin, Meletin, Quercetine, Xanthaurine, Quercetol, Quercitin, Quertine, Flavin meletin
| |
| Identifiers | |
3D model (JSmol)
|
|
| 317313 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.807 |
| EC Number |
|
| 579210 | |
| KEGG | |
PubChem CID
|
|
| UNII |
|
| UN number | 2811 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H10O7 | |
| Molar mass | 302.236 g/mol |
| Appearance | yellow crystalline powder[1] |
| Density | 1.799 g/cm3 |
| Melting point | 316 °C (601 °F; 589 K) |
| Practically insoluble in water; soluble in aqueous alkaline solutions[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Quercetin is a plant flavonol from the flavonoid group of polyphenols. It is found in many fruits, vegetables, leaves, seeds, and grains; capers, red onions and kale are common foods containing appreciable amounts of quercetin.[2][3] Quercetin has a bitter flavor and is used as an ingredient in dietary supplements, beverages, and foods.
Occurrence
Quercetin is a flavonoid widely distributed in nature.[2] The name has been used since 1857, and is derived from quercetum (oak forest), after the oak genus Quercus.[4][5] It is a naturally occurring polar auxin transport inhibitor.[6]
Quercetin is one of the most abundant dietary flavonoids,[2][3] with an average daily consumption of 25–50 milligrams.[7]
| Foods | Quercetin (mg/100g) |
|---|---|
| capers, raw | 234[3] |
| capers, canned | 173[3] |
| lovage leaves, raw | 170[3] |
| buckwheat seeds | 90 |
| dock like sorrel | 86[3] |
| radish leaves | 70[3] |
| carob fiber | 58[3] |
| dill | 55[8] |
| cilantro | 53[3] |
| Hungarian wax pepper | 51[3] |
| fennel leaves | 49[3] |
| onion, red | 32[3] |
| radicchio | 32[3] |
| watercress | 30[3] |
| kale | 23[3] |
| chokeberry | 19[3] |
| bog blueberry | 18[3] |
| cranberry | 15[3] |
| lingonberry | 13[3] |
| plums, black | 12[3] |
In red onions, higher concentrations of quercetin occur in the outermost rings and in the part closest to the root, the latter being the part of the plant with the highest concentration.[9] One study found that organically grown tomatoes had 79% more quercetin than non-organically grown fruit.[10] Quercetin is present in various kinds of honey from different plant sources.[11]
Biosynthesis
In plants, phenylalanine is converted to 4-coumaroyl-CoA in a series of steps known as the general phenylpropanoid pathway using phenylalanine ammonia-lyase, cinnamate-4-hydroxylase, and 4-coumaroyl-CoA-ligase.[12] One molecule of 4-coumaroyl-CoA is added to three molecules of malonyl-CoA to form tetrahydroxychalcone using 7,2′-dihydroxy-4′-methoxyisoflavanol synthase. Tetrahydroxychalcone is then converted into naringenin using chalcone isomerase.
Naringenin is converted into eriodictyol using flavanoid 3′-hydroxylase. Eriodictyol is then converted into dihydroquercetin with flavanone 3-hydroxylase, which is then converted into quercetin using flavonol synthase.[12]
Glycosides

Quercetin is the aglycone form of a number of other flavonoid glycosides, such as rutin and quercitrin, found in citrus fruit, buckwheat and onions.[2] Quercetin forms the glycosides quercitrin and rutin together with rhamnose and rutinose, respectively. Likewise guaijaverin is the 3-O-arabinoside, hyperoside is the 3-O-galactoside, isoquercitin is the 3-O-glucoside and spiraeoside is the 4′-O-glucoside. CTN-986 is a quercetin derivative found in cottonseeds and cottonseed oil. Miquelianin is the quercetin 3-O-β-D-glucuronopyranoside.[13]
Rutin degradation pathway
The enzyme quercitrinase can be found in Aspergillus flavus.[14] This enzyme hydrolyzes the glycoside quercitrin to release quercetin and L-rhamnose. It is an enzyme in the rutin catabolic pathway.[15]
Pharmacology
Pharmacokinetics
The bioavailability of quercetin in humans is low and highly variable (0–50%), and it is rapidly cleared with an elimination half-life of 1–2 hours after ingesting quercetin foods or supplements.[16] Following dietary ingestion, quercetin undergoes rapid and extensive metabolism that makes the biological effects presumed from in vitro studies unlikely to apply in vivo.[17][18][19]
