Cannabichromene
| |
| Names | |
|---|---|
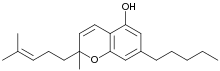
| IUPAC name
2-Methyl-2-(4-methylpent-3-enyl)-7-pentyl-5-chromenol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.236.929 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H30O2 | |
| Molar mass | 314.469 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cannabichromene (CBC), also called cannabichrome, cannanbichromene, pentylcannabichromene or cannabinochromene,[1] is one of the 120 cannabinoids found in the Cannabis plant,[2] and is therefore a phytocannabinoid. It bears structural similarity to the other natural cannabinoids, including tetrahydrocannabinol (THC), tetrahydrocannabivarin (THCV), cannabidiol (CBD), and cannabinol (CBN), among others.[2][3] Cannabichromene and its derivatives are as abundant as cannabinols in cannabis.[2] It is not active at CB1 or CB2 receptors, but is an agonist of TRPA1 and less potently, an agonist of TRPV3 and TRPV4.[2] CBC has two stereoisomers. It is not scheduled by the Convention on Psychotropic Substances.
Biosynthesis
Within the Cannabis plant, CBC occurs mainly as cannabichromenic acid (CBCA, 2-COOH-CBC, CBC-COOH). Geranyl pyrophosphate and olivetolic acid combine to produce cannabigerolic acid (CBGA; the sole intermediate for all other phytocannabinoids), which is cyclized by the enzyme CBCA synthase to form CBCA. Over time, or when heated somewhere above 200° F, CBCA is decarboxylated, producing CBC. See also the biosynthetic scheme image below.[citation needed]

Pharmacology
As of 2017, CBC is under laboratory research to identify its possible pharmacological properties. No in vivo human studies exist yet.[2][4][5][6][7][8][9][10][11][12]
References
- ^ "Cannabichromene". PubChem. National Center for Biotechnology Information. 2019-02-04. Retrieved 12 February 2019.
- ^ a b c d e Turner, Sarah E.; Williams, Claire M.; Iversen, Leslie; Whalley, Benjamin J. (2017). "Molecular Pharmacology of Phytocannabinoids". In Kinghorn, A. Douglas; Falk, Heinz; Gibbons, Simon; Kobayashi, Jun'ichi (eds.). Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa. Progress in the Chemistry of Organic Natural Products. Vol. 103. Springer International Publishing. pp. 61–101. doi:10.1007/978-3-319-45541-9_3. ISBN 978-3-319-45539-6. PMID 28120231.
- ^ Aizpurua-Olaizola, Oier; Soydaner, Umut; Öztürk, Ekin; Schibano, Daniele; Simsir, Yilmaz; Navarro, Patricia; Etxebarria, Nestor; Usobiaga, Aresatz (2016). "Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes". Journal of Natural Products. 79 (2): 324–331. doi:10.1021/acs.jnatprod.5b00949. PMID 26836472.
- ^ Morales, Paula; Hurst, Dow P.; Reggio, Patricia H. (2017). "Molecular Targets of the Phytocannabinoids: A Complex Picture". In Kinghorn, A. Douglas; Falk, Heinz; Gibbons, Simon; Kobayashi, Jun'ichi (eds.). Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa. Progress in the Chemistry of Organic Natural Products. Vol. 103. Springer International Publishing. pp. 103–131. doi:10.1007/978-3-319-45541-9_4. ISBN 978-3-319-45539-6. PMC 5345356. PMID 28120232.
- ^ Delong, G. T.; Wolf, C. E.; Poklis, A.; Lichtman, A. H. (2010). "Pharmacological evaluation of the natural constituent of Cannabis sativa, cannabichromene and its modulation by Δ(9)-tetrahydrocannabinol". Drug and Alcohol Dependence. 112 (1–2): 126–33. doi:10.1016/j.drugalcdep.2010.05.019. PMC 2967639. PMID 20619971.
- ^ https://www.ncbi.nlm.nih.gov/pubmed/22300105
- ^ https://www.ncbi.nlm.nih.gov/pubmed/23373571
- ^ https://www.ncbi.nlm.nih.gov/pubmed/20942863
- ^ https://www.ncbi.nlm.nih.gov/pubmed/25311884
- ^ https://www.ncbi.nlm.nih.gov/pubmed/20118579
- ^ https://www.ncbi.nlm.nih.gov/pubmed/22300105
- ^ https://www.ncbi.nlm.nih.gov/pubmed/26197538