Quercetin supplements in the aglycone form are far less bioavailable than the quercetin glycoside often found in foods, especially red onions.[2][20] Ingestion with high-fat foods may increase bioavailability compared to ingestion with low-fat foods,[20] and carbohydrate-rich foods may increase absorption of quercetin by stimulating gastrointestinal motility and colonic fermentation.[2]
Metabolism
In rats, quercetin did not undergo any significant phase I metabolism.[21] In contrast, quercetin did undergo extensive phase II (conjugation) to produce metabolites that are more polar than the parent substance and hence are more rapidly excreted from the body. In vitro, the meta-hydroxyl group of catechol is methylated by catechol-O-methyltransferase. Four of the five hydroxyl groups of quercetin are glucuronidated by UDP-glucuronosyltransferase. The exception is the 5-hydroxyl group of the flavonoid ring which generally does not undergo glucuronidation. The major metabolites of orally absorbed quercetin are quercetin-3-glucuronide, 3'-methylquercetin-3-glucuronide, and quercetin-3'-sulfate.[21] A methyl metabolite of quercetin has been shown in vitro to be more effective than quercetin at inhibiting lipopolysaccharide-activated macrophages.[19]
Compared to other flavonoids quercetin is one of the most effective inducers of the phase II detoxification enzymes.[22]
Quercetin is a strong inhibitor of the cytochrome P450 enzymes CYP3A4 and CYP2D6.[23] Drugs that are metabolized by these pathways may have increased effect.
In vitro pharmacology
Quercetin has been reported to inhibit the oxidation of other molecules and hence is classified as an antioxidant in vitro.[17] It contains a polyphenolic chemical substructure that stops oxidation in vitro by acting as a scavenger of free radicals. Quercetin has been shown to inhibit the PI3K/AKT pathway leading to downregulation of the anti-apoptotic protein Bcl-w.[24][25]
Quercetin also activates or inhibits the activities of a number of proteins in vitro. For example, quercetin is a non-specific protein kinase enzyme inhibitor.[17] In human breast cancer cell lines, quercetin has also been found to act as an agonist of the G protein-coupled estrogen receptor (GPER).[26]
Health claims
Quercetin has been studied in basic research and small clinical trials.[2][27][28][29] While supplements have been promoted for the treatment of cancer and various other diseases,[2][30] there is no high-quality evidence that quercetin (via supplements or in food) is useful to treat cancer[31] or any other disease.[2][32]
The US Food and Drug Administration has issued warning letters to several manufacturers advertising on their product labels and websites that quercetin product(s) can be used to treat diseases.[33][34] The FDA regards such quercetin advertising and products as unapproved – with unauthorized health claims concerning the anti-disease products – as defined by "sections 201(g)(1)(B) and/or 201 (g)(1)(C) of the Act [21 U.S.C. § 321(g)(1)(B) and/or 21 U.S.C. § 321(g)(1)(C)] because they are intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease",[33][34] conditions which were not met by the manufacturers.
Safety
In preliminary human studies, oral intake of quercetin in doses up to one gram per day over three months did not cause adverse effects.[2] The safety of using quercetin in dietary supplements during pregnancy and lactation has not been established.[2]
See also
References
- ^ a b c "Quercetin dihydrate safety sheet". Archived from the original on September 16, 2011.
- ^ a b c d e f g h i j k "Flavonoids (Review)". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis, OR. November 2015. Retrieved 1 April 2018.
- ^ a b c d e f g h i j k l m n o p q r s t "USDA Database for the Flavonoid Content of Selected Foods, Release 3" (PDF). U.S. Department of Agriculture. 2011.
- ^ "Quercetin". Merriam-Webster.
- ^ "Quercetin (biochemistry)". Encyclopædia Britannica.
- ^ Fischer C, Speth V, Fleig-Eberenz S, Neuhaus G (Oct 1997). "Induction of Zygotic Polyembryos in Wheat: Influence of Auxin Polar Transport". The Plant Cell. 9 (10): 1767–1780. doi:10.1105/tpc.9.10.1767. PMC 157020. PMID 12237347.
- ^ Formica JV, Regelson W (1995). "Review of the biology of quercetin and related bioflavonoids". Food and Chemical Toxicology. 33 (12): 1061–80. doi:10.1016/0278-6915(95)00077-1. PMID 8847003.
- ^ Justesen U, Knuthsen P (May 2001). "Composition of flavonoids in fresh herbs and calculation of flavonoid intake by use of herbs in traditional Danish dishes". Food Chemistry. 73 (2): 245–50. doi:10.1016/S0308-8146(01)00114-5.
- ^ Slimestad R, Fossen T, Vågen IM (December 2007). "Onions: a source of unique dietary flavonoids". Journal of Agricultural and Food Chemistry. 55 (25): 10067–80. doi:10.1021/jf0712503. PMID 17997520.
- ^ Mitchell AE, Hong YJ, Koh E, Barrett DM, Bryant DE, Denison RF, Kaffka S (Jul 2007). "Ten-year comparison of the influence of organic and conventional crop management practices on the content of flavonoids in tomatoes". Journal of Agricultural and Food Chemistry. 55 (15): 6154–9. doi:10.1021/jf070344+. PMID 17590007.
- ^ Petrus K, Schwartz H, Sontag G (Jun 2011). "Analysis of flavonoids in honey by HPLC coupled with coulometric electrode array detection and electrospray ionization mass spectrometry". Analytical and Bioanalytical Chemistry. 400 (8): 2555–63. doi:10.1007/s00216-010-4614-7. PMID 21229237. S2CID 24796542.
- ^ a b Winkel-Shirley B (Jun 2001). "Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology". Plant Physiology. 126 (2): 485–93. doi:10.1104/pp.126.2.485. PMC 1540115. PMID 11402179.
- ^ Juergenliemk G, Boje K, Huewel S, Lohmann C, Galla HJ, Nahrstedt A (Nov 2003). "In vitro studies indicate that miquelianin (quercetin 3-O-beta-D-glucuronopyranoside) is able to reach the CNS from the small intestine". Planta Medica. 69 (11): 1013–7. doi:10.1055/s-2003-45148. PMID 14735439.
- ^ "Information on EC 3.2.1.66 - quercitrinase". BRENDA (BRaunschweig ENzyme DAtabase). Helmholtz Centre for Infection Research.
- ^ Tranchimand S, Brouant P, Iacazio G (Nov 2010). "The rutin catabolic pathway with special emphasis on quercetinase". Biodegradation. 21 (6): 833–59. doi:10.1007/s10532-010-9359-7. PMID 20419500. S2CID 30101803.
- ^ Graefe EU, Derendorf H, Veit M (1999). "Pharmacokinetics and bioavailability of the flavonol quercetin in humans" (PDF). (review). International Journal of Clinical Pharmacology and Therapeutics. 37 (5): 219–33. PMID 10363620.
- ^ a b c Williams RJ, Spencer JP, Rice-Evans C (Apr 2004). "Flavonoids: antioxidants or signalling molecules?". (review). Free Radical Biology & Medicine. 36 (7): 838–49. doi:10.1016/j.freeradbiomed.2004.01.001. PMID 15019969.
- ^ Barnes S, Prasain J, D'Alessandro T, Arabshahi A, Botting N, Lila MA, Jackson G, Janle EM, Weaver CM (May 2011). "The metabolism and analysis of isoflavones and other dietary polyphenols in foods and biological systems". (review). Food & Function. 2 (5): 235–44. doi:10.1039/c1fo10025d. PMC 4122511. PMID 21779561.
- ^ a b Luca SV, Macovei I, Bujor A, Trifan A (2020). "Bioactivity of dietary polyphenols: The role of metabolites". Critical Reviews in Food Science and Nutrition. 60 (4): 626–659. doi:10.1080/10408398.2018.1546669. PMID 30614249. S2CID 58651581.
- ^ a b Dabeek WM, Marra MV (2019). "Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans". Nutrients. 11 (10): 2288. doi:10.3390/nu11102288. PMC 6835347. PMID 31557798.
- ^ a b Day AJ, Rothwell JA, Morgan R (2004). "Characterization of polyphenol metabolites". In Bao Y, Fenwick R (eds.). Phytochemicals in health and disease. New York, NY: Dekker. pp. 50–67. ISBN 0-8247-4023-8.
- ^ Procházková D, Boušová I, Wilhelmová N (2011). "Antioxidant and prooxidant properties of flavonoids". Fitoterapia. 82 (4): 513–523. doi:10.1016/j.fitote.2011.01.018. PMID 21277359.
- ^ Elbarbry, Fawzy; Ung, Aimy; Abdelkawy, Khaled (2017). "Studying the Inhibitory Effect of Quercetin and Thymoquinone on Human Cytochrome P450 Enzyme Activities". Pharmacognosy Magazine. 13 (Suppl 4): S895–S899. doi:10.4103/0973-1296.224342 (inactive 2021-09-18). ISSN 0973-1296. PMC 5822518. PMID 29491651.
{{cite journal}}: CS1 maint: DOI inactive as of September 2021 (link) CS1 maint: unflagged free DOI (link) - ^ Hartman ML, Czyz M (2020). "BCL-w: apoptotic and non-apoptotic role in health and disease". Cell Death & Disease. 11 (4): 2260. doi:10.1038/s41419-020-2417-0. PMC 7174325. PMID 32317622.
- ^ Paez-Ribes M, González-Gualda E, Doherty GJ, Muñoz-Espín D (2019). "Targeting senescent cells in translational medicine". EMBO Molecular Medicine. 11 (12): e10234. doi:10.15252/emmm.201810234. PMC 6895604. PMID 31746100.
- ^ Prossnitz ER, Barton M (May 2014). "Estrogen biology: new insights into GPER function and clinical opportunities". (review). Molecular and Cellular Endocrinology. 389 (1–2): 71–83. doi:10.1016/j.mce.2014.02.002. PMC 4040308. PMID 24530924.
- ^ Yang F, Song L, Wang H, Wang J, Xu Z, Xing N (June 2015). "Quercetin in prostate cancer: Chemotherapeutic and chemopreventive effects, mechanisms and clinical application potential (Review)". Oncol. Rep. 33 (6): 2659–68. doi:10.3892/or.2015.3886. PMID 25845380.
- ^ Gross P (March 1, 2009), New Roles for Polyphenols. A 3-Part Report on Current Regulations & the State of Science, Nutraceuticals World
- ^ Miles SL, McFarland M, Niles RM (2014). "Molecular and physiological actions of quercetin: need for clinical trials to assess its benefits in human disease". Nutrition Reviews. 72 (11): 720–34. doi:10.1111/nure.12152. PMID 25323953.
- ^ D'Andrea G (2015). "Quercetin: A flavonol with multifaceted therapeutic applications?". Fitoterapia. 106: 256–71. doi:10.1016/j.fitote.2015.09.018. PMID 26393898.
- ^ Ades TB, ed. (2009). Quercetin (2nd ed.). American Cancer Society. ISBN 9780944235713.
{{cite book}}:|work=ignored (help) - ^ European Food Safety Agency (EFSA) NDA Panel (Dietetic Products, Nutrition and Allergies) (8 April 2011). "Scientific Opinion on the substantiation of health claims related to quercetin and protection of DNA, proteins and lipids from oxidative damage (ID 1647), "cardiovascular system" (ID 1844), "mental state and performance" (ID 1845), and "liver, kidneys" (ID 1846) pursuant to Article 13(1) of Regulation (EC) No 1924/2006". EFSA Journal. 9 (4): 2067–82. doi:10.2903/j.efsa.2011.2067. Retrieved 24 September 2014.
- ^ a b Janice L King (2 March 2017). "Warning Letter to Cape Fear Naturals". Inspections, Compliance, Enforcement, and Criminal Investigations, US Food and Drug Administration. Retrieved 29 November 2018.
- ^ a b Ronald Pace (17 April 2017). "Warning Letter to DoctorVicks.com". Inspections, Compliance, Enforcement, and Criminal Investigations, US Food and Drug Administration. Retrieved 29 November 2018.
External links
 Media related to Quercetin at Wikimedia Commons
Media related to Quercetin at Wikimedia Commons
